Over the Christmas festive period an interesting study was published in the journal Proceedings of the National Academy of Sciences (PNAS). It was about a protein called Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2) that has some impressive properties that could be good for Parkinson’s disease.
In today’s post we will review the results of the study and discuss what they mean for Parkinson’s disease.
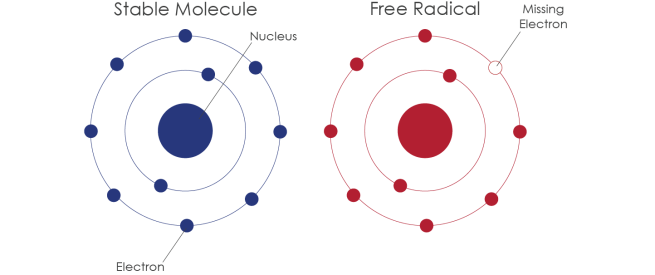
We are going to be talking about free radicals. Source: PRIMOH2
Antioxidants are one of those subjects that is often discussed, but not well understood. So before we review the study that was published last week, let’s first have a look at what we mean when we talk about antioxidants.
What is an antioxidant?
An antioxidant is simply a molecule that prevents the oxidation of other molecules.
OK, but what does that mean?
Well, the cells in your body are made of molecules. Molecules are combinations atoms of one or more elements joined by chemical bonds. Atoms consist of a nucleus, neutrons, protons and electrons.
Oxidation is simply the loss of electrons from a molecule, which in turn destabilises the molecule.
Think of iron rusting. Rust is the oxidation of iron – in the presence of oxygen and water, iron molecules will lose electrons over time. Given enough time, this results in the complete break down of objects made of iron.

Rust, the oxidation of metal. Source: TravelwithKevinandRuth
The exact same thing happens in biology. Molecules in your body go through a similar process of oxidation – losing electrons and becoming unstable. This chemical reaction leads to the production of what we call free radicals, which can then go on to damage cells.
What is a free radical?
A free radical is an unstable molecule – unstable because it is missing electrons. They react quickly with other molecules, trying to capture the needed electron to re-gain stability. Free radicals will literally attack the nearest stable molecule, stealing an electron. This leads to the “attacked” molecule becoming a free radical itself, and thus a chain reaction is started. Inside a living cell this can cause terrible damage, ultimately killing the cell.
Antioxidants are thus the good guys in this situation. They are molecules that neutralize free radicals by donating one of their own electrons. The antioxidant don’t become free radicals by donating an electron because by their very nature they are stable with or without that extra electron.

How free radicals and antioxidants work. Source: h2miraclewater
Interesting, but what does all this have to do with this new gene Nrf2?
Well, Nrf2 is a ‘transcription factor’ with some interesting properties.
What is a transcription factor?
So you remember your high school science class when some adult at the front of the class was talking about biology 101 – DNA gives rise to RNA, RNA gives rise to protein.

The basic of biology. Source: Youtube
Ultimately this is a circular cycle, because the protein that is produced using RNA is required at all levels of this process. Some of the protein is required for making RNA from DNA, while other proteins are required for making protein from the RNA instructions.
A transcription factor is a protein that is involved in the process of converting (or transcribing) DNA into RNA.
Now, a transcription factor can be an ‘activator’ of transcription – that is initiating or helping the process of generating RNA from DNA.
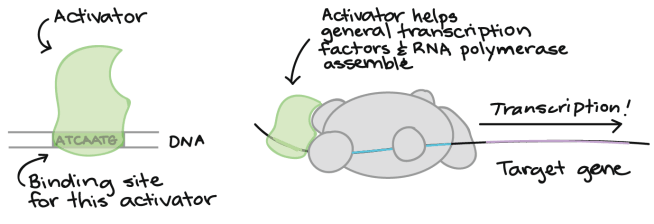
An example of a transciptional activator. Source: Khan Academy
Or it can be a repressor of transcription – blocking the machinery (required for generating RNA) from doing it’s work.

An example of a transciptional repressor. Source: Khan Academy
Nrf2 is an activator of transcription. When it binds to DNA to aids in the production of RNA, which then results in specific proteins being produced.
And this is where Nrf2 gets interesting.
You see, Nrf2 binds to antioxidant response elements (ARE).
What are ARE?
Antioxidant response elements (ARE) are regions of DNA is commonly found in the regulatory region of genes encoding various antioxidant and cytoprotective enzymes.
The regulatory region of genes is the section of DNA where transcription is initiated for each gene. They are pieces of DNA that a transcription factor like Nrf2 binds to and activates the production of RNA.
ARE are particularly interesting because these regions reside in the regulatory regions of genes that encode naturally occurring antioxidant and protective proteins. And given that antioxidants and protective proteins are generally considered a good thing for sick/dying cells, you can see why Nrf2 is an interesting protein to investigate.
By binding to ARE, Nrf2 is directly encouraging the production of naturally occurring antioxidant and protective proteins. And this is why a lot of people are excited by Nrf2 and call it the ‘next big thing’.
So what did the new research study report?
Well, this is where the story gets really interesting.
The researchers in the new study found that Nrf2 has some additional features that may be completely unrelated to the antioxidant properties:

Title: Nrf2 mitigates LRRK2- and α-synuclein-induced neurodegeneration by modulating proteostasis.
Authors: Skibinski G, Hwang V, Ando DM, Daub A, Lee AK, Ravisankar A, Modan S, Finucane MM, Shaby BA, Finkbeiner S.
Journal: Proc Natl Acad Sci U S A. 2016 Dec 27. pii: 201522872.
PMID: 28028237
The researchers wanted to determine what effect introducing exaggerated amounts of Nrf2 into cell culture models of Parkinson’s disease would have on the behaviour and survival of the cells. There were two types of cell culture models of Parkinson’s disease used in the study: one produced a lot of the Parkinson’s associated protein alpha synuclein (normal un-mutated) and the other cell culture model involved two mutations in the Lrrk2 gene (we have previously discussed Lrrk2 – click here to read that post).
The researchers had previously demonstrated that both of these cell culture models of Parkinson’s disease exhibited increased levels of cell death when compared with normal cells. In the current study, when the researchers artificially exaggerated the amounts of Nrf2 in both sets of cell cultures, they found that not only did Nrf2 reduce Lrrk2 and alpha-synuclein toxicity in cell culture, but it also influenced alpha-synuclein protein regulation, by increasing the degradation of the protein. This means that Nrf2 increased the disposal of the unnecessary excess of alpha synuclein.
In addition, Nrf2 also promoted the collection of free-floating mutant Lrrk2 and bundling it up into dense ‘inclusion bodies’ – dense clusters which are similar to the Lewy bodies of Parkinson’s disease but inclusion bodies are not associated with cell death. The scientists concluded that excessive levels of Nrf2 help to make the cells healthier and that this could represent a new target for future therapies of Parkinson’s disease. The researchers acknowledge that the ARE-related features of Nrf2 may be also playing a beneficial role in the cells, but this is the first time the alpha synuclein and Lrrk2 features have been identified.
Sounds great. Are there any catches?
Yes, a very interesting one.
The response of Nrf2 is time-dependent. The researchers found that over stimulation with Nrf2 leads to natural compensation from cells that eventually limits the activity of Nrf2. In other words, too much of a good thing loses it’s affect over time. Biology is one giant balancing act and sometimes when one factor is artificially introduced, cells will compensate regardless of whether it’s a good thing or not.
The researchers suggested that this issue could potentially be over come by periodic use of Nrf2, rather than simply chronic (or continuous) use of the protein. This still needs to be determined, however, in follow up experiments.
What does it all mean?
This new study provides us with new data relating to a protein that has been seen as holding great promise in the treatment of neurodegenerative conditions (not just Parkinson’s disease). The new research, however, demonstrates some interesting characteristic of Nrf2 specific to two Parkinson’s disease related genes.
Nrf2 has been considered a drug target for some time and agents targeting this protein have been patented and are under investigation (Click here to read more on this). We will be keeping an eye out for these compounds and we’ll report here the results of any research being conducted on them.
Interesting side note here:
We have previously discussed the treatments for Parkinson’s disease that were prescribed in India over 2000 years ago (Click here for that post). Outlined in the ancient texts, called the ‘Ayurveda’ (/aɪ.ərˈveɪdə/; Sanskrit for “the science of life” or “Life-knowledge”) was the use of the seeds of Mucuna pruriens in treating conditions of tremor. The seeds of this tropical legume we now know have extremely high levels of L-dopa in them (L-dopa being the standard therapy for Parkinson’s disease in modern medicine).
Here’s the interesting bit:
A second popular Ayurvedic treatment that is popular for Parkinson’s disease is Curcumin.

Tumeric. Source: Cerebrum
Curcumin is an active component of turmeric (Curcuma longa), a dietary spice used in Indian cuisine and medicine. Curcumin exhibits antioxidant, anti-inflammatory and anti-cancer properties, crosses the blood-brain barrier and there are numerous studies that indicate neuroprotective properties in various models of neurological disorders.
Curcumin has also been shown to activate Nrf2 (Click here , here and here for more on this).
It has also been shown to prevent the aggregation of alpha synuclein (click here for more on this).
We are always amazed at the curious little connection with ancient remedies that can be found in modern research and medical practice, and we thought we’d share this one here.
EDITORIAL NOTE: The content provided by the Science of Parkinson’s website is for information purposes only. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. The information provided on this website should under no circumstances be considered medical advice, and any actions taken by readers should firstly be discussed with a qualified healthcare professional.
The banner for today’s post was sourced from NRF2 science