A wise man once told me:
“When trying to understand genetics, think of DNA as the stream of words in a book. The nucleotides (A, G, T and C) are the individual letters. These ‘letters’ collect together to make up the genes (the sentences) which give the book meaning and convey information. And the chromosomes are the chapters in that book.
Some of these ‘books’ are short reads – the fly has only 139.5 million nucleotides (‘words’) and 15,682 genes (sentences) spread across just 4 chromosomes (‘chapters’), while others are long books – humans = 3 billion words, divided into 22,000 sentences, and 23 chapters.
They were helpful words – putting things in perspective – and I hope that they might aid you dear reader as we tackle the topic of this post – a genetic mutation in a particular location of chromosome 22 and its relationship with Parkinson’s disease.
Oh, and do not be fooled into thinking that size matters when it comes to chromosomes. The mighty hedgehog and faultless pigeon have almost twice as many chromosomes as we do (45 and 40 pairs, respectively), and yet…
As most of you will be aware, human beings have 23 pairs of chromosomes.
Chromosomes are a concept that many people are aware of (a pub quiz type of topic), but what are they?
What exactly is a chromosome?
In a nutshell, a chromosome is a very efficient way of packing a lot of DNA into a cell.
Within most of the cells in your body, DNA is densely coiled into discrete packages called chromosomes. Without such packaging, the stringy DNA molecules would be too long to fit inside the cell. In fact, if you uncoiled all of the DNA molecules in a single human cell and placed them end-to-end, they would stretch for at least 6 feet. And that’s just for one cell – remember that the humans have approx. 40 trillion cells in their body!
 A schematic demonstrating the arrangement of DNA- Genes-Chromosomes. Source: cancergenome.nih.gov
A schematic demonstrating the arrangement of DNA- Genes-Chromosomes. Source: cancergenome.nih.gov
When a cell is not dividing, the chromosomes usually sit in the nucleus of the cell in loose strands called chromatin. When the cell decides to divide, the chromatin condenses and wrap up very tightly, becoming chromosomes. Both loose chromatin and tightly wound chromosomes are very difficult to see, even with a microscope.
Chromosomes come in pairs – one set of 23 chromosomes from each parent, giving us a total of 46 chromosomes per cell. All of these pairs reside inside the nucleus of each cell, where their DNA is read and instructions (RNA) are sent off to be made into proteins which performs functions within the cell.
Within the DNA in the chromosomes there are sometimes mistakes (think of them as spelling mistakes in the book example we mentioned above). The mistakes are called ‘mutations’ or variants. They can involve sections of DNA being absent or sections of DNA being replicated multiple times.
This week new research was published dealing with Parkinson’s disease and a mutation in chromosome 22.
What do we know about Chromosome 22?
Chromosome 22 is the second smallest human chromosome, being only slightly larger than chromosome 21. Chromosome 22 spans approximately 50 million DNA base pairs and represents 1.5-2% of the total DNA in each cell.
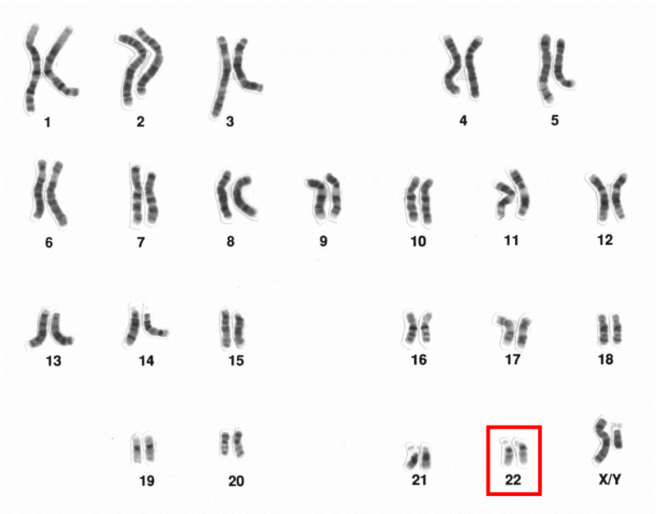
The 23 chromosomes of humans (this set is from a male). Chromosome 22 is highlighted. Source: Wikipedia
There are approx. 1000 genes on chromosome 22. The functions of many of these genes (what they tell the cell/body to do) is still being determined. Mutations in some of those genes, however, are associated with certain diseases. One particular disease associated with Chromosome 22 is called chromosome 22q11.2 deletion syndrome.
What is 22q11.2 deletion syndrome?
Chromosome 22q11.2 deletion syndrome (also known as DiGeorge syndrome) is a condition that arises from a section of chromosome 22 being absent. The ’22q11.2′ code part of the name relates to the specific location on chromosome 22 where the missing sections become apparent. About 87% of deletions occur within a 3 million base pair (nucleotides) region in the middle of one copy of chromosome 22 in each cell (remember that chromosomes come in pairs). The region contains at least 52 known genes.
Given the number of possible gene affected, there are numerous clinical features associated with 22q11.2 deletion syndrome, including heart defects, an opening in the roof of the mouth (a cleft palate), subtle facial features, learning issues, and low calcium levels.
Small ‘micro deletions’ within chromosome 22 are some of the most frequent known deletions found in human beings, occurring in about 25 in 100 000 people. These micro deletions are inherited from an affected parent in 5–10% of cases, while the rest occur spontaneously.
So what does Chromosome 22 have to do with Parkinson’s disease?
In 2009, this research report was published:

Title: The co-occurrence of early onset Parkinson disease and 22q11.2 deletion syndrome.
Authors: Zaleski C, Bassett AS, Tam K, Shugar AL, Chow EW, McPherson E.
Journal: Am J Med Genet A. 2009 Mar;149A(3):525-8.
PMID: 19208384
In this report the researchers described two patients, both with chromosome 22q11.2 deletion syndrome and early onset Parkinson’s disease (diagnosed before 45 years of age). The researchers suggested that this co-occurrence of chromosome 22q11.2 deletion syndrome and Parkinson’s disease in two unrelated patients was unlikely to be coincidence (given the low frequency of the conditions).
That first study was followed up by a second study:

Title: Association between early-onset Parkinson disease and 22q11.2 deletion syndrome: identification of a novel genetic form of Parkinson disease and its clinical implications.
Authors: Butcher NJ, Kiehl TR, Hazrati LN, Chow EW, Rogaeva E, Lang AE, Bassett AS.
Journal: JAMA Neurol. 2013 Nov;70(11):1359-66.
PMID: 24018986
In this report, the scientists conducted an observational study of the occurrence of Parkinson’s disease in the world’s largest cohort of well-characterized adults with a chromosome 22q11.2 deletion syndrome (n = 159; age range = 18.1-68.6 years). They found that people with chromosome 22q11.2 deletion syndrome had a significantly elevated occurrence of Parkinson’s disease compared with standard population estimates.
Curiously, they suggested that the common use of antipsychotics in patients with chromosome 22q11.2 deletion syndrome (to manage associated psychiatric symptoms) delayed diagnosis of Parkinson’s disease by up to 10 years. And in postmortem analysis of the brains of people with both conditions, they found the loss of dopamine neurons and the occurrence of Lewy bodies – characteristic features of Parkinson’s disease.
This was proof that people with chromosome 22q11.2 deletion syndrome were more vulnerable to developing Parkinson’s disease. But what about people with Parkinson’s disease? Do they have deletions with chromosome 22q11.2?
This week we got the answer to that question:

Title: Deletions at 22q11.2 in idiopathic Parkinson’s disease: a combined analysis of genome-wide association data.
Authors: Mok KY, Sheerin U, Simón-Sánchez J, Salaka A, Chester L, Escott-Price V, Mantripragada K, Doherty KM, Noyce AJ, Mencacci NE, Lubbe SJ; International Parkinson’s Disease Genomics Consortium (IPDGC), Williams-Gray CH, Barker RA, van Dijk KD, Berendse HW, Heutink P, Corvol JC, Cormier F, Lesage S, Brice A, Brockmann K, Schulte C, Gasser T, Foltynie T, Limousin P, Morrison KE, Clarke CE, Sawcer S, Warner TT, Lees AJ, Morris HR, Nalls MA, Singleton AB, Hardy J, Abramov AY, Plagnol V, Williams NM, Wood NW.
Journal: Lancet Neurol. 2016 Mar 23. [Epub ahead of print]
PMID: 27017469
The researchers analysed the DNA of 9387 people with Parkinson’s disease and 13 863 controls. They identified eight unrelated people with Parkinson’s disease who carried the chromosome 22q11.2 deletions. None of the controls had any of these deletions. Those people with Parkinson’s disease who had chromosome 22q11.2 deletions had earlier ages of onset (average age of diagnosis = 41 years old) than people with Parkinson’s disease who did not have the deletions (average age of diagnosis = 60.3 years). The researchers concluded that chromosome 22q11.2 deletions are associated with early onset Parkinson’s disease.
Some concluding thoughts
While the results of the Lancet Neurology study are very interesting, there are several important aspects to consider.
Firstly, the researchers noted that the estimated prevalence of 22q11.2 deletion syndrome in the general population is 0·024% or 24 in every 100,000 people. More importantly, as the study indicated the frequency of a 22q deletion among people with early-onset Parkinson’s disease is also very low (0·49% or 5 in every 1000 people with early-onset Parkinson’s disease). In fact, the number of people with the 22q11.2 deletion syndrome mutation and Parkinson’s disease is less than 20. So obviously this is a very low frequency event.
It is also interesting to consider that only 3% of patients with 22q11.2 deletion syndrome go on to develop Parkinson’s disease. Also a low frequency event. But studying this small population of people with a very specific genetic circumstance may enlighten us to some of the biological mechanisms causing this low frequency occurrence. And that may further aid us in better understanding other forms of Parkinson’s disease.
And that really is the take home message from this study: we are gradually building a map of the connections between genetics and Parkinson’s disease, and while genetics will not explain every case of this condition, the knowledge we gain from this process will allow us to better target the disease in the long run.



 A schematic demonstrating the arrangement of DNA- Genes-Chromosomes. Source:
A schematic demonstrating the arrangement of DNA- Genes-Chromosomes. Source: 


