|
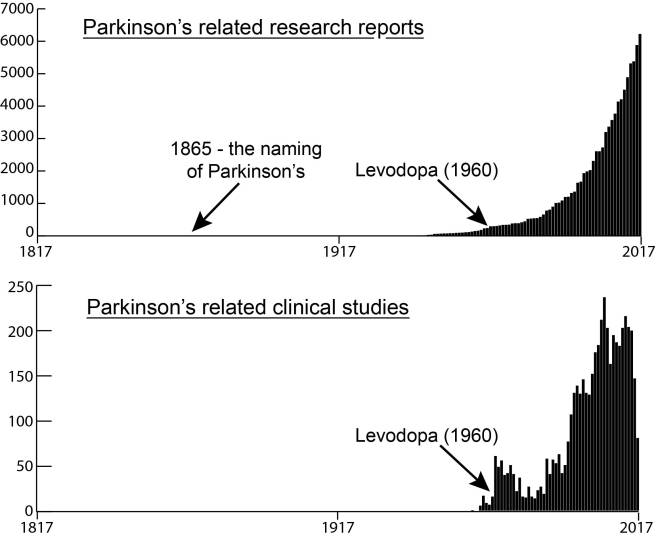
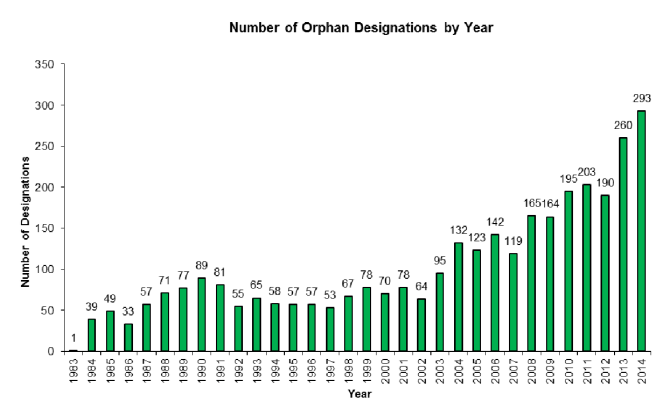
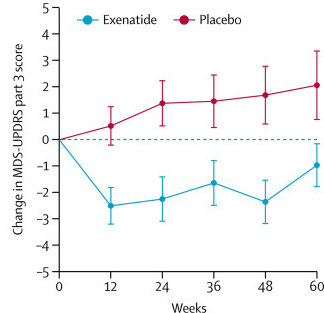
On Saturday 7th January, 2018, one of the world’s largest pharmaceutical companies – Pfizer – announced that it was abandoning research efforts focused on finding new drugs for Alzheimer’s and Parkinson’s. Naturally, the Parkinson’s and Alzheimer’s communities reacted with disappointment to the news, viewing it as a demoralising tragedy. And there was genuine concern that other pharmaceutical companies would follow suit in the wake of this decision. Those fears, however, are unfounded. In today’s post we will look at some of the reasons underlying Pfizer’s decision, why our approach to failure is wrong, why Pfizer will definitely be back, and what the Parkinson’s community can do about it all. |
Photo by David Kovalenko on Unsplash
1. Our approach to failure
I am currently reading “Black box thinking: The Surprising Truth About Success“. It is a really interesting book by journalist Matthew Syed that investigates how we approach to failure.
Matthew Syed. Source: Amazon
In the first chapter of his book, Syed makes comparisons between the way the aviation industry and the medical profession approach failure, pointing out the processes that follow situations when a disasters occur. In the aviation industry, when any event occurs there is a major investigative process that starts with the recovery of the black boxes. The aviation industry uses this system of investigation to learn from every single incident. It makes the information available to all and this helps with re-thinking everything from cockpit ergonomics and design to air traffic controller procedures. Even the airline companies are keen to be seen to be involved in this process of investigation. Failure, while unfortunate, is not shameful or stigmatising, but rather embraced and enlightening.
In addition, Syed points out that when an airline pilot sits down in his/her cockpit, their neck is also on the line if something goes wrong. Thus, it is in their best interest that the flight should be successful. And this is another reason why the aviation industry takes the reporting of failure so seriously. Everyone benefits from learning from previous situations. And all of this comes together with the observation that 2017 was the safest year on record for flying (based on deaths/flights – Source).