For more than 50 years, L-dopa (a critical ingredient used by the brain to produce the chemical dopamine) has been one of the primary therapies used in the treatment of Parkinson’s disease. Over those years, there have been several different versions of L-dopa, providing advantages over previous forms. Last week, the results of clinical trials involving a new inhalable version of L-dopa were published.
In this post we will review the results of those studies.

Inhalers. Source: Verywell
The motor features (a resting tremor in one of the limbs, slowness of movement, and rigidity in the limbs) of Parkinson’s disease begin to appear when most of the dopamine producing neurons in the brain have been lost (specifically, >60% of the midbrain dopamine neurons). Thus for the last 50 years the primary means of treating Parkinson’s disease has been via dopamine replacement therapies.
Why don’t we just inject people with dopamine?
The chemical dopamine has a very difficult time crossing the blood-brain barrier, which is a thick membrane surrounding the brain. This barrier protects the brain from unwanted undesirables (think toxic chemicals), but it also blocks the transfer of some chemicals that exert a positive impact (such as dopamine).
When dopamine is blocked from entering the brain, other enzymes can convert it into another chemical called ‘norepinephrine’ (or epinephrine) and this conversion can cause serious side effects in blood pressure and glucose metabolism.
In addition, any dopamine that does find its way into the brain is very quickly broken down by enzymes. Thus, the amount of time that dopamine has to act is reduced, resulting in a very limited outcome. And these reasons are why doctors turned to L-dopa instead of dopamine in the treatment of Parkinson’s disease.
What is L-dopa?
Basically, Levodopa (L-dopa) is a chemical intermediary in the production of dopamine. That is to say, you need L-dopa to make dopamine. L-dopa is very stable inside the body and crosses the blood-brain-barrier very easily.
In the UK, a commonly used version is known as ‘Sinemet®‘(produced by Merck).

The chemical structure of L-dopa. Source: Wikipedia
The best way to understand what L-dopa is probably be to explain the history of this remarkable chemical.
The history of L-dopa
Until the 1950s there were few treatment options for Parkinson’s disease, but a young scientist in Sweden was about to change that.
This is Arvid Carlsson.

Prof Arvid Carlsson. Source: Alchetron
He’s a dude.
In 1957, he discovered that when he injected the brains of rabbits with a neurotoxin (reserpine) it killed the dopamine neurons (and the animals exhibited reduced movement). He also discovered that by injecting the dopamine precursor –L-dopa – into those same animals, he was able to rescue their motor ability. Importantly, he found that the serotonin precursor (called 5-hydroxytryptophan) was not capable of reversing the reduction in motor ability, indicating that the effect was specific to L-dopa.
Here is the 1957 report:

Title: 3,4-Dihydroxyphenylalanine and 5-hydroxytryptophan as reserpine antagonists.
Authors: Carlsson A, Lindqvist M, Magnusson T.
Journal: Nature. 1957 Nov 30;180(4596):1200. No abstract available.
PMID: 13483658 (the article on the Nature website – access required)
This was a fantastic discovery. A Nobel prize winning discovery in fact.
But what to do with it?
At the time, we did not know that dopamine was depleted in Parkinson’s disease. And people with Parkinson’s continued to suffer.
It was not until 1960 that the critical discovery of Parkinson’s disease was made by another young European scientist. Carlsson’s research (and that of others) inspired the Austrian researcher, Oleh Hornykiewicz to look at dopamine levels in people with Parkinson’s disease.
And what he found changed everything.

Prof Oleh Hornykiewicz. Source: Kurienwissenschaftundkunst
In his study, Hornykiewicz found very high levels of dopamine in the basal ganglia of normal postmortem adult brains, but a marked and consistent reduction (approx. 10-fold) in six postmortem cases of Parkinsonisms. The basal ganglia is one of the main regions of the brain that dopamine neurons communicate with (releasing dopamine there).

Title: Distribution of noradrenaline and dopamine (3-hydroxytyramine) in the human brain and their behavior in diseases of the extrapyramidal system
Authors: Ehringer H, Hornykiewicz O.
Journal: Parkinsonism Relat Disord. 1998 Aug;4(2):53-7. No abstract available.
PMID: 18591088
Importantly, Hornykiewicz did not stop there.
In November 1960, Hornykiewicz approached Walther Birkmayer, a doctor at a home for the aged in Vienna, and together they began some clinical trials of L-dopa in July 1961. Birkmayer injected 50 to 150 mg intravenously in saline into 20 volunteers with Parkinsonism. In their report, Birkmayer and Hornykiewicz wrote this regarding the results:
“The effect of a single intravenous injection of l-dopa was, in short, a complete abolition or substantial relief of akinesia. Bedridden patients who were unable to sit up, patients who could not stand up when seated, and patients who when standing could not start walking performed after l-dopa all of these activities with ease. They walked around with normal associated movements, and they could even run and jump. The voiceless, aphonic speech, blurred by palilalia and unclear articulation, became forceful and clear as in a normal person. For short periods of time the people were able to perform motor activities, which could not be prompted to any comparable degree by any other known drug”
Despite their initial excitement, Birkmayer and Hornykiewicz found that the response to L-dopa was very limited in its duration. In addition, subsequent trials by others were not able to achieve similar results, with many failing to see any benefit at all.
And that was when George stepped into the picture.

Dr George Cotzias…and yes, he is holding a brain. Source: New Scientist
Dr George Cotzias was a physician working in New York who became very interested in the use of L-dopa for Parkinson’s disease. And he discovered that by starting with very small doses of L-dopa, given orally every two hours and gradually increasing the dose gradually he was able to stabilize patients on large enough doses to cause a dramatic changes in their symptoms. His studies led ultimately to the Food and Drug Administration (FDA) approving the use of L-dopa for use in PD in 1970. Cotzias and his colleagues were also the first to describe L-dopa–induced dyskinesias.
How does L-dopa work?
When you take an L-dopa tablet, the chemical will enter your blood. Via your bloodstream, it arrives in the brain where it will be absorbed by cells. Inside the cells, another chemical (called DOPA decarboxylase) then changes it into dopamine. And that dopamine is released, and that helps to alleviate the motor features of Parkinson’s disease.
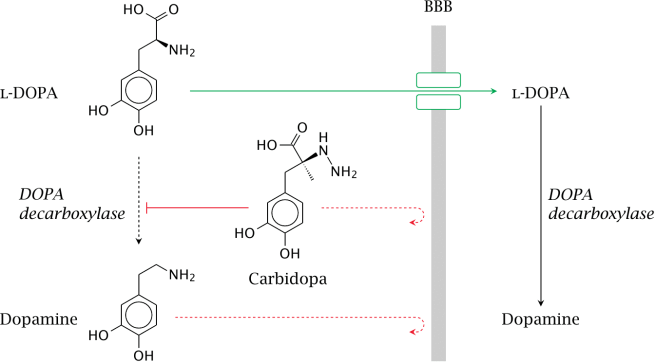
The production of dopamine, using L-dopa. Source: Watcut
Outside the brain, there is a lot of DOPA decarboxylase in other organs of the body, and if this is not blocked then the effect of L-dopa is reduced in the brain, as less L-dopa reaches the brain. To this end, people with Parkinson’s disease are also given Carbidopa (Lodosyn) which inhibits DOPA decarboxylase outside of the brain (Carbidopa does not cross the blood-brain-barrier).
How does the L-dopa inhaler work?
The company behind this new product, Acorda Therapeutics, spent many years developing a powdered version of levodopa that could be delivered to the lungs. Early on in this developmental process the scientists realised a problem: while normal asthma inhalers only need to release micrograms of their medicine to the lungs, a L-dopa inhaler would need to deliver 1,000 times more than that to have any effect. The huge amounts were needed to ensure that enough L-dopa would get from the lungs into the brain to be effective. Thus, the ARCUS inhaler delivers 25 to 50 milligrams in two breaths.
The inhaler contains capsules of L-dopa, which are designed to break open so that the powder can escape. By sucking on the inhaler (see image below), the open capsule starts spinning, releasing the levodopa into the air and subsequently into the lungs.

The ARCUS inhalation technology. Source: ParkinsonsLife
Pretty straightforward, right? Nice idea, cool design, easy to use.
But does it work?
What were the results of the clinical trials?

Title: Preclinical and clinical assessment of inhaled levodopa for OFF episodes in Parkinson’s disease.
Authors: Lipp MM, Batycky R, Moore J, Leinonen M, Freed MI.
Journal: Sci Transl Med. 2016 Oct 12;8(360):360ra136.
PMID: 27733560 (This article is OPEN ACCESS if you would like to read it)
In their research report, the scientists provided data from three studies: preclinical, phase one clinical, and phase two clinical. In the preclinical work, they measured the levels of L-dopa in dogs who had inhaled levodopa powder. When they looked at blood samples, they found that levodopa levels peaked in all of the animals 2.5 min after administration. This represented a very quick route to the blood system, as dogs that were given levodopa plus carbidopa orally did not exhibit peak blood levodopa levels until 30 min after administration.
In the phase one (safety) clinical trial, 18 healthy persons were enrolled, and again comparisons were made between inhaled CVT-301 and orally administered carbidopa/levodopa. This study demonstrated that CVT-301 was safe and had a similar rapidity of action as in the preclinical dog study.
Next, the researchers conducted a phase two (efficacy) clinical study. This involve 24 people with Parkinson’s disease inhaling CVT-301 as a single 50mg dose during an OFF episode (periods of no prescribed medication). 77% of the CVT-301 treated subjects showed an increase in plasma levodopa within 10 min. By comparison, only 27% of a group of subjects taking oral doses of carbidopa/levodopa at a 25-mg/100-mg dose achieved the same levels within that time. Improvements in timed finger tapping and overall motor function (as measured by the Unified Parkinson’s Disease Rating Scale) were observed between 5 and 15 minutes after administration.
The most common adverse event was cough, but all of the coughing events were considered mild to moderate, generally occurring at the time of inhalation. In most cases, they were resolved rapidly and became less frequent after initial dosing.
So what does it all mean?
Inhalation of L-dopa may represent a novel means of treating people with Parkinson’s disease, especially those who struggle with swallowing pills. The most obvious benefit is the speed with which the subjects see results.
The amount of L-dopa being used is very high, however, and we will be interested to see the results of more long term studies before passing judgement on the inhaler approach. We’ll keep you informed as more information comes to hand.
The banner for today’s post is sourced from the BBC