|
Today’s post discusses some interesting new research that could potentially have ‘blue sky’ implications for the treatment of Parkinson’s (‘blue sky‘ meaning somewhere in the longer-term framework). The research deals with the amazing activity of a newly discovered protein, isolated from tiny little fish. The protein responds to magnetic fields. And with a lot more work, the researchers behind this discovery think that this protein could be useful in the future for the treatment of Parkinson’s. In today’s post, we will have a look at this new protein, review the research report, and explore how this novel discovery could possibly be used for treating Parkinson’s.
|
Source: Flickr
The two fish in the image above are called Kryptopterus bicirrhis (or more commonly “glass catfish”).
These wondrous little creatures are typically found in the river waters of Thailand and Indonesia, they grow to a final length of approximately 8-12 cm (4-5 inches), and they live for 6-8 years.
But immediately you notice their most striking feature: the semi-transparent-ness of their bodies:
Why am I starting this post by talking about these curious little fish?
Some researchers in Baltimore (Maryland) recently made an amazing discovery related to them.
Which was?
The glass cat fish produces a protein which responds to magnetic fields.
And the researchers published their finding this report:
Title: Wireless control of cellular function by activation of a novel protein responsive to electromagnetic fields
Authors: Krishnan V, Park SA, Shin SS, Alon L, Tressler CM, Stokes W, Banerjee J, Sorrell ME, Tian Y, Fridman GY, Celnik P, Pevsner J, Guggino WB, Gilad AA & Pelled G
Journal: Scientific Reports (2018) 8, 8764
PMID: N/A (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in learning more about the behavioral response of fish to magnetic fields.
Why are they interested in magnetic fields?
It is well known that many forms of aquatic life use the Earth’s magnetic and electric fields for orientation and navigation themselves:
Some species also use electrical fields to detection of prey and predators (Click here to learn more about electroreception). But the cellular mechanisms underlying this amazing ability are only just starting to be uncovered and understood.
In their study of the glass catfish, the researchers were interested in learning more the biology of how glass catfish respond to magnetic fields, and to do this they set up an experiment in which the fish were exposed to magnetic stimulation.
In a series of 10 trials, a Neodymium Rare Earth Magnet was positioned on one side of the fish tank for 10 seconds and the fish were subjected to magnetic field (strength of 23 mT). As you can see in the image below, when the stimulation was not active, the fish were scattered as they swam around the tank (image A). During the stimulation, all the fish swam away from the source of the stimulation (image B), which was located on the right of the image. When stimulation was turned off, the fish once again swam in all directions (image C). In image D, there is a graph quantifying the amount of ‘stimulation-induced’ avoidance behaviour based on the amount of distance the fish swam away from the stimulation.
The researchers repeated the experiment with a alternating magnetic field – a transcranial magnetic stimulation system (at a strength of 250 mT), and they observed the same results: all fish swam away from the electromagnetic field, suggesting that the fish display behavioral responses caused by both static and alternating magnetic stimulation.
Now this was no major discovery – it was basically just a control experiment to make sure the fish were doing what they should do before the researchers continued with the rest of the study.
And this is where things start to get interesting.
‘Electroreceptor organs‘ – these are particular parts of the body on fish that are able to detect electrical fields – were first identified physiologically way back in the early 1960s by American neuroscientist Theodore H. Bullock and colleagues and by French scientists Thomas Szabo and Alfred Fessard. And slightly more recent research had suggested that the electroreceptors were particularly clustered in the anal fin of the glass catfish (Click here for that previous research).
The researchers in the study published last week isolated RNA from the anal fin of glass catfish and began looking for which piece of RNA would make a cell responsive to a magnetic field. They were trying to find RNA (the instructions for making a protein), which would allow them to identify the gene (the region of DNA that gives rise to a particular piece of RNA) that is associated with magnetic field responsiveness.
And after a large screening experiment they found a gene that provides the instruction for a protein (of 133 amino acids; ~15 kDa) that responds to magnetic fields.
They called it the ‘electromagnetic perceptive gene‘ (or EPG).
Interesting, but what does any of this have to do with Parkinson’s?
Well, the researchers next put the DNA for their ‘electromagnetic perceptive gene’ into rodent brain cells that they were growing in cell cultures.
Why did they do that?
These brain cells do not naturally have the electromagnetic perceptive gene, and when they are exposed to a magnetic field they usually do not respond in any way, shape or form to the stimulation. By adding the electromagnetic perceptive gene to the cells, the researchers wanted to see if they could introduce the function of magnetic field responsiveness.
And when the investigators inserted the electromagnetic perceptive gene into those cells and exposed them to a magnetic field, they did respond!
Cool!
The researchers found that the introduced electromagnetic perceptive gene was functional in these brain cells: In response to magnetic fields, the EPG protein would increases levels of calcium inside of cells. Such a situation, makes cells more excitable – that is, it lowers the threshold for activating the cells, making them more likely to respond to a signal.
And this got the scientists thinking: If this electromagnetic perceptive gene works in cells, maybe it will also work inside the brain.
So they decided to test the idea by making a virus which could infect cells with the electromagnetic perceptive gene. Once the virus was injected into a particular part of the brain, and the cells in that area were infected, they would start making the EPG protein. But would those cells respond to a magnetic field?
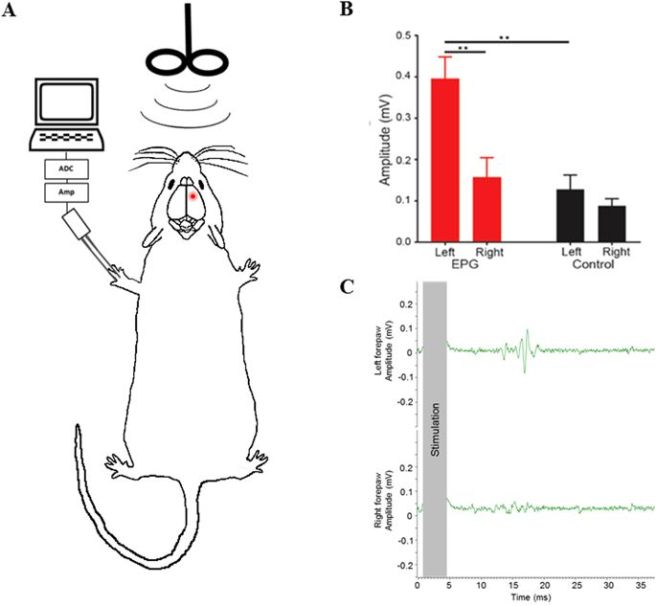
The researchers tested this question by injecting the virus into the primary motor cortex on the right side of the brain in a group of rats – the cells in this region regulate the limbs on the left side of the body.
A rat brain with the left primary motor cortex of the rat brain in green. Source: Nibb
Three weeks later when the investigators stimulated the right side of the head with a transcranial magnetic stimulation system, they observed an increase in levels of nerve stimulation in the limbs on the left side of the body (see panels A & B in the image below). Stimulation of the left side of the head resulted in no change in nerve stimulation in the right limbs, and stimulation of control animals who did not receive the EPG virus also displayed no response to stimulation (see panel B in the image below).
These results suggested to the investigators that neuronal activation of the EPG protein by a magnetic stimulation produced a measurable behavioral output.
Which is pretty amazing – you have to admit!
Remotely controlling very specific brain activity using a magnetic field. Wow!
And while noting that a lot more research and characterisation of the gene/protein is required (for example, exactly how EPG works is not clear), the researchers did speculate in the discussion of the report about various potential applications, from remote control of regions of the brain to whether the technique will work on non-neuronal tissues (such as the heart).
That is pretty cool, but how does it relate to Parkinson’s?
Remote control of neuronal activity might sound very dodgy, but some researchers are wondering if this technology could one day (in the distant future) be applied to the method of deep brain stimulation.
What is deep brain stimulation?
Deep brain stimulation (or DBS) is a treatment method that involves embedding electrodes into the brain to help modulate the brain activity involved in movement.
First introduced in 1987, deep brain stimulation consists of three components: the pulse generator, an extension wire, and the leads (which the electrodes are attached to). All of these components are implanted inside the body. The system is turned on, programmed and turned off remotely.
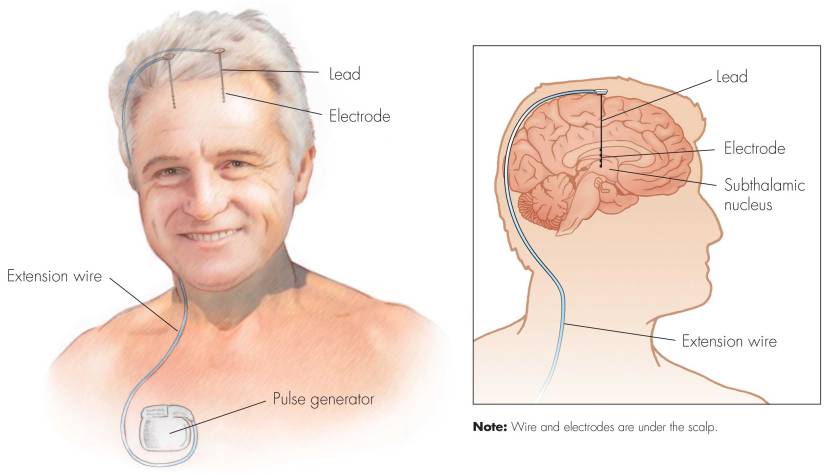
Source: Ucdmc
The electrodes that are implanted deep in the brain are tiny, and the very tip of them has small metal plates (each less than a mm in width) which provide the pulses that will help mediate the activity in the brain.

DBS electrode tip. Source: Oxford
The electrode extends up into the leads (or extension wire) which continue up and out of the brain, across the top of the skull, down the neck and to the pulse generator which is generally located on the chest.

Xray image demonstrating the leads. Source: Fineartamerica
There are many different brands/types of pulse generators, but they all largely do the same basic function. They are titanium packets containing the electronics and power supply for the leads and electrodes. They are implanted in a subcutaneous spot, usually located under the collar bone on the chest.

Pulse generators. Source: Cambridge (also a good read on DBS)
The pulse generator is programmable (which the doctor usually does several weeks post surgery). A small hand held device is used to turn on/off the generator, generally by holding the device over it.

Remote control. Source: Cambridge
For more information on the topic, click here to read about a case report of deep brain stimulation implantation.
Here is a video – kindly provided by fellow kiwi Andrew Johnson – that demonstrates the benefits of DBS:
How does deep brain stimulation actually work?
I have previously discussed how we initiate movement (Click here to read that post), and I will only briefly summarise the basics here.
Movement is largely controlled by the activity in a specific group of brain regions, collectively known as the ‘Basal ganglia‘.
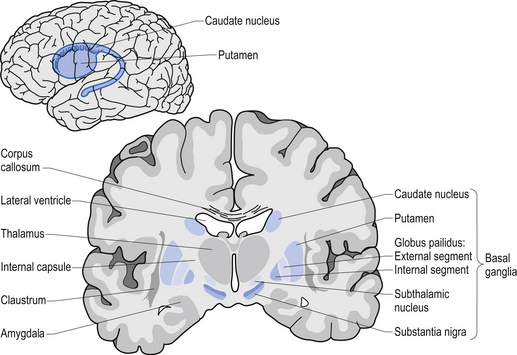
The basal ganglia structures (blue) in the human brain.
Source: iKnowledge
The basal ganglia receives signals from the overlying cerebral cortex regarding possible movements to make, and it processes that information before sending a signal on to another important participant in the regulation of movement: the thalamus.

A brain scan illustrating the location of the thalamus in the
human brain. Source: Wikipedia
The thalamus is a structure deep inside the brain that acts like the central control unit of the brain. Everything coming into the brain from the spinal cord, passes through the thalamus. And everything leaving the brain, passes through the thalamus. It is aware of most everything that is going on and it plays an important role in the regulation of movement. Importantly it is biased towards inhibiting movement – a signal from the basal ganglia has to be pretty strong, for the thalamus to give the green light and a particular movement can then be made.
Under normal circumstances, dopamine producing neurons release dopamine in the basal ganglia where it helps to mediate the local environment. It acts as a kind of lubricant for movement, the oil in the machine if you like. It helps to reduce the inhibitory bias of the basal ganglia.
Thus, with the loss of dopamine neurons in Parkinson’s, there is an increased amount of inhibitory activity. And as a result, the thalamus is kept in an overly inhibited state. And this is the reason why people with Parkinson’s have trouble initiating movement.

The workflow of the movement decision making process. Source: BJP
Now, as you can see from the image above, the globus pallidus is one of the main conduits of information into the thalamus. Given this pivotal position in the regulation of movement, the globus pallidus has been a region of major research focus for a long time.
It is also one of the sites targeted in deep brain stimulation therapy for Parkinson’s. Another target of deep brain stimulation therapy is the subthalamic nucleus which help to regulate the activity of the globus pallidus. By placing electrodes in one of these two brain regions (see image below), doctors can regulate the signal being passed from the basal ganglia to the thalamus and thus provide people with Parkinson’s relief from their motor-associated symptoms.

Two of the target sites for deep brain stimulation. Source: JAMA
But what do the electrodes do?
The electrodes will be programmed to release tiny electrical pulses that will help regulate the firing of the surrounding cells. By increasing the firing of those cells, deep brain stimulation reduces the inhibitory state of the thalamus.
The efficacy of deep brain stimulation is based on high frequency stimulation. Rates (or frequencies) below 50 pulses per second typically do not have an impact on stimulating the surrounding cells. In fact, low frequency can actually cause a stimulation-induced worsening of tremors. The most commonly used rates are between 130 and 185 pulses per second for Parkinson’s. And of course this is programmable and can be adjusted over time.
Ok, but how does the glass cat fish research relate to deep brain stimulation?
Imagine if instead of permanently embedding electrodes into the brain, we simply injected a virus that infected the cells of the subthalamic nucleus with the EPG DNA. Those cells would then become responsive to magnetic fields, which could be applied by (perhaps) wearing a hat or head band with particular magnets in it. Such a system would ideally be programmable – with the ability to increase the strength of the magnetic field in the hat, fine tuning the level of activity inside the brain.
That would be cool!
Yes, but before anyone gets too excited, please remember that we are talking hypotheticals here.
As I said in the introduction above, this is very ‘blue sky’ stuff. The electromagnetic perceptive gene has only just been discovered – we don’t know how it works and the research needs to be independently replicated before it can be utilised in any interesting way. And those interesting way will largely be limited to research purposes initially – so please don’t delay or put off a DBS procedure in the hope of waiting for the “magnetic fish DNA” approach.
Plus, we live in a world full of magnetic fields. You have to ask yourself what would happen if a person with “electromagnetic perceptive gene-based deep brain stimulation” walks past a microwave, makes a cell phone call, or walks through a metal detector at the airport? I for one am not in a rush to find out.
As the researchers suggest in their report, the “EPG technology could provide an exciting and valuable tool for studying neural activity at the network, cellular, and molecular levels”. And until further developments are made, that is where we stand.
So what does it all mean?
How animals can navigate or detect prey using magnetic or electrical fields is an absolute wonder of biology – one which we are only just starting to uncover the basics underlying mechanism of. With a better understanding of those processes, comes the possibility of using that knowledge to study other aspects of biology, as well as improving the lives of people affected by particular conditions.
Recently American researchers have identified a protein in fish that responds to magnetic fields. When they introduced that protein into cells in the brains of rodents, they found that they could remotely control the activity of those brain cells using a magnetic field. Although this achievement is exciting, the discovery will require a lot more research before this technology could ever be considered for use in people with Parkinson’s (as has been suggested in the popular media).
The new discovery does, however, provide researchers with an amazing new tool to further explore the biology of conditions like Parkinson’s, which will hopefully give rise to other ‘blue sky’ discoveries as well as more immediately impactful findings.
The banner for today’s post was sourced from Sciencesource





Simon,
Thanks yet again for another interesting article,
In the context of your article, the paper by Kondaveeti et al. looks interesting [1]. Unfortunately, this is behind a paywall. But, from what I can gather, “levodopa is loaded into magnetic hydrogels”. This is then released “under external magnetic field”. In my view, there would be considerable advantage in having a very slow delivery method for levodopa (that is, a long THALF), not because it might be more convenient, but because it might smooth out the peaks and troughs of plasma levodopa. This could reduce dyskinesia.
Reference:
[1] “Magnetic hydrogels for levodopa release and cell stimulation triggered by external magnetic field”
Stalin Kondaveeti, Ana Teresa, Silva Semeano, Daniel R.Cornejo, Henning Ulrich, Denise Freitas Siqueira Petria
Colloids and Surfaces B: Biointerfaces
Volume 167, 1 July 2018, Pages 415-424
https://www.sciencedirect.com/science/article/pii/S0927776518302509
John
LikeLike
interesting. I wonder if Parkinson’s brains respond to the Stephin Merritt version of Magnetic Fields as well. I think mine does (sorry – couldn’t resist).
LikeLike
Great post, Simon. Very interesting. And just as you recently observed with the spiny dogfish, progress often occurs at a breathtaking pace. If I understand correctly, the electric stimulation of DBS is inhibiting the inhibition imposed by the thalamus. In effect, it’s cancelling out signals rather than actually stimulating signals. On the other hand, the eps-gene activated neurons generate an actual ‘positive’ stimulation, causing action in the rat’s forepaw. Would this type of activation result in the same type of ‘inhibitiry Inhibition’ , or the opposite?
Thanks again for the post
LikeLike