|
# # # # Astrocytes are a non-neuronal cell type in the brain that play an important supportive role – nurturing neurons and helping to maintain homeostasis in the central nervous system. When neurons get damaged or stressed, however, astrocytes can do a Jekyll & Hyde-like transformation and start releasing a toxic substance that helps to kill neurons. This nasty change in the temperament of astrocytes is believed to play a role in neurodegenerative conditions. Exactly what the released toxic substance is has long been a mystery. Until now it seems. But the nature of the presumed substance is something of a surprise to the research community. In today’s post, we will review a new research report that points towards saturated lipids as the mediators of astrocyte-induced toxicity and we will consider what this could mean for future therapies for neurodegenerative conditions. # # # # |
One of my favourite scientists to listen to is Ben Barres.
It is wonderful to go back and watch some of his old videos. Not just because you learn so much from him, but also for the passion that he always had when discussing general science, his research, and other things he believed in.
For example, watch the first 10-15 minutes of this video:
The presentation above was made on January 9th 2017, and despite knowing that he had less than a year to live, you can hear the energy and excitement in his voice for the material he is presenting. He desperately wanted to share the information and to learn what others might think about it.
He was truly an amazing individual.
Even more so, because almost 4 years after he died, Ben is still publishing spectacular research.
Wait. What?
Such is the nature of modern scientific research that many experiments can take 5-6 years to complete and get published.
Recently, researchers who were collaborating with Ben before he died published this report:
 Title: Neurotoxic reactive astrocytes induce cell death via saturated lipids.
Title: Neurotoxic reactive astrocytes induce cell death via saturated lipids.
Authors: Guttenplan KA, Weigel MK, Prakash P, Wijewardhane PR, Hasel P, Rufen-Blanchette U, Münch AE, Blum JA, Fine J, Neal MC, Bruce KD, Gitler AD, Chopra G, Liddelow SA, Barres BA.
Journal: Nature. 2021 Oct 6. Online ahead of print.
PMID: 34616039
In this report, the investigators were interested in the role of astrocytes in neurodegenerative conditions.
What exactly are astrocytes?
There are different cell types in the brain. Neurons are one of the main ‘prima donnas’, and they are usually get all the attention, because they are believed to be the primary engines in cognitive activity. But in addition to these there are numerous supporting (or “helper”) cells, which include microglia, oligodendrocytes, or astrocytes.
 Different types of cells in the brain. Source: Dreamstime
Different types of cells in the brain. Source: Dreamstime
Microglia are the resident immune cells in the brain, keeping an eye out for disease causing pathogens and cleaning up when there is damage. Oligodendrocytes insulate neurons and help with rapid transmission of signals across the brain (Click here to read a recent SoPD post on oligos).
Astrocytes are really the key supportive cells for neurons.
They provide them with nutrients and make sure the environment surrounding the neurons is balanced and agreeable. They are plentiful and work extremely hard to keep neurons happy and functioning.
 A human astrocyte. Source: Wikipedia
A human astrocyte. Source: Wikipedia
What do astrocytes have to do with neurodegenerative conditions?
Well, while astrocytes have this supportive role for neurons, they will occasionally turn on them and can do them serious harm. This situation often occurs when the neurons are stressed and perceived to be beyond rescue.
At this point, astrocyte become activated (often termed ‘reactive’) and they are believed to release a mystery agent that helps to encourage the neuron to die. Such a scenario is believed to occur in neurodegenerative conditions like Parkinson’s.
 Source: Sciencedirect
Source: Sciencedirect
Exactly what that lethal substance is has been a mystery for a long time.
To resolve the nature of the mystery factor, Prof Barres and collaborators undertook a series of experiments in which they compared the secretions of normal astrocytes grown in cell culture to the secretions of reactive astrocytes grown in culture.
What is meant by “reactive” astrocytes?
Reactive astrocytes are astrocytes that have undergone “morphological, molecular, and functional changes in response to pathological situations in surrounding tissue” (Source – a very good review on this topic). Reactive astrocytes differ from their non-reactive, resting counterparts in that they swell up in size and the number of branches can increase.
See the image below, in which the non-reactive astrocyte – lean and relaxed (left panel) – are compared to reactive astrocytes – fat and angry (me!) – in the right hand panel:
 Source: Sciencedirect
Source: Sciencedirect
To achieve reactive astrocytes, the researchers treated the astrocytes with a mix of cytokines.
What is a cytokine?
Cytokines (from the Greek: kýtos meaning ‘container, body, cell’; and kī́nēsis meaning ‘movement’) are small proteins that are secreted by certain cells in the body and they have an effect on other cells. Cytokines are a method of communication for cells.
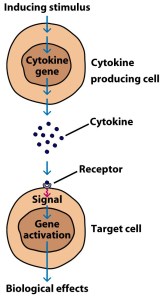
How cytokines work. Source: SBS
In their experiments, the researchers treated their astrocytes with pro-inflammatory cytokines (such as C1q, Interleukin-1α, and tumor necrosis factor), which caused the astrocytes to do their Jekyll & Hyde-like transformation and become extremely reactive.
The investigators then collected the solution that the reactive astrocytes were growing in and compared it to the solution that normal relaxed astrocytes were growing in. They analysed the different solutions for any secreted molecules.
It is important to appreciate here that scientists have been conducting these types of experiments for years and they have never identified any conclusive “mystery toxic agent”. And Prof Barres’ team also found nothing.
But in all of these previous experiments, the scientists were looking mainly at proteins.
The key difference in this new study, is that the researchers noticed an abundance of lipoproteins in the solution from the reactive astrocyte cultures.
What are lipoproteins?
Lipoproteins are substances that are combinations of protein and fat. They are typically spherical in shape and they are involved in the transportation and redistribution of lipids (fats) in the body.
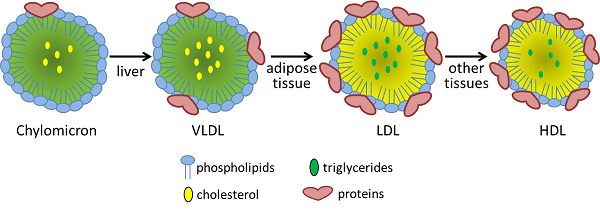 Different types of lipoproteins. Source: Study
Different types of lipoproteins. Source: Study
The high levels of lipoproteins in the reactive astrocyte culture solution suggested that these constructs could be the potential bearers of astrocyte-induced toxicity, and the researchers decided to test this idea.
They analysed the levels of 1,501 lipids from different solutions of normal astrocytes and reactive astrocyte cultures, and they found that in the latter there were many more long-chain saturated fatty acids.
What are long-chain saturated fatty acids?
Fatty acids represent the basic building blocks of lipids and fats in our body. They are made up of chains of carbon atoms, and they play important roles in our bodies. We have recently discussed them in another SoPD post (Click here to read that post).
Fatty acid can be classified by the number of carbon atoms in their tails. Long-chain fatty acids are those with 14 or more carbons.
 A long-chain fatty acid. Source: Zoeharcombe
A long-chain fatty acid. Source: Zoeharcombe
And each carbon atom has a number of hydrogen atoms attached to it – the exact number of hydrogen atoms on each carbon determines whether the fat is saturated or unsaturated. If a fatty acid chain contains the maximum level of hydrogen atoms possible, it is considered a saturated fatty acid. By comparison, in unsaturated fatty acids some of the hydrogen atoms are missing. These are replaced with double bonds between the carbon atoms.
 Source: Expii
Source: Expii
Ok, so there were more long-chain saturated free fatty acids in the reactive astrocyte culture solution. So what?
Well, given the high levels of long-chain saturated fatty acids in the solution from reactive astrocyte cultures, the researchers questioned whether they could be the mystery toxic agent we discussed above.
To test this, the scientist genetically engineered astrocytes that had no ELOVL1 gene in their DNA.
What is the ELOVL1 gene and why is it important?
A gene is a section of DNA that provides the instructions for producing a particular protein. In the case of the ELOVL1 (“Elongation of very long chain fatty acids protein 1“) gene, it offers the instructions for making an enzyme that lengthens saturated fatty acids. It generates (as the label on the can suggests) very long chain fatty acids (up to 30 carbons long).
When the researchers grew astrocytes with no ELOVL1 in culture, and they tried to switch them to reactive astrocytes,… guess what happened?
They didn’t turn reactive?
No.
They still turned reactive, but they released much lower levels of long-chain saturated fatty acids.
AND, the solution from reactive astrocytes with no ELOVL1 was significantly less toxic to the other cells in the culture (compared to reactive astrocytes with ELOVL1 still present).
This result suggested to the investigators that lipoapoptosis might be the cause of cell death.
Lipo-what now?
Lipoapoptosis is a form of apoptosis (or programmed cell death) caused by exposure to an excess of fatty acids.
Apoptosis is a process of programmed cell death by which a cell will undergo an orderly sequence of events which ultimately leads to death of that cell. It is a very clean, orderly, and efficient way of removing old cells.
 Apoptosis. Source: Genome
Apoptosis. Source: Genome
The remains of the cell can be cleaned up and absorbed by surrounding cells.
During lipoapoptosis, the high levels of saturated fatty acids activate something called the PERK (protein kinase R-like endoplasmic reticulum kinase) endoplasmic reticulum stress response pathway, which then leads to cell death via two death activators:
- PUMA (p53 upregulated modulator of apoptosis; encoded by Bbc3), or
- CHOP (C/EBP homologous protein; Ddit3)
These two proteins are killers of cells. If the cell starts producing high levels of either PUMA or CHOP, it’s basically game over.
If these two proteins were the activators of apoptosis in response to the high levels of long-chain saturated free fatty acids in the reactive astrocyte culture solution, then the researchers assumed that removal of these proteins from cells should make them resistant to this astrocyte-induced lipoapoptosis.
And this is what they found… in the case of PUMA.
When the investigators removed PUMA from cells and exposed them to reactive astrocyte culture solution, the cells did not die.
BUT, when they removed CHOP, the investigators found that the cells were still vulnerable to lipoapoptosis via the reactive astrocyte culture solution.
The researchers concluded that “these results suggest that saturated lipids mediate
the toxic activity of reactive astrocytes” and they proposed that reducing ELOVL1 levels could be a means of blocking the production of these toxic lipids.
Are there any drugs targeting ELOVL1?
At present, no.
And it may not be a very viable drug target. As others have pointed out “some long-chain fatty acids have beneficial effects, so if you interfere with their synthesis, you might have deleterious effects, for example disrupting myelin integrity” (Source).
But more research is required in this area of astrocyte-induced neurotoxicity. In some of the experiments of the study reviewed today, the effects were only partial suggesting other pathways might also be involved. In addition, more work in in vivo (or animal) models of neurodegenerative conditions is required to determine if the effect is biologically relevant (most of the experiments in this report were conducted in vitro – in cell culture).
So what does it all mean?
Researchers have recently identified an unexpected potential neurotoxic messenger that is released from astrocytes when they become reactive to stressed neurons: Saturated lipids can induce a form of programmed cell – called lipoapoptosis – that could be playing a role in neurodegenerative conditions.
If these new results can be independently replicated, it would point towards a novel means of cell death not previously considered in neurodegenerative conditions, which would represent a significant step forward. And if astrocytes are actively involved with all neurodegenerative conditions, such a therapeutic angle could potentially have a very broad impact.
While not dealing with the specific underlying cause of a condition like Parkinson’s or Alzheimer’s, inhibiting the release of astrocyte-originating neurotoxic agents could represent a means of slowing disease progression and give stressed neurons a chance to heal and return to health. Such a therapeutic could be used in combination with other agents (such as neurotrophic factors or more specific disease halting therapies).
As with much of the research discussed here, it will be interesting to see where this work leads.
# # # # # # # # #
Ben Barres (the senior scientist on the paper we have reviewed today) died on the 27th December, 2017. For the previous 20 months he had been dealing with pancreatic cancer.
When talking about his life, he said:
“I lived life on my terms: I wanted to switch genders, and I did. I wanted to be a scientist, and I was. I wanted to study glia, and I did that too. I stood up for what I believed in and I like to think I made an impact, or at least opened the door for the impact to occur. I have zero regrets and I’m ready to die. I’ve truly had a great life” (Source– take a moment to learn a little more about a fascinating individual).
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced and adapted from Wikipedia

Great post Simon. This is super interesting science and Barres and scientific offspring are /were great folks.
Peter
LikeLike