|
# # # # At the end of each month the SoPD writes a post which provides an overview of some of the major pieces of Parkinson’s-related research that were made available during October 2025. The post is divided into 10 parts based on the type of research:
# # # # |
So, what happened during October 2025?
In world news:
October 1st – Scientists report the detection of organic molecules on Enceladus, based on plume samples taken by the Cosmic Dust Analyzer on the Cassini spacecraft (Click here to read more about this).
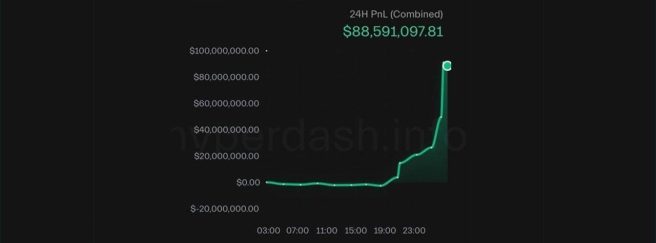
October 10th – Crypto enthusiasts were left puzzled as to how just 30 minutes before President Trump’s surprise announcement of 100% China tariffs, an individual (@bigwinner01) opened a massive, multi-million dollar leveraged short position on Bitcoin, and made $88 million in just two hours… and then closed the position (Click here to read more about this).
October 20th – Japan finally joined the list of countries to have female leaders – Sanae Takaichi made history as Japan’s first female prime minister. There are 73 women in Japan’s Lower House, representing just 15.7% of the 465-seat chamber (Click here to read more about this).
October 27th – “At the direction of President Trump, the Department of War carried out three lethal kinetic strikes on four vessels operated by Designated Terrorist Organizations (DTO) trafficking narcotics in the Eastern Pacific” (Click here to read more about this).
October 28th – OpenAI was founded as a non-profit in 2015 with a mission to safely build “artificial general intelligence” for humanity’s benefit. Now, it’s not so ‘open’. OpenAI has now “converted its main business into a for-profit corporation, the conclusion of a lengthy and fraught legal saga” (Click here to read more about this – Sam Altman scares me…)
In the world of Parkinson’s research, a great deal of new research and news was reported:
In October 2025, there were 1,257 research articles added to the Pubmed website with the tag word “Parkinson’s” attached (11,069 for all of 2025 so far). In addition, there was a wave to news reports regarding various other bits of Parkinson’s research activity (clinical trials, etc).