‘Prana’ is a Hindu Sanskrit word meaning “life force”.
An Australian biotech company has chosen this word for their name.
Recently Prana Biotechnology Ltd announced some exciting results from their Parkinson’s disease research programme.
In today’s post we will look at what the company is doing, the science underlying the business plan, and review the results they have so far.

Source: ADPD2017
At the end of March, over 3000 researchers in the field of neurodegeneration gathered in the Austrian capital of Vienna for the 13th International Conference on Alzheimer’s and Parkinson’s Diseases and Related Neurological Disorders (also known as ADPD2017).

The Vienna city hall. Source: EUtourists
A lot of interesting new research in the field of Parkinson’s disease was presented at the conference (we will look at some other presentation in future posts), but one was of particular interest to us here at SoPD HQ.
The poster entitled: ‘Abstract: 104 – PBT434 prevents neuronal loss, motor function and cognitive impairment in preclinical models of movement disorders by modulation of intracellular iron’, was presented by Associate Professor David Finkelstein, of the Florey Institute of Neuroscience and Mental Health (Melbourne, Australia).
Unfortunately the ADPD2017 conference’s scientific programme search engine does not allow for individual abstracts to be linked to on the web so if you would like to read the abstract, you will need to click here for the search engine page and search for ‘PBT434’ or ‘Finkelstein’ in the appropriate boxes.
Prof Finkelstein was presenting preclinical research that had been conducted by an Australian biotech company called Prana Biotechnology Ltd.

Source: Prana Biotechnology Ltd
What does the company do?
Prana Biotechnology Ltd has a large portfolio of over 1000 small chemical agents that they have termed ‘MPACs’ (or Metal Protein Attenuating Compounds). These compounds are designed to interrupt the interactions between particular metals and target proteins in the brain. The goal of this interruption is to prevent deterioration of brain cells in neurodegenerative conditions.
For Parkinson’s disease, the company is proposing a particular iron chelator they have called PBT434.
What is an iron chelator?
Iron chelator therapy involves the removal of excess iron from the body with special drugs. Chelate is from the Greek word ‘chela’ meaning “claw”.

Chelator therapy. Source: Stanford
Iron overload in the body is a common medical problem, sometimes arising from disorders of increased iron absorption such as hereditary haemochromatosis. Iron chelator therapy represents one method of reducing the levels of iron in the body.
But why is iron overload a problem?

Iron. Source: GlobalSpec
Good question. It involves the basic properties of iron.
Iron is a chemical element (symbol Fe). It has the atomic number 26 and by mass it is the most common element on Earth (it makes up much of Earth’s outer and inner core). It is absolutely essential for cellular life on this planet as it is involved with the interactions between proteins and enzymes, critical in the transport of oxygen, and required for the regulation of cell growth and differentiation.
So why then – as Rosalind asked in Shakespeare’s As You Like It – “can one desire too much of a good thing?”
Well, if you think back to high school chemistry class you may recall that there are these things called electrons. And if you have a really good memory, you will recall that the chemical hydrogen has one electron, while iron has 26 (hence the atomic number 26).
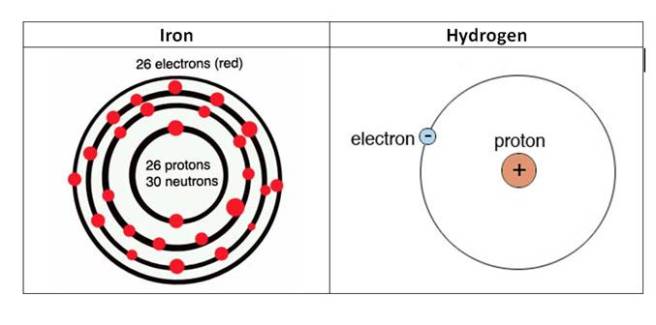
The electrons of iron and hydrogen. Source: Hypertonicblog
Iron has a really interesting property: it has the ability to either donate or take electrons. And this ability to mediate electron transfer is one of the reasons why iron is so important in the body.
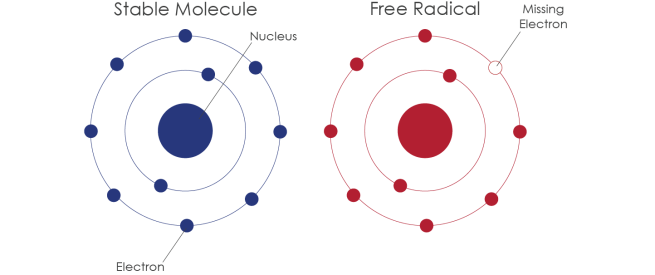
Iron’s ability to donate and accept electrons means that when there is a lot of iron present it can inadvertently cause the production of free radicals. We have previously discussed free radicals (Click here for that post), but basically a free radical is an unstable molecule – unstable because they are missing electrons.

How free radicals and antioxidants work. Source: h2miraclewater
In an unstable format, free radicals bounce all over the place, reacting quickly with other molecules, trying to capture the much needed electron to re-gain stability. Free radicals will literally attack the nearest stable molecule, to steal an electron. This leads to the “attacked” molecule becoming a free radical itself, and thus a chain reaction is started. Inside a living cell this can cause terrible damage, ultimately killing the cell.
Antioxidants can help try and restore the balance, but in the case of iron overload iron doctors will prescribe chelator treatment to deal with the situation more efficiently. By soaking up excess iron, we can limit the amount of damage caused by the surplus of iron.
So what research has been done regarding iron content and the Parkinsonian brain?
Actually, quite a lot.
In 1968, Dr Kenneth Earle used an X-ray based technique to examine the amount of iron in the substantia nigra of people with Parkinson’s disease (Source). The substantial nigra is one of the regions in the brain most badly damaged by the condition – it is where most of the brain’s dopamine neurones resided.
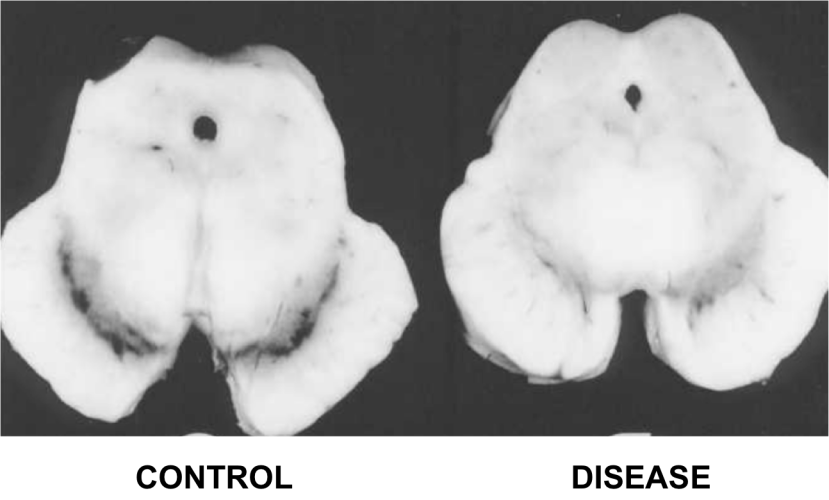
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinson’s disease brain (right). Source:Memorangapp
Earle examined 11 samples and compared them to unknown number of control samples and his results were a little startling:
The concentration of iron in Parkinsonian samples was two times higher than that of the control samples.
Since that first study, approximately 30 investigations have been made into levels of iron in the Parkinsonian brain. Eleven of those studies have replicated the Earle study by looking at postmortem tissue. They have used different techniques and the results have varied somewhat:
- Sofic et al. (1988) 1.8x increase in iron levels
- Dexter et al. (1989) 1.3x increase in iron levels
- Uitti et al. (1989) 1.1x increase in iron levels
- Riederer et al 1989 1.3x increase in iron levels
- Griffiths and Crossman (1993) 2.0x increase in iron levels
- Mann et al. (1994) 1.6x increase in iron levels
- Loeffler et al. (1995) 0.9 (lower)
- Galazka-Friedman et al., 1996 1.0 (no difference)
- Wypijewska et al. (2010) 1.0 (no difference)
- Visanji et al, 2013 1.7x increase in iron levels
Overall, however, there does appear to be a trend in the direction of higher levels of iron in the Parkinsonian brains. A recent meta-analysis of all this data confirmed this assessment as well as noting an increase in the caudate putamen (the region of the brain where the dopamine neuron branches release their dopamine – Click here for that study).
Brain imaging of iron (using transcranial sonography and magnetic resonance imaging (MRI)) has also demonstrated a strong correlation between iron levels in the substantia nigra region and Parkinson’s disease severity/duration (Click here and here to read more on this).
Thus, there appears to be an increase of iron in the regions most affected by Parkinson’s disease and this finding has lead researchers to ask whether reducing this increase in iron may help in the treatment of Parkinson’s disease.
How could iron overload be bad in Parkinson’s disease?
Well in addition to causing the production of free radicals, there are many possible ways in which iron accumulation could be aggravating cell loss in Parkinson’s disease.

Possible causes and consequences of iron overload in Parkinson’s disease. Source: Hindawi
High levels of iron can cause the oxidation of dopamine, which results in the production of hydrogen peroxide (H2O2 – a reactive oxygen species – the stuff that is used to bleach hair and is also used as a propellant in rocketry!). This reaction can cause further oxidative stress that can then lead to a range of consequences including protein misfolding, lipid peroxidation (which can cause the accumulation of the Parkinson’s associated protein alpha synuclein), mitochondrial dysfunction, and activation of immune cells in the brain.
And this is just a taster of the consequences.
For further reading on this topic we recommend two very good reviews – click here and here.
Ok, so iron overload is bad, but what was the research presented in Austria?
The abstract:
Title: PBT434 prevents neuronal loss, motor function and cognitive impairment in preclinical models of movement disorders by modulation of intracellular iron
Authors: D. Finkelstein, P. Adlard, E. Gautier, J. Parsons, P. Huggins, K. Barnham, R. Cherny
Location: C01.a Posters – Theme C – Alpha-Synucleinopathies
The researchers at Prana Biotechnology Ltd assessed the potential of one of their candidate drugs, PBT434, in both cell culture and animal models of Parkinson’s disease. The PBT434 drug was selected for further investigation based on its performance in cell culture assays designed to test the inhibition of oxidative stress and iron-mediated aggregation of Parkinson’s associated proteins like alpha synuclein.
PBT434 significantly reduced the accumulation of alpha synuclein and markers of oxidative stress, and prevented neuronal loss.
The investigators also demonstrated that orally administered PBT434 readily crossed the blood brain barrier and entered the brain. In addition the drug was well-tolerated in the experimental animals and improved motor function in toxin-induced (MPTP and 6-hydroxydopamine) and transgenic mouse models of Parkinson’s disease (alpha synuclein -A53T and tau – rTg4510).
These results are in agreement with previous studies that have looked at iron chelator therapy in models of Parkinson’s disease (Click here, here and here for some examples)
Interestingly, PBT434 also demonstrated neuroprotective properties in animal models of multiple systems atrophy (or MSA). Suggesting that perhaps iron chelation could be a broad neuroprotective approach.
The researchers concluded that this preclinical data demonstrates the efficacy of PBT434 as a clinical candidate for Parkinson’s disease. PBT434 shows a strong toxicology profile and favourable therapeutic activity. Prana is preparing its pre-clinical development package for PBT434 to initiate human clinical trials.
Does Prana have any other drugs in clinical trials?
Yes, they do.

Source: Prana
Prana Biotechnology has another product called PBT2.
The company currently has two clinical trial programs for PBT2 focused on two other neurodegenerative diseases: Alzheimer’s disease and Huntington’s disease.
The Alzheimer’s study was called the IMAGINE Trial, but (there is always a ‘but’) recently PBT2 failed to meet its primary endpoint (significantly reducing levels of beta-amyloid – the perceived bad guy in Alzheimer’s disease) in a phase III trial of mild Alzheimer’s disease. PBT2 was, however, shown to be safe and very well tolerated over the 52 week trial, with no difference in the occurrence of adverse events between the placebo and treated groups.
In addition, there was less atrophy (shrinkage) in the brains of those patients treated with PBT2 when compared to control brains, 2.6% and 4.0%, respectively (based on brain imaging). The company is tracking measures of brain volume and cognition in a 12 month extension study. It could be interesting to continue that follow up long term to evaluate the consequences of long term use of this drug on Alzheimer’s disease – even if the effect is minimal, any drug that can slow the disease down is useful and could be used in conjunction with other neuroprotective medications.
For Huntington’s disease, the company is also using the PBT2 drug and this study has had a bit more success. The study, called Reach2HD, was a six month phase II clinical trial in 109 patients with early to mid-stage Huntington’s disease, across 20 sites in the US and Australia. The company was aiming to assess the safety profile of this drug in this particular condition, as well as determining the motor and behavioural benefits.
In the ReachHD study, PBT2 showed signs of improving some aspects of cognitive function in the study, which potentially represents a major event for a disease for which there is very little in the way of medical treatments.
For a full description of the PBT2 trials, see this wikipedia page on the topic.
Is Prana the only research group working on iron chelators technology for Parkinson’s disease?
No.
There is a large EU-based consortium called FAIR PARK II, which is running a five year trial (2015 – 2020) of the iron chelator deferiprone (also known as Ferriprox). The study is a multi-centre, placebo-controlled, randomised clinical trial involving 338 people with recently diagnosed Parkinson’s disease.

The population will be divided into two group (169 subjects each). They will then be assigned either deferiprone (15 mg/kg twice a day) or a placebo. Each subject will be given 9-months of treatment followed by a 1-month post-treatment monitoring period, in order to assess the disease-modifying effect of deferiprone (versus placebo).
Deferiprone. Source: SGPharma
As far as we are aware, this FAIR PARK II clinical trial is still recruiting participants – please click here to read more about this – thus it will most likely be some time before we hear the results of this study.
Are there natural sources of chelators?
Yes there are. In fact, many natural antioxidants exert some chelating activities.
Prominent among the natural sources of chelators: Green tea has components of plant extracts, such as epigallocatechin gallate (EGCG – which we have previously discussed in regards to Parkinson’s disease, click here to read that post) which possess structures which infer metal chelating properties.
As we have said before people, drink more green tea!

Anyone fancy a cuppa? Source: Expertrain
So what does it all mean?
Summing up: We do not know what causes Parkinson’s disease. Most of our experimental treatments are focused on the biological events that occur in the brain around and after the time of diagnosis. These include an apparent accumulation of iron in affected brain regions.
Research groups are currently experimenting with drugs that reduce the levels of iron in the brain as a potential treatment for Parkinson’s disease. Preclinical data certainly look positive. We will now have to wait and see if those results translate into the human.
Previous clinical trials of metal chelators in neurodegeneration have had mixed success in demonstrating positive benefits. It may well be, however, that this treatment approach should be used in conjunction with other neuroprotective approaches – as a supplement. It will be interesting to see how Prana Biotechnology’s drug PBT434 fares in human clinical trials for Parkinson’s disease.
Stay tuned for more on this.
UPDATE – 3rd May 2017
Today the results of a double-blind, phase II clinical trial of iron chelator deferiprone in Parkinson’s disease were published. The results of the study indicate a mildly positive effect (though not statistically significant) after 6 months of daily treatment.

Title: Brain iron chelation by deferiprone in a phase 2 randomised double-blinded placebo controlled clinical trial in Parkinson’s disease
Authors: Martin-Bastida A, Ward RJ, Newbould R, Piccini P, Sharp D, Kabba C, Patel MC, Spino M, Connelly J, Tricta F, Crichton RR & Dexter DT
Journal: Scientific Reports (2017), 7, 1398.
PMID: 28469157 (This article is OPEN ACCESS if you would like to read it)
In this Phase 2 randomised, double-blinded, placebo controlled clinical trial, the researchers recruited 22 people with early stage Parkinson’s disease (disease duration of less than 5 years; 12 males and 10 females; aged 50–75 years). They were randomly assigned to either a placebo group (8 participants), or one of two deferiprone treated groups: 20 mg/kg per day (7 participants) or 30 mg/kg per day (7 participants). The treatment was two daily oral doses (taken morning and evening), and administered for 6 months with neurological examinations, brain imaging and blood sample collections being conducted at 0, 3 and 6 months.
Deferiprone therapy was well tolerated and brain imaging indicated clearance of iron from various parts of the brain in the treatment group compared to the placebo group. Interestingly, the 30 mg/kg deferiprone treated group demonstrated a trend for improvement in motor-UPDRS scores and quality of life (although this was not statistically significance). The researchers concluded that “more extensive clinical trials into the potential benefits of iron chelation in PD”.
Given the size of the groups (7 people) and the length of the treatment period (only 6 months) in this study it is not really a surprise that the researchers did not see a major effect. That said, it is very intriguing that they did see a trend towards motor score benefits in the 30 mg/kg deferiprone group – remembering that this is a double blind study (so even the investigators were blind as to which group the subjects were in).
We will now wait to see what the FAIR PARK II clinical trial finds.
UPDATE: 28th June 2017
Today, the research that Prana biotechnology Ltd was presenting in Vienna earlier this year was published:

Title: The novel compound PBT434 prevents iron mediated neurodegeneration and alpha-synuclein toxicity in multiple models of Parkinson’s disease.
Authors: Finkelstein DI, Billings JL, Adlard PA, Ayton S, Sedjahtera A, Masters CL, Wilkins S, Shackleford DM, Charman SA, Bal W, Zawisza IA, Kurowska E, Gundlach AL, Ma S, Bush AI, Hare DJ, Doble PA, Crawford S, Gautier EC, Parsons J, Huggins P, Barnham KJ, Cherny RA.
Journal: Acta Neuropathol Commun. 2017 Jun 28;5(1):53.
PMID: 28659169 (This article is OPEN ACCESS if you would like to read it)
The results suggest that PBT434 is far less potent than deferiprone or deferoxamine at lowering cellular iron levels, but this weakness is compensated by the reduced levels of alpha synuclein accumulation in models of Parkinson’s disease. PBT434 certainly appears to be neuroprotective demonstrating improvements in motor function, neuropathology and biochemical markers of disease state in three different animal models of Parkinson’s disease.
The researchers provide little information as to when the company will be exploring clinical trials for this drug, but in the press release associated with the publication, Dr David Stamler (Prana’s Chief Medical Officer and Senior Vice President, Clinical Development) was quoted saying that they “are eager to begin clinical testing of PBT434”. We’ll keep an eye to the ground for any further news.
FULL DISCLOSURE: Prana Biotechnology Ltd is an Australasian biotechnology company that is publicly listed on the ASX. The information presented here is for educational purposes. Under no circumstances should investment decisions be made based on the information provided here. The SoPD website has no financial or beneficial connection to either company. We have not been approached/contacted by the company to produce this post, nor have we alerted them to its production. We are simply presenting this information here as we thought the science of what the company is doing might be of interest to other readers.
In addition, under absolutely no circumstances should anyone reading this material consider it medical advice. The material provided here is for educational purposes only. Before considering or attempting any change in your treatment regime, PLEASE consult with your doctor or neurologist. Metal chelators are clinically available medications, but it is not without side effects (for more on this, see this website). We urge caution and professional consultation before altering a treatment regime. SoPD can not be held responsible for any actions taken based on the information provided here.
The banner for today’s post was sourced from Prana








 Acetylcysteine. Source:
Acetylcysteine. Source: