|
Genetic mutations (or ‘variants’) in the Leucine-rich repeat kinase 2 (or LRRK2; also known as Dardarin) gene are associated with increased risk of Parkinson’s. As a result this gene has become the focus of a lot of genetic research. But what about LRRK2’s less well-known, rather neglected sibling LRRK1? In today’s post, we will look at new research that suggests the LRRK siblings could both be involved with Parkinson’s disease. |
I recommend to the reader that today’s post should be read with the following music playing in the background:
Inspired by a poem of the same title, English composer Ralph Vaughan Williams wrote ‘The Lark Ascending’ in 1914. It is still to this day, a tune that remains a firm favourite with BBC listeners here in the UK (Source).
On to business:
While the music and the poem are about a songbird, today’s SoPD post deals with a different kind of Lark.
Or should I say LRRK.
This is Sergey Brin.

Nice guy.
He was one of the founders of a small company you may have heard of – it’s called “Google”.
Having changed the way the world searches the internet, he is now turning his attention to other projects.
One of those other projects is close to our hearts: Parkinson’s disease.
In 1996, Sergey’s mother started experiencing numbness in her hands. Initially it was believed to be a bit of RSI (Repetitive strain injury). But then her left leg started to drag. In 1999, following a series of tests and clinical assessments, Sergey’s mother was diagnosed with Parkinson’s disease. It was not the first time the family had been affected by the condition though: Sergey’s late aunt had also had Parkinson’s disease.
Given this coincidental family history of this particular condition, both Sergey and his mother decided to have their DNA scanned for genetic mutations that increase the risk of Parkinson’s disease. And they discovered that they were both carrying a genetic variation on their 12th chromosome, in a gene called PARK8 – one of the Parkinson’s disease associated genes (Click here to read more about the genetics of Parkinson’s). Autosomal dominant mutations (meaning if you have just one copy of the altered gene) in the PARK8 gene dramatically increase one’s risk of developing Parkinson’s disease.
PARK8 provides the instructions for making an enzyme called Leucine-rich repeat kinase 2 (or LRRK2).
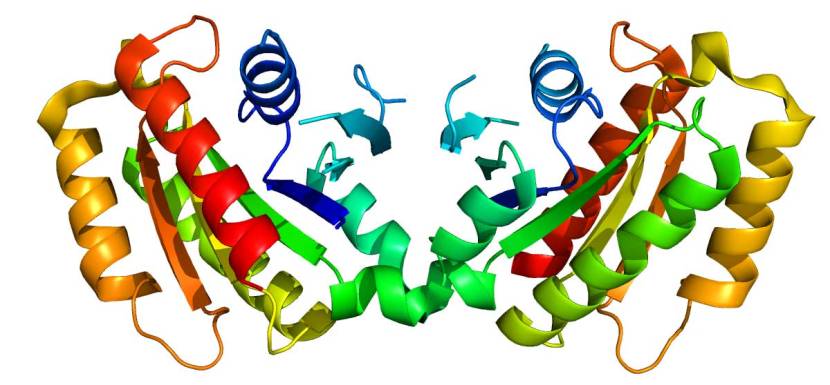
The structure of Lrrk2. Source: Wikipedia
Also known as ‘Dardarin‘ (from the Basque word “dardara” which means ‘trembling’), LRRK2 has many functions within a cell – from helping to move things around inside the cell to helping to keep the power on (involved with mitochondrial function).
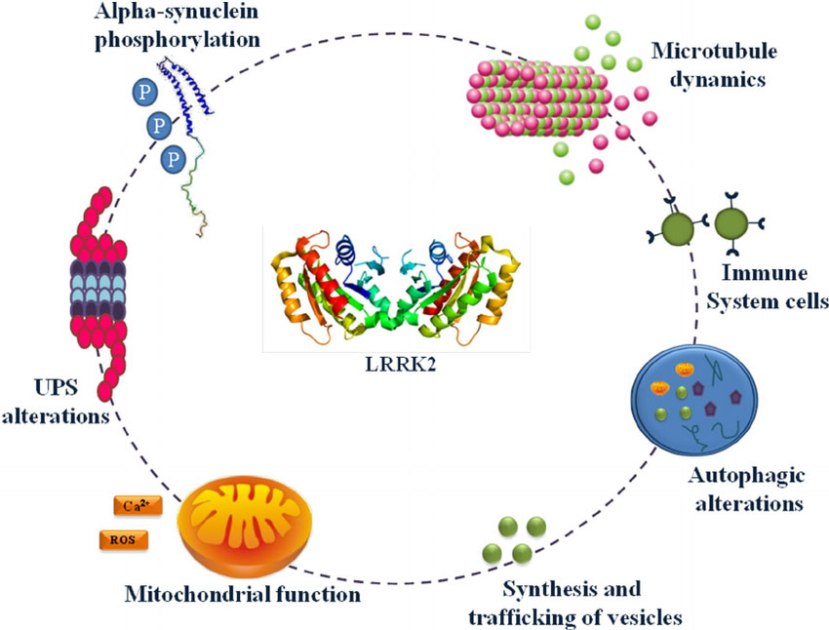
Source: Researchgate
The LRRK2 enzyme is made up of many different regions, each of which are involved with these different functions of the protein highlighted above.
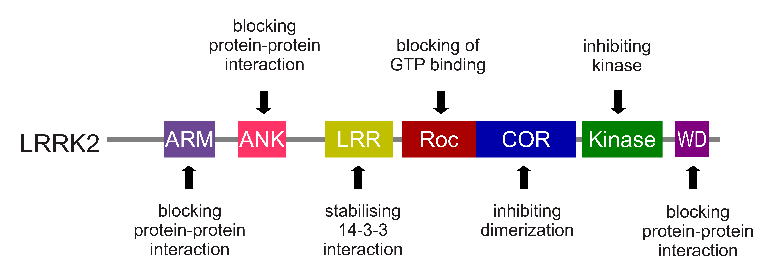
The regions and associated functions of the LRRK2 gene. Source: Intechopen
Genetic variants within the LRRK2 gene result in some of these regions being unable to do their function, or in other cases doing their function too much. Below are some of the mutations associated with the human LRRK2 gene:
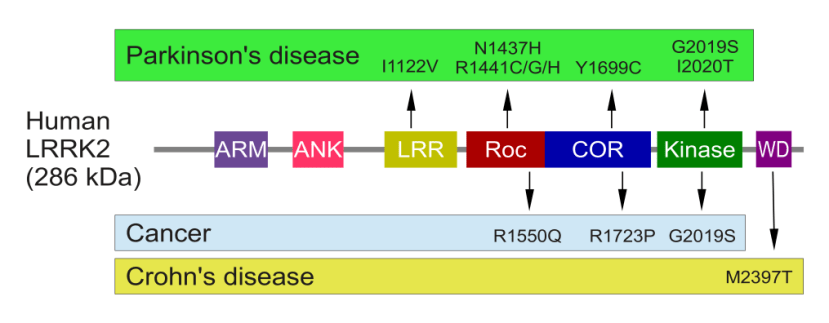
The structure of Lrrk2 and where various mutations lie. Source: Intech
As the image above suggests, one mutation in the LRRK2 gene is associated with Crohn’s disease (Click here and here for more on this), but that mutation is in a different location to those associated with Parkinson’s disease and it is not associated with an increased risk of developing Parkinson’s. And one particularly common Parkinson’s-associated LRRK2 mutation, however, called ‘G2019S’ is associated with an increased risk of certain types of cancer, especially for hormone-related cancer and breast cancer in women – Click here to read more about this. If you have a G2019S mutation, it is good to be aware of this association and have regular check ups (just to be safe).
The G2019S mutation (the name designates its location on the gene) is the most common LRRK2 variant. In some populations of people it can be found in 40% of people with Parkinson’s (Click here to read more about this). But what is interesting about this particular mutation is that it gives rise to a LRRK2 enzyme that is extremely hyperactive.
And it is this hyperactive behaviour of the protein that is believed to be involved with the cellular dysfunction seen in LRRK2-associated Parkinson’s. This hyperactive version of LRRK2 has been the topic of a great deal of research – some of which is currently out of this world (Click here to read more about this) – and pharmaceutical companies have been developing LRRK2 inhibitors with the goal of using them to treat people with Parkinson’s (Click here to read more about this).
And this is where Mr Brin comes back into the picture.
You see: Sergey Brin and his mother have the G2019S variant.
Now, to be very clear: Not everyone with this LRRK2 variant will go on to develop Parkinson’s disease. But Sergey has decided that his chances are 50:50. And he does not appear to be taking any chances. Being one of the founders of a large company like Google has left Sergey with considerable resources at his disposal. And he has chosen to focus some of those resources on LRRk2 research (call it an insurance policy). He has done this via considerable donations to groups like the Michael J Fox foundation (he gives approximately $20 million per year in donations to the MJFF).
Actor Michael J Fox was diagnosed at age 30. Source: MJFox foundation
Ok, so LRRK2 may be a problem, but what about it’s sibling LRRK1 you mentioned above? What do we know about it?
Several years ago, this research report was published:
Title: Rare variants in LRRK1 and Parkinson’s disease
Authors: Schulte EC, Ellwanger DC, Dihanich S, Manzoni C, Stangl K, Schormair B, Graf E, Eck S, Mollenhauer B, Haubenberger D, Pirker W, Zimprich A, Brücke T, Lichtner P, Peters A, Gieger C, Trenkwalder C, Mewes HW, Meitinger T, Lewis PA, Klünemann HH, Winkelmann J.
Journal: Neurogenetics. 2014 Mar;15(1):49-57.
PMID: 24241507 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers conducted genetic sequencing on two affected individuals from a Southern German family that was badly affected by late-onset Parkinson’s. From their analysis they identified 15 potentially genetic variants. They next search available genetic datasets from 862 cases of Parkinson’s and 1,014 ethnically matched controls. Of the 15 genetic variants identified in the original family, two were found to also be present in other individuals affected by Parkinson’s: EEF1D and LRRK1.
And that is not a typo.
It is supposed to be LRRK1, not LRRK2.
The researchers next wanted to conduct some functional analysis on the LRRK1 genetic variants that they had found. They genetically engineered cells that carried the LRRK1 genetic variants, but the investigators found that the genetic mutations did not affect the overall survival of the cells.
This was not the first time LRRK1 had popped up on the radar. Several previous studies had also looked at it in the context of Parkinson’s disease (Click here and here to read those reports).
And this next research report actually found that LRRK1 and LRRK2 interact directly:
Title: Heterodimerization of Lrrk1-Lrrk2: Implications for LRRK2-associated Parkinson disease.
Authors: Dachsel JC, Nishioka K, Vilariño-Güell C, Lincoln SJ, Soto-Ortolaza AI, Kachergus J, Hinkle KM, Heckman MG, Jasinska-Myga B, Taylor JP, Dickson DW, Gibson RA, Hentati F, Ross OA, Farrer MJ.
Journal: Mech Ageing Dev. 2010 Mar;131(3):210-4.
PMID: 20144646 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers first looked at where in the human brain LRRK1 and LRRK2 could be found. Their results from postmortem specimens revealed that in most areas of the brain LRRK1 and LRRK2 levels were largely the same (except in the cortex where LRRK1 was higher). Curiously, in the midbrain region where the dopamine producing cells reside, levels ofLRRK1 and LRRK2 were generally low.
Next they investigated whether there was a specific interaction between LRRK1 and LRRK2. They wanted to know if the two protein work together since they are found in the same regions. Their results suggested that this is the case (LRRK1 and LRRK2 form what is called a heterodimer – a pairing of two different proteins that results in specific activity being initiated).
Given this close working relationship, the investigators next looked at a genetic database based on the DNA from people with familial and sporadic Parkinson’s from Tunisia. Given that there is a very high frequency of the Lrrk2 G2019S variant in this population, the dataset provided a unique opportunity to assess the role of LRRK1 genetic modifications on the risk of developing Parkinson’s.
Interestingly, the researchers found that one particular LRRK1 mutation (rs55739947) appeared to lower the average age of diagnosis of Parkinson’s by 6.2 years. Thus the combination of LRRK1 and LRRK2 mutations could be influencing the age of onset of Parkinson’s.
So what’s the difference between LRRK1 and LRRK2?
Not much.
Source: Het paper
As you can see from the image above, LRRK1 and LRRK2 share a high degree of ‘homology‘ (in other words, sharing a lot of the similar sequences of As, Gs, Ts, & Cs that make up DNA), and both of the LRRK proteins possess an almost identical structure. This level of similarity has led to speculation that the two genes may be compensate for one another – when something is wrong with one of the LRRK siblings, the other one can step in to replace it.
This may be the reason why many of the mice that have been genetically engineered to include LRRK2 mutations have not exhibited the neurodegeneration or other features associated with Parkinson’s (Click here, here, here, here and here to see examples of this). Perhaps in these LRRK2 mutant mice, LRRK1 is compensating for the loss of LRRK2.
So what happens when there is something wrong with both of the LRRK genes?
That’s a good question.
One that has recently been answered in mice:
Title: Age-Dependent Dopaminergic Neurodegeneration and Impairment of the Autophagy-Lysosomal Pathway in LRRK-Deficient Mice
Authors: Giaime E, Tong Y, Wagner LK, Yuan Y, Huang G, Shen J.
Journal: Neuron. 2017 Oct 18. pii: S0896-6273(17)30905-4.
PMID: 29056298
In this study the researchers generated mice that had genetic disruption of both of the LRRK genes, resulting in no LRRK1 or LRRK2 protein being present in the mice. This complete inactivation of LRRK1 and LRRK2 resulted in an age-dependent loss of DA neurons (starting at approximately 14 months of age in the mice – basically middle age for mice who will live for 2-2.5 years in captivity). This cell loss was accompanied by an increase in levels of the Parkinson’s associated protein alpha synuclein and a disruption of the autophagy process – the waste-disposal/recycling system of the cell. A similar effect was observed in cells in other regions that are affected by Parkinson’s disease, such as the locus coeruleus. Curiously though, the loss of both LRRK genes did not cause any neurodegeneration in other regions of the brain (such as the cerebral cortex and cerebellum).
Is this effect limited to just LRRK1 and LRRK2 mutations?
No. A recent analysis of Parkinson’s genetic data indicates that the appearance of a common Parkinson’s-associated genetic variant is often associated with another rarer variant in people with Parkinson’s:
Title: Additional rare variant analysis in Parkinson’s disease cases with and without known pathogenic mutations: evidence for oligogenic inheritance.
Authors: Lubbe SJ, Escott-Price V, Gibbs JR, Nalls MA, Bras J, Price TR, Nicolas A, Jansen IE, Mok KY, Pittman AM, Tomkins JE, Lewis PA, Noyce AJ, Lesage S, Sharma M, Schiff ER, Levine AP, Brice A, Gasser T, Hardy J, Heutink P, Wood NW, Singleton AB, Williams NM, Morris HR; for International Parkinson’s Disease Genomics Consortium.
Journal: Hum Mol Genet. 2016 Dec 15;25(24):5483-5489.
PMID: 27798102 (This article is OPEN ACCESS if you would like to read it)
The researchers used two large independent datasets of sequenced DNA (totalling 7,900 cases of Parkinson’s and 6,166 controls). The investigators were trying to determine how many people with one common genetic variant that is associated with Parkinson’s disease (such as LRRK2 variants) also carry another ‘rarer’ genetic variant. Their results showed that approximately 30% of people with Parkinson’s with a known causal mutation also carry additional variants (this was compared to 16% in unaffected controls). And interestingly these 30% of cases had on average a younger ages at onset.
So what does it all mean?
Summing up: In a previous post, we discussed the idea of rare genetic variants having a major impact on conditions like Parkinson’s (Click here to read that post). It now appears that rare variants might be playing a more important role than previously thought. It should be added, however, that our understanding of how genes influence Parkinson’s disease is still evolving and it is difficult to come to any solid conclusions. Folks reading this should not panic and jump to the wrong conclusions. I would suggest that we pay more attention to the ascending LRRK (that being LRRK1) and investigate what factors could be influencing vulnerability to Parkinson’s in these LRRK genes.
Let’s finish today’s post where we started: Composer Vaughan Williams inscribed the opening and closing lines from Meredith’s poem on the flyleaf of his album. It seems a nice way to finish up:
He rises and begins to round,
He drops the silver chain of sound,
Of many links without a break,
In chirrup, whistle, slur and shake.
For singing till his heaven fills,
‘Tis love of earth that he instils,
And ever winging up and up,
Our valley is his golden cup
And he the wine which overflows
to lift us with him as he goes.
Till lost on his aerial rings
In light, and then the fancy sings.
ADDENDUM:
So, the day after making this post live I had a chat with LRRK research Dr Patrick Lewis of the University of Reading. He is one of the researchers on the “Rare variants in LRRK1 and Parkinson’s disease” discussed above. He suggested to me that the picture is a lot less clear with the human data than one might be lead to believe. His basic message was: LRRK2 is certainly important, but the jury is still out on LRRk1. But of more concern, he suggested, is what impact the LRRK2 inhibitors are having on LRRK1. Many of the inhibitors under development are designed to target the ‘kinase‘ region of LRRK2, which is one of the homologous (similar) regions between the two proteins. Thus there is the possibility that the inhibitors could reduce LRRK1 activity as well as LRRK2.
The banner for today’s post was sourced from Bio







hi simon,
i’ve been enjoying this site for about a year. diagnosed in 2014.
a lot of the science is above my pay grade, but I get more out of your explanations than from most sites.
I’ve had a swab done from 23andme and also Indiana university looking for possible markers so I can focus potential treatments. on L-dopa (ten sinemet per day) but also isradipine, NAC, turmeric, methyl b-12, methylfolate (MTHFR c677t T/T). I’d like to offer an alternate soundtrack for your LRRK1 post: King Crimson’s “lark’s tongue in aspic”
https://www.youtube.com/watch?v=vy3UiXb2uDQ
LikeLiked by 1 person
Hi Billy,
Thanks for the comment and the music link. Great choice! I would be very interested to hear what sort of information Indiana university offers you. The personalised approach is the best way forward. There is no point to taking a particular supplement if your body is not designed to process it, so it will be interesting to see if Indiana university can provide you with more useful information. Thanks for sharing and the kind words about the site.
Kind regards,
Simon
LikeLike
As directed I listened to Vaughn Williams while reading – nice touch. In other news – will most neurologists agree to genetic testing? Seems like it could make a difference. (Sorry I think you covered this somewhat in a previous post)
LikeLike
DIdn’t see Billy’s post till now. how much is 23 and me or other testing – is this advised to find out? Seems like Simon recommended it in one post.
LikeLiked by 1 person
Hi DKDC,
Hope all is well. I suspect the genetic testing will become more of a thing in the near future, if only to help people determine what they should and shouldn’t be taking supplement/treatment-wise. But the genetic testing should not be taken lightly. Recently Benyamin Stecher over at the Tomorrow Edition blog interviewed Prof John Hardy from UCL in London. Hardy is a world leading researcher on the genetics of PD (probably #1), but he says:
“Genome sequencing is a private decision. I did it 20 years ago and found that I was APOE3 homozygous. I’m not sure I would do it now because if I found out I was APOE4 homozygous(an allele strongly correlated with the onset of Alzheimer’s disease) it would be pretty devastating as I’d likely now be in my last decade of non-dementia”
It is a very interesting interview (https://tmrwedition.com/2017/11/07/q-a-with-acclaimed-geneticist-prof-john-hardy/) and the blog is a must read for the PD community – Ben was diagnosed with PD at age 29. He has been travelling the world and learning as much as he can about PD from the best in the business. I am honestly amazed at some of the doors he has been able to open. VERY interesting blog.
23andMe (https://www.23andme.com) is free for folks with PD in the US, but it cost about £150 here in the UK.
Hope this helps,
Simon
LikeLike
Saw my MDS doc – she said there is no need for genetic testing at present – but I think her attitude is no supplements work – until proven that they do. So genetic info is not needed to throw sinemet at PD. Either way the treatment is the same. I will see what another researcher says next week. (Simon is one researcher, my doc used to be a researcher, I will get a third opinion.)
Would love to hear more opinions on how people look at this. I am a little slow to change I think. I need to read Ben’s blog.
LikeLike
Love the King Crimson – Eddie Jobson era?
LikeLike
david cross on this one, just before eddie I believe. the testing was free after confirming dx.
LikeLike