|
# # # # Tiny variations our DNA can have a significant impact on our lives. For the last 20 years, Parkinson’s researchers have been collecting data highlighting ‘genetic risk factors’ that are associated with increasing one’s risk of developing the condition. More recently, however, these same scientists have started shifting their attention to the factors that modulate these genetic risk factors – and some of those influences are also genetic. In today’s post, we will look at new research exploring genetic variations that influence the effect of the Parkinson’s-associated GBA genetic variants, and discuss why this research has huge implications not only on how we conduct clinical trials, but also on how we will treat Parkinson’s in the future. # # # # |
 Prof Craig Venter. Source: ScienceMag
Prof Craig Venter. Source: ScienceMag
In June 2000, when the results of the first human genome sequencing were announced during a ceremony at the White House, the DNA sequencing pioneer Prof Craig Venter observed that “The concept of race has no genetic or scientific basis“.
He was suggesting that due to genetic variations among human individuals and populations, the term ‘race’ cannot be biologically defined. There was simply no evidence that the broad groups we commonly refer to as “races” have any distinct or unifying genetic identities (Click here for interesting additional reading on this).
 Source: Phillymag
Source: Phillymag
Prof Venter’s words were a powerful statement regarding the incredible variability within our genetic make up.
And that variability is even more remarkable considering that we are all 99.9 percent genetically identical.
So how do we explain the variability then?
Well, firstly that 0.1% difference is rather bigly.
If you plucked any two people random off the street and sequenced their DNA, you would find that they will differ at approximately 1 in every 1000 to 1500 DNA base pairs (base pairs being the familiar A-T & C-G’s that make up our DNA code).
 The basics of genetics. Source: CompoundChem
The basics of genetics. Source: CompoundChem
Given that the human genome contains approximately 3 billion of these base pairs, there will be 3-6 million base pairs that are different between the two randomly picked people from the street (Source).
But this is only a tiny part of the ‘variability’ story.
What do you mean?
The difference in effect that some of these 3 million or so different base pairs have is… well variable.
For example, a variation in a region of DNA (called MC1R) on chromosome 16 will result in red hair. If the variation is present: red hair. If not there: no red hair.
 Source: weheartit
Source: weheartit
But there are additional levels of complexity.
And to explain this we will use disease: We know that random changes within certain regions of our DNA can influence our vulnerability to particular medical conditions – these are called ‘genetic risk factors‘. But then on top of these, there are additional genetic variants that can modulate the impact of those risk factors.
These we will refer to as ‘genetic modifiers‘.
The risk factors puts you at risk, while the modifiers influence the level of risk.
And this is where things start to get really complex.
How does it get complex?
In the schematic below, the red square (#7) represents a genetic risk factor for a particular medical condition, and the blue square (#3) represents a genetic modifier, which is influencing the genetic risk factor (the line with the arrow). This ‘influence’ could be increasing or reducing the risk.
 Source: ScienceDirect
Source: ScienceDirect
I understand that, but how is that complex?
Well, on a very basic level, any genetic risk factor probably has multiple genetic modifiers modulating its influence (see panel B in the image below). And again these modifiers are each increasing or reducing the risk – resulting in a cumulative effect.
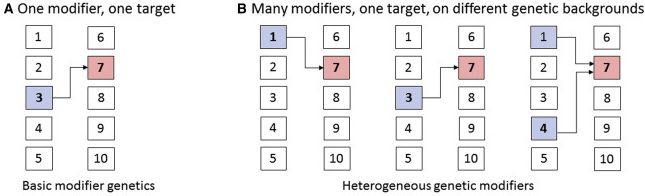 Source: ScienceDirect
Source: ScienceDirect
In this scenario, the right balance of genetic modifiers might actually reduce the risk significantly.
But it is probably even more complex than this, given that one genetic modifier may impact multiple genetic risk factors (see panel C in the image below). In addition, the effect of any modifier on a genetic risk factor will probably have down stream effects on other related risk factors (panel D in the image below).
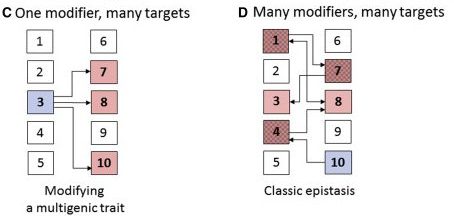 Source: ScienceDirect
Source: ScienceDirect
Imagine these simple schematics scaled up to 3 billion base pairs. You can hopefully begin to see how the complexity starts to snowball.
|
# RECAP #1: There is tremendous variability between each human being, and part of these differences can be explained by tiny variations in our DNA. Genetic variants may increase one’s risk of developing a medical condition (a “risk factor”), while other variations in our DNA may influence the impact of a risk factor (a “genetic modifier”). # |
Very interesting, but what does this have to do with Parkinson’s?
Potentially a lot.
Recently, researchers in New Zealand (yay!) and Australia published this report:
 Title: Common Variants Coregulate Expression of GBA and Modifier Genes to Delay Parkinson’s Disease Onset.
Title: Common Variants Coregulate Expression of GBA and Modifier Genes to Delay Parkinson’s Disease Onset.
Authors: Schierding W, Farrow S, Fadason T, Graham OEE, Pitcher TL, Qubisi S, Davidson AJ, Perry JK, Anderson TJ, Kennedy MA, Cooper A, O’Sullivan JM.
Journal: Mov Disord. 2020 Jun 18. Online ahead of print.
PMID: 32557794 (This report is OPEN ACCESS if you would like to read it)
In this study, the scientists wanted to explore whether genetic variations could explain the variability in age of onset of people with Parkinson’s. For their analysis, the researchers focused their attention on a specific genetic risk factor: the GBA gene.
What is the GBA gene?
The GBA gene (a gene is a functional section of DNA) provides the instructions for making a protein called glucocerebrosidase, which plays an important role in cellular waste disposal (discussed further below).
Importantly, genetic variations in the GBA gene are associated with an increased risk of developing Parkinson’s.
 The GBA gene and mutations associated with Parkinson’s. Source: Neurology
The GBA gene and mutations associated with Parkinson’s. Source: Neurology
In fact, genetic variations in the GBA gene are some of the most common genetic risk factors for the condition. It is believed that approximately 5%–8% of people with Parkinson’s may have a genetic mutation in their GBA gene (Click here and here to read more about this).
What does the Glucocerebrosidase protein do?
Glucocerebrosidase (also known as GCase) is an enzyme that helps with the digestion and recycling of various proteins (particularly glucocerebrosides) inside cells.
The enzyme is located and active inside small bags of degradative enzymes. These bags are the lysosomes and they can be found floating around inside of cells.
What do these lysosomes do?
On a relatively continual basis, small parts of a cells outer surface (or membrane) are being brought inside the cell. This is a process called endocytosis.
It occurs when the cell needs to consume resources from the outside world in order to find what it requires to function and survive. As a section of cell membrane is brought into the cell, it forms what is called a vesicle (which is a term used to refer to small spherical bags of stuff inside cells). Given the process by which these outer membrane vesicles are formed, they is referred to as endosomes (sometimes it is also called a vacuole).
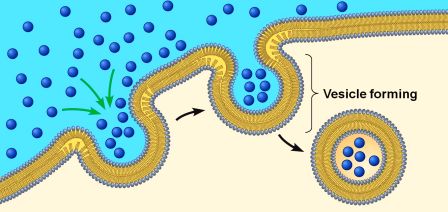 Source: Socratic
Source: Socratic
Once the endosome is inside the cell and detached from the rest of the membrane, it will bind to another vesicle which is called a lysosome. And as I mentioned above, lysosome is a small bag that is full of digestive enzymes, which help to break down the contents of the endosome.

How lysosomes work. Source: Prezi
The lysosome will fuse with the endosome/vacuole and the enzymes from the lysosome will mix with the material in the vacuole and digest it (or it break down into more manageable components).
This enzymatic process works in a very similar fashion to the commercial products that you use for washing your clothes.
 Enzymatic degradation. Source: Samvirke
Enzymatic degradation. Source: Samvirke
The reagents that you put into the washing machine with your clothes contain a multitude of enzymes, which help to break down the dirty, bacteria, flakes of skin, etc that cling to your clothes. Each enzyme breaks down a particular protein, fat or such like. And this is very similar to the collection of enzymes in the lysosome. All of them are needed to break down all of the contents of the endosome.
And if one of those enzymes – such as glucocerebrosidase/GCase – is faulty (due to a genetic mutation), then the enzymatic process is disrupted, which could result in the build up of un-degraded material over time.
Do people with GBA-associated Parkinson’s differ from people with ‘normal’ Parkinson’s?
There is a lot of variability, but people with GBA-associated Parkinson’s generally exhibit an earlier age of diagnosis and more progressive symptoms than people without a GBA mutation.
And this has recently been confirmed in this large clinical observation study:
 Title: Features of GBA-associated Parkinson’s disease at presentation in the UK Tracking Parkinson’s study.
Title: Features of GBA-associated Parkinson’s disease at presentation in the UK Tracking Parkinson’s study.
Authors: Malek N, Weil RS, Bresner C, Lawton MA, Grosset KA, Tan M, Bajaj N, Barker RA, Burn DJ, Foltynie T, Hardy J, Wood NW, Ben-Shlomo Y, Williams NW, Grosset DG, Morris HR; PRoBaND clinical consortium.
Journal: J Neurol Neurosurg Psychiatry. 2018 [Epub ahead of print]
PMID: 29378790 (This article is OPEN ACCESS if you would like to read it)
In this multi-research centre study, the investigators studied 1893 people with Parkinson’s. Of these 48 (2.5%) had one copy of a known GBA mutation, 117 (6.2%) had a non-GBA genetic variant that had previously been associated with Parkinson’s (such as mutations in genes like LRRK2 or alpha synuclein), and 28 (1.5%) carried genetic variants of unknown significance in the GBA gene.
Individuals with Parkinson’s associated GBA mutations were on average diagnosed 5 years earlier compared with non-carriers. They were also more likely to have postural instability and gait difficulties compared with non-carriers. In addition, they had more progressive forms of Parkinson’s (as determined by more advanced Hoehn and Yahr staging – after adjustment for age – compared with non-carriers).
No differences was observed in cognitive function between GBA mutation carriers and non-carriers. Cognitive impairment/dementia have been reported in other studies at a later stages of the condition (Click here to read more about this).
So, yes, GBA-associated Parkinson’s is different.
But even within this subtype of Parkinson’s there is variability within how the condition presents itself.
|
# # RECAP #2: Tiny variations within the GBA gene are some of the most common genetic risk factors for Parkinson’s – increasing the risk of developing the condition. GBA provides the instructions for a protein (called GCase) involved in cellular waste disposal, and genetic mutations in the gene can result in a non-functioning version of the protein being produced. This leads to problems with waste disposal. People diagnosed with GBA-associated Parkinson’s have earlier onset of symptoms and typically exhibit more progressive symptoms than people without a GBA mutation. But there is a lot of variability between cases. # # |
Ok, so what did the Kiwi and Aussie researchers report in their study?
Right, so as I said above the researchers wanted to explore whether genetic variations could explain the variability in age of onset of people with Parkinson’s, and they chose GBA as a focal point for their analysis. They were seeking to better understand the regulatory control of GBA and identify potential genetic modifiers.
To do this, they firstly conducted an analysis of GBA activity (or more specifically, ‘expression’) across different tissues, using data collected from the genotype‐tissue expression (GTEx) database.
 The GTEx project is an ongoing effort by the Broad Institute of MIT and Harvard to build a comprehensive public resource to study tissue-specific gene activity (or expression) and regulation. They have collected and analysed different aspects of DNA and RNA within samples from 54 non-diseased tissue sites across nearly 1000 individuals, and made this information available to scientists. It is an amazing resource!
The GTEx project is an ongoing effort by the Broad Institute of MIT and Harvard to build a comprehensive public resource to study tissue-specific gene activity (or expression) and regulation. They have collected and analysed different aspects of DNA and RNA within samples from 54 non-diseased tissue sites across nearly 1000 individuals, and made this information available to scientists. It is an amazing resource!
Can you explain what you mean by gene expression?
The DNA is all of your cells is the same, but the proteins being produced can differ between cells. Gene expression is the process by which the instructions encoded in our DNA are converted into a functional product (like a protein). If a cell is producing GCase protein, this means that the GBA gene is being ‘expressed’ within that cell.
Thus, gene expression could be considered a measure of activity for a gene within a particular cell.
This short video of Prof Nick Spitzer will hopefully help you to understand gene expression:
Using the GTEx database, the kiwi/Aussie researchers were able to explore the levels of GBA expression across different brain regions and different peripheral tissues (including the heart, esophagus, stomach, pancreas, and salivary and adrenal glands). At the same time, they were analysing the DNA to identify genetic variants that may be modulating levels of GBA differently across tissue types.
Specifically, they screened 128 common variants within (or near) the GBA gene and found that 56 were associated with decreased GBA expression in 14 of 29 peripheral tissue types.
And this is a really important finding as it suggests that the impact of the GBA gene is not limited to genetic mutations that simply mess with the structure of GCase protein (such as the Parkinson’s-associated variants like N370S, E326K, V394L, L444P).
 The structure of normal GCase protein. Source: ScienceDirect
The structure of normal GCase protein. Source: ScienceDirect
The researcher had found that there are genetic variations within the GBA gene that can directly affect levels of normal functional GCase protein within tissues (reducing the amount of GCase) in the absence of a recognised Parkinson’s-associated GBA variant. Normal levels of normal GCase were being generated in other tissues, but these genetic variations within the GBA gene were potentially affecting normal GCase protein levels in specific tissues.
The researchers proposed that having these particular GBA variants could reduce GBA expression in specific peripheral tissues possibly rendering them susceptible to aspects of Parkinson’s (think alpha synuclein in gut tissues – click here to read a previous SoPD post on this topic). Such data may thereby support “the view of Parkinson’s as a multisystem disorder”.
In addition to this, the researchers found that 73 of the 128 common GBA variants were able to regulate 143 potential modifier genes across the various tissues analysed (including different regions of the brain):
 Source: Wiley
Source: Wiley
Before we go any further, I just want to spell out the potential implications of these findings.
If you look at the image above, you will see in the top left corner, the names of genes that are considered potential modifiers of GBA expression. They vary across different regions, and they differ in their influence – some increase expression (written in red) and others reduce expression (written in blue). If we zero in on one region of the brain called the substantia nigra – an area badly affected by Parkinson’s – you will see that the researchers identified two potential modifiers of GBA expression (AGAP1 & EPS15) that reduce GCase levels.
Now, I want to you consider this: There could be individuals with Parkinson’s that do not have a recognised PD-associated GBA variant, but they might be carrying one of these potential modifiers that lowers the levels of GCase protein in their substantia nigra – possibly making them vulnerable to developing Parkinson’s.
Perhaps some of the GCase targetting therapies currently being clinically tested could be useful for these individuals (even though they do not have recognised Parkinson’s-associated GBA variant).
It is also of interest that the researchers found that 24% of these modifiers were associated with Parkinson’s in some way (for example, genes like DNAJC6 [PARK19] and SYT11 were among the modifiers).
It gives a flavour of the complexity we were discussion above.
 Source: Imperial
Source: Imperial
And that complexity is about to get even more…. well, complex.
You see, next the research looked for potential modifiers of GBA expression outside of the GBA gene. And what they found is rather remarkable.
They identified 6 genetic variants that had strong regulatory impacts on GBA expression. Four of them were expressed in the cortex, while 2 were expressed in the substantia nigra (that Parkinson’s vulnerable region).
But here’s the catch: All of them were on completely different chromosomes to each other.
What is a chromosome?
In a nutshell, a chromosome is a very efficient way of packing a lot of DNA into a cell.
Within most of the cells in your body, DNA is densely coiled into discrete packages called chromosomes. Without such packaging, the stringy DNA molecules would be too long to fit inside the cell. In fact, if you uncoiled all of the DNA molecules in a single human cell and placed them end-to-end, they would stretch for at least 6 feet. And that’s just for one cell – remember that the average human has more than 10 trillion cells in their body!
Fun fact: All of the DNA in your body could stretch to the sun and back….more than 60 times (Source).

The 23 chromosomes of humans (this set is from a male). Chromosome 22 is highlighted. Source: Wikipedia
Chromosomes come in pairs – one set of 23 chromosomes from each parent, giving us a total of 46 chromosomes per cell. All of these pairs reside inside the nucleus of each cell, where their DNA is read and instructions (RNA) are sent off to be made into proteins which performs functions within the cell.
Got it. So some of these potential modifiers of GBA were on different chromosomes?
Exactly. Of the 6 highlighted, no two on the same chromosome.
And none of them on the same chromosome as GBA.
These are long distance modifiers.
And again, consider the implications: if someone with Parkinson’s that does not have a recognised PD-associated GBA variant, but does carry one of these distant potential modifiers that lowers the levels of GCase protein in their substantia nigra, could this possibly make them vulnerable to developing Parkinson’s?
Could these variant explain some of the variability in people with Parkinson’s?
It’s a great question and this is what the researchers wanted to explore in their study.
To assess the clinical impact of their findings, the researchers collected genetic data from 229 people with Parkinson’s (21 of whom had a known GBA mutation). To remove the effect of known GBA mutations, they removed data from the 21 individuals with known GBA mutations. And then they clustered the rest into 3 groups based on the nature of their potential GBA modifier variant (minor to major effect).
And when they looked at the age of symptom onset and age of diagnosis, they found that the 3 groups were significantly different. For example, the average age of diagnosis for the first group (minor variants) was 5.7 years later than group 3 (major variants).
|
# # # RECAP #3: Researchers in New Zealand and Australia have identified genetic variants within the GBA gene (and distant to it) that modify the levels of GCase protein in various tissues (including different areas of the brain). They also found that these variants can impact the age of diagnosis of Parkinson’s. # # # |
Wow. That is some influence. But why is knowing all of these potential GBA modifiers important?
Multiple reasons.
Firstly, it suggests that GCase targetting therapies currently being developed for GBA-associated Parkinson’s may (if found to be effective on PD) have wider use within the affected community than simply on just the folks with known GBA variants.
Second, at The Cure Parkinson’s Trust (where the ridiculously tall and freakishly good looking author of blog works) we are supporting the repurposing of a drug called ambroxol for Parkinson’s. Our hope is that this respiratory medication will have a disease modifying effect on the progression of the condition.
 Source: Skinflint
Source: Skinflint
The results of a recent Phase 2 clinical trial suggest that ambroxol increases levels of GCase protein in the brains of people with Parkinson’s (both with and without GBA variants – click here to read a SoPD post about those results).
In the next phase of clinical testing, we will be seeking to conduct a large placebo-controlled trial in which the participants in the trial will be randomly assigned to be treated with either ambroxol or a placebo. Ideally, we will have evenly balanced groups to provide a fair evaluation of the drug.
But (hypothetically speaking) what happens if the treatment group has a larger number of people with a major GBA modifier variant than the placebo group. Could such a distribution affect the result, even lead to a false negative result?
Hopefully you can see why a better understanding of these genetic modifiers is crucial to the future of clinical trials for Parkinson’s.
So what does it all mean?
If a term like ‘race’ can not be biologically defined due to all of the genetic variability that exists between us, where do we stand on vague labels like “Parkinson’s disease”?
At present we have a collection of genetic risk factors associated with the condition, which allow us to build some variable subtypes. And more recently researchers in New Zealand and Australia have pointed towards some ‘genetic modifiers’ that could be playing an important role in influencing the impact of one of those Parkinson’s associated genes (GBA).
It will be important to replicate the findings of this new study and expand on them in bigger datasets, exploring other aspects of the condition that could be affected by them. A better understanding of the genetic modifiers of Parkinson’s will allow us to conduct more balanced clinical trials, providing a cleaner evaluation of the experimental therapy being evaluated. In addition, it might allow for better stratification of the condition, ideally resulting in better treatment.
Hopefully with more research, some order will come from all of this variability and complexity.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
Further, the author of this post is an employee of the Cure Parkinson’s Trust. The Trust has not asked for this post to be written, and there has been no effort to highlight the work of the Trust over others (perceptions of any bias should be directed to the author). This post has been written by the author solely for the purpose of sharing what the author considers interesting information.
The banner for today’s post was sourced from Tum.

A 2019 scientific study reported: “exposure to pesticides is a risk factor for Parkinson’s Disease.”
Research in the Netherlands and USA have shown a correlation between exposure to pesticides containing GLYPHOSATE and the number of ‘exposed’ people with PD symptoms. Thailand, El Salvador, Sri Lanka, Bermuda, Columbia, Vietnam and Austria have totally banned the use of glyphosate. Germany will ban its use after 2023. The Netherlands and France have banned it’s use in residential areas.
Glyphosate is used on a third of UK crops.
Glyphosate contains a chemical compound, called MPTP. It destroys dopamine-producing brain cells and is the ONLY known proven trigger of Parkinsonism. This has been known for over 30 years!
Despite that, it is not banned in the UK or the EU.
I have never seen Parkinson’s UK, or any other PD support group, campaign about it – even though reducing its use would be an immediate and effective way to reduce PD in the UK.
However, Antonia Staats (Professor Neil Ferguson’s girlfriend), successfully campaigned for the EU to ban the use of neonicotinoid insecticides in 2018, because of their effect on bees!
https://pubmed.ncbi.nlm.nih.gov/30799335/
https://www.sciencedirect.com/science/article/abs/pii/S0304394020303025
Click to access oxford-economics-summary-report-final.pdf
https://www.scientificamerican.com/article/parkinsons-disease-and-pesticides-whats-the-connection/
LikeLike
Hi simonmcksays,
you claim that glyphosate contains MPTP. To the best of my knowledge, this claim is false. Furthermore, I see no connection between your comment and the topic of this post.
zz
LikeLiked by 1 person
I don’t know whether or not MPTP is in glyphosate, although I certainly agree that glyphosate should be banned. But you should not post this information here on a post that has no relationship to the article.
LikeLike
Sorry – feel free to remove my comment.
LikeLike
Check the science.
However, sorry – feel free to remove it.
LikeLike
Hi Simon,
you mention the existence of 6 potential modifiers on different chromosomes which might participate in up- or downregulating GBA. If I am not mistaken, none of these modifiers is part of the exome. Wouldn’t this suggest that whole exome sequencing is no longer sufficient for genetic PD diagnostics?
I have young onset PD and had a PD panel diagnostic done 4 years ago. No pathogenic variants were detected. Recently, I had my whole genome sequenced and since then I am having fun digging around in the raw data. After reading this post and the original research paper I checked my variants of these potential modifiers. I came to the conclusion that only about 20% of people with European ancestry have at least as many of these modifiers in the downregulating version as I have. So I think I finally found something which might contribute to my PD. As a consequence, I doubled my daily dose of ambroxol. My neurologist was not particularly happy when I told him about that, he suggested to wait for the results of the ambroxol studies, but my primary care physician understood and sort of approved my decision. You see, your post had some (bench-to-bedside) impact on me.
Thank you for keeping us updated on PD research!
zz
LikeLike
Hi Zz,
Thanks for the comment and for sharing. Glad you found the post interesting. Yes, exome seq was never going to be the answer, and I for one welcome this new age of genetic modifier research.
If I am completely honest, I find this area really exciting – certainly more than simply identifying novel, rare risk variants. The variants have been useful in pointing towards specific biological pathways that may be involved with PD, and giving rise to agents like ambroxol being clinically tested. But they are only ever going cover (at most) 20% of PD cases. Whereas the genetic modifier research may be able to increase that percentage considerably. It is going to be incredibly complex moving forward, but it will be necessary in order to get a full picture.
And I am hugely impressed with your proactive initiative – go you! (I agree with your neurologist, but I also understand your position).
You might like to share your observations with Prof Antony Cooper at the Garvan in Sydney (https://www.garvan.org.au/people/antcoo). He was one of the senior authors on the paper and I am sure he will be very interested to learn more about this.
Thanks again for the comment.
Kind regards,
Simon
LikeLike