|
# # # # Oligodendrocytes are a supportive cell in the brain. They produces a protective coating – called myelin – which insulates the primary projecting branch of neuronal (called axons) and aids in the rapid transmission of signals in the brain. Medical conditions associated with these cells include the inflammatory condition of multiple sclerosis. It is fair to say that oligodendrocytes have been largely neglected in the context of Parkinson’s research. But that might be about to rapidly change. In today’s post, we will discuss what oligodendrocytes do, and explore new research that may point towards a role for oligodendrocytes in Parkinson’s. # # # # |
 Source: Youtube
Source: Youtube
The name “oligodendrocyte” comes from the Greek.
‘Oligos’ (ολιγος) meaning small or little; ‘dendros’ (δενδρος) meaning tree or bush; and ‘kytos‘ (κύτος) meaning ‘cavity’ or ‘cell’.
The name was given to cells in the brain with small branches by the Spanish neuroscientist Dr Pío del Río Hortega (1882–1945):
 Pío del Río Hortega. Source: RTVE
Pío del Río Hortega. Source: RTVE
Del Río Hortega developed a staining method that allowed him to differentiate what the great Ramón y Cajal referred to as the “third element” of cells in the brain. Initially calling them “interfascicular cells”, Del Río Hortega later changed the name to oligodendroglia.
 Title: La glía de escasas radiaciones (oligodendroglía).
Title: La glía de escasas radiaciones (oligodendroglía).
Author: Del Río-Hortega P.
Journal: Bol. Real Soc. Esp. Hist. Nat.(1921). 21, 63–92.
PMID: N/A
(For those who would like to read more about Pio del Río-Hortega – click here for an excellent review)
Cool. But what do oligodendrocytes do?
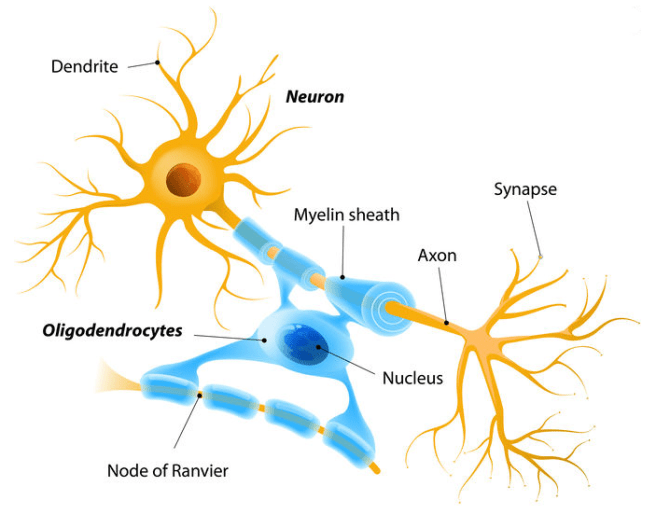
Oligodendrocytes are a type of glial cell (or ‘non-neuronal cells’) in the brain. Their main function is to provide insulation to the main projection of neurons (these branches are called axons). The insulation provided by oligodendrocytes is called the myelin sheath.
 Source: ElectroSpinning
Source: ElectroSpinning
Oligodendrocytes extend many processes, each of which contacts and repeatedly envelopes a stretch of axon with layers of myelin. On the same axon, adjacent sections of myelin will belong to different oligodendrocytes. And a single oligodendrocyte can have 40-50 processes making contact with axons.
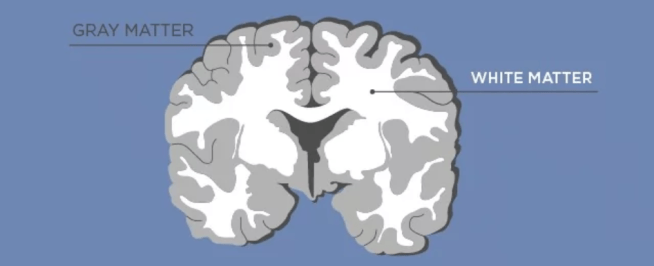
You know how people talk about gray matter and white matter in the brain?
Well, the myelin surrounding all of the axons is what make the white matter white (Gray matter being where the main body (or soma) of neurons is).
 Source: TN
Source: TN
So the oligodendrocyte is protecting the main branch of the neuron? The axon?
Not just protecting it.
Myelin plays a very important role in the transmission of signals around the brain.
You may have noticed in the image above small gaps between sections of myelin. These are called nodes of Ranvier.
These gaps allow signals (or action potentials) to pass along the axon faster.
 Source: Britannica
Source: Britannica
In the absence of myelin and nodes of ranvier, the signal would slow down and lose its strength very quickly (see the animation below).
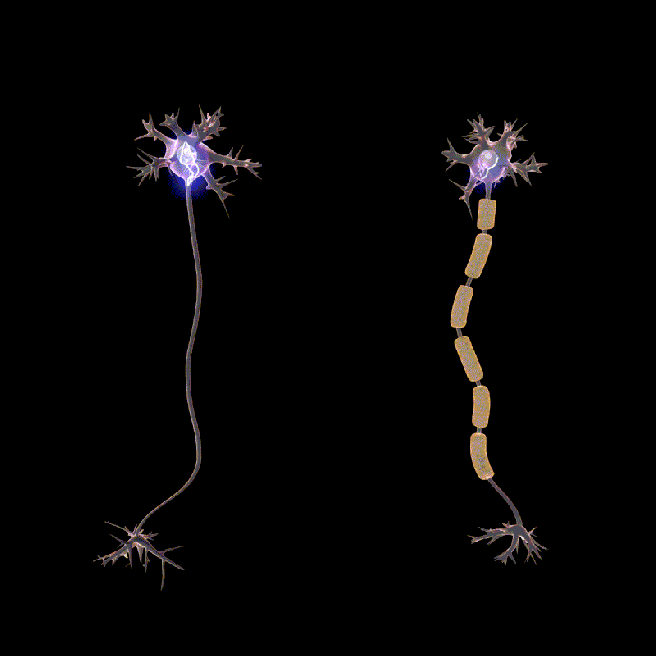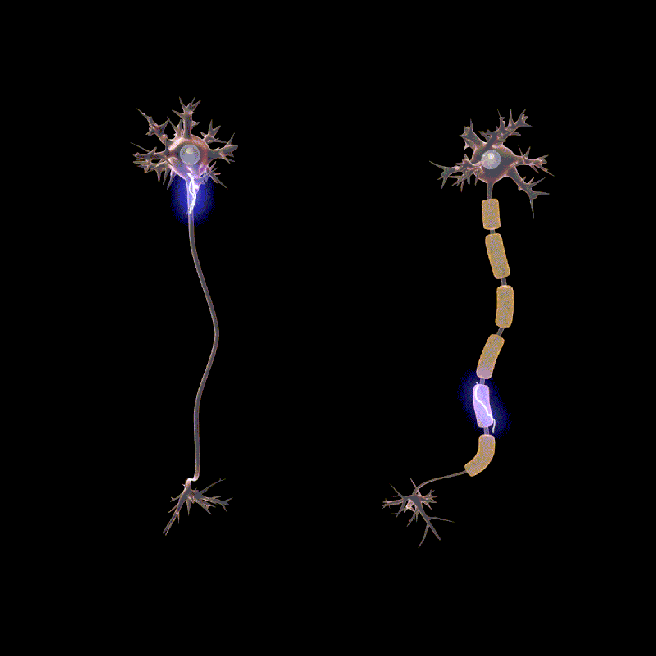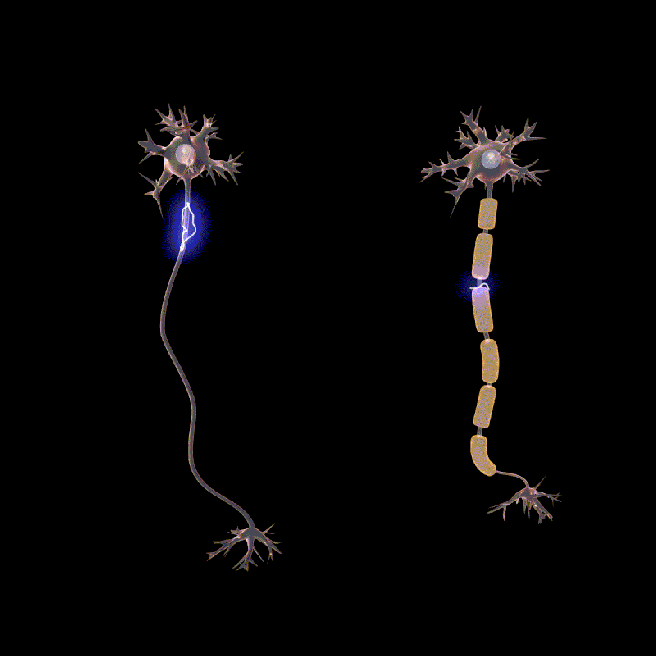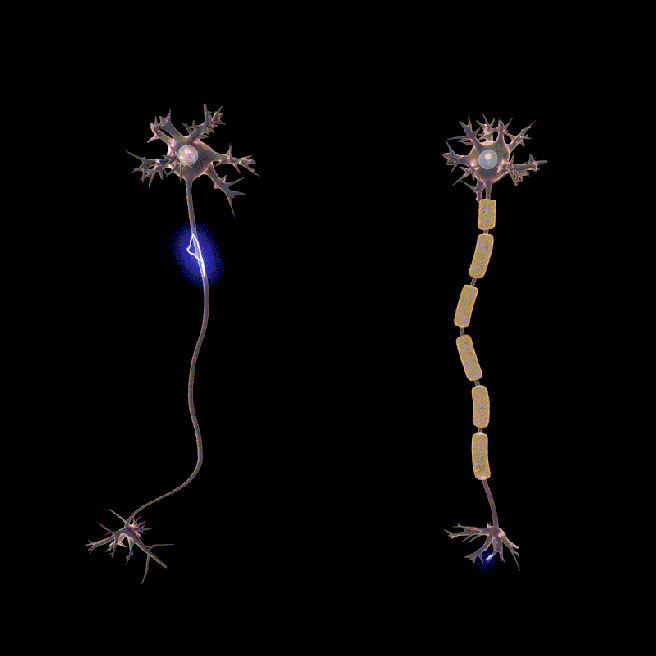 Neurons don’t usually rotate. Source: Wikipedia
Neurons don’t usually rotate. Source: Wikipedia
|
– SMALL SIDE NOTE HERE – Oligodendrocytes as a cell type fascinate me for one simple reason: their place in the world. How on earth did this class of cells ever evolve? How did they come to have such a complicated role (and importance) in cognitive processing? When did they collectively think we’re going to wrap these axons with a bit of waxy insulation, but leave just enough of a gap between sheaths for stuff to happen? Other cells are happy just to connect with each other, but oligodendrocytes obviously wanted to be different. I am in awe of the biology of it. – |
The importance of myelin and nodes of ranvier is apparent in demyelinating conditions like multiple sclerosis (or MS). Underlying this disease is a cell mediated loss of the insulating mylein on axons. As that myelin is lost, a range of symptoms start to show themselves. Symptoms can include double vision, blindness in one eye, muscle weakness and trouble with sensation or coordination.
Like Parkinson’s, there is no cure for MS.
This video explains MS:
|
# RECAP #1: Oligodendrocytes are a type of glia (non-neuronal) cell in the brain that functions by insulating the main projections of neurons with a protective coating, called myelin sheath. This insulation aids in the transmission of signals around the brain. Issues oligodendrocytes and myelin sheath underlie the demyelinating condition known as multiple sclerosis (or MS). # |
This is all very interesting, but what do oligodendrocytes have to do with Parkinson’s?
Not much.
At least that was what we used to think.
The aggregation of alpha synuclein protein (the assumed villan in PD research) has been observed in oligodendrocytes in the Parkinsonian brain (click here to read a report about this), but “glial cell” accumulation of alpha synuclein is typically associated more with “multiple systems atrophy” (or MSA – a condition very similar to Parkinson’s, but distinctly different – click here to read a previous SoPD post on MSA).
Parkinson’s has long been considered a ‘gray matter’ condition, affecting neuronal populations like dopamine neurons.
It is fair to say that oligodendrocytes have not really been on the radar for many Parkinson’s researchers.
And that is why several recent reports have resulted in the scratching of many heads.
What did the research reports suggest?
So last year a report was published by researchers associated with the International Parkinson Disease Genomics Consortium (or IPDGC – click here to read a previous SoPD post about this group).
This is the report in question:
 Title: Moving beyond neurons: the role of cell type-specific gene regulation in Parkinson’s disease heritability.
Title: Moving beyond neurons: the role of cell type-specific gene regulation in Parkinson’s disease heritability.
Authors: Reynolds RH, Botía J, Nalls MA; International Parkinson’s Disease Genomics Consortium (IPDGC); System Genomics of Parkinson’s Disease (SGPD), Hardy J, Gagliano Taliun SA, Ryten M.
Journal: NPJ Parkinsons Dis. 2019 Apr 17;5:6.
PMID: 31016231 (This report is OPEN ACCESS if you want to read it)
In this study, the researchers wanted to examined whether genes (or regions of DNA) associated Parkinson’s were more (or less) active in different brain cell types.
What do you mean ‘different brain cell types’?
Above we have been discussing oligodendrocytes, but there are different cell types in the brain. The main ones are neurons, astrocytes, oligodendrocytes, or microglia.
 Different types of cells in the brain. Source: Dreamstime
Different types of cells in the brain. Source: Dreamstime
Neurons are the main cell involved in cognitive activity, they are the work horses of cognitive activity. Astrocytes are one of the ‘helper cells’ in the brain. They provide nutrients to neurons and make sure the environment surrounding the neurons is balanced and supportive.
Microglia are another type of the helper cells in the brain. They act as the resident immune cells. When infection or damage occurs, the microglia become ‘activated’ and start cleaning up the area. And then we also have oligodendrocytes (discussed above).
For a long time, neurons have been the focus of much neurodegenerative research. The science world has viewed neurons as the prima donnas and the rest of the cell types as a mix of back up singers.
So the researchers wanted to know if genes associated Parkinson’s were more (or less) active in one of these types of cells?
Exactly. They were asking questions like “does a Parkinson’s-associated gene like LRRK2 has higher levels of activty in neurons or astrocytes?”
And what did they find?
Curiously, they found that genetic variations associated with Parkinson’s were not enriched in global and regional brain annotations or brain-related cell-type-specific annotations. Likewise, we found no enrichment of PD susceptibility genes in brain-related cell types.
A negative result?
No, the researchers changed tack and looked at Parkinson’s-associated genes linked to specific functions. They found that gene associated with lysosomal function (a component of waste disposal and recycling) had higher levels of activity in astrocytes, microglia, and oligodendrocytes.
This finding suggests that lysosome function is different in the helper cells.
It raises the idea that the molecular risk factors associated with Parkinson’s may not lie entirely in those cell types that display the characteristic pathology of Parkinson’s (think neurons with Lewy bodies, etc), but rather in some of the helper cells.
Perhaps the backup singers not supporting the prima donnas like they should be?
But what about oligodendrocytes specifically?
Yes, so they (in particular) have popped up on everyone’s radar more recently due to research published this year.
Reynolds et al (the report discussed above) shifted the attention towards the glial cell types, and then in April (2020) this report published:
 Title: Genetic identification of cell types underlying brain complex traits yields insights into the etiology of Parkinson’s disease.
Title: Genetic identification of cell types underlying brain complex traits yields insights into the etiology of Parkinson’s disease.
Authors: Bryois J, Skene NG, Hansen TF, Kogelman LJA, Watson HJ, Liu Z; Eating Disorders Working Group of the Psychiatric Genomics Consortium; International Headache Genetics Consortium; 23andMe Research Team, Brueggeman L, Breen G, Bulik CM, Arenas E, Hjerling-Leffler J, Sullivan PF.
Journal: Nat Genet. 2020 Apr 27. [Epub ahead of print]
PMID: 32341526 (A preprint of the report is available to read by clicking here)
In this study, the researchers pooled together a huge amount of genetic data from different psychiatric disorders and neurological conditions. They then integrated those results with single-cell data from the entire nervous system – samples of each type of cell in multiple regions of the brain had their RNA sequenced and identified, allowing for a representation of which genes are active in each cell type (this is referred to as single cell sequencing).
In this fashion, the researchers could explore which genes are more (or less) active in each cell type of the brain across all of the neurological conditions (including Parkinson’s).
And to their surprise, Parkinson’s gave them one of the strangest results of their study: oligodendrocytes!
When they looked at genes that were particularly active in the substantia nigra part of the brain (the region where the dopamine neurons reside, and an area of the brain badly affected by Parkinson’s), one of the biggest differences that they saw between unaffected control brains and PD brains was oligodendrocyte-related genes were very active.
In the image below, you will see oligodendrocyte-related genes (listed across the bottom) are rather elevated (red bar on the left side of the bar lines) across multiple datasets (listed down the right in grey boxes). As expected, dopamine neuron-related genes are reduced (blue bars on the far left side of the bar lines).
 Source: BioRxiv
Source: BioRxiv
And one interesting detail in this image, is that the top lines represents different stages of Parkinson’s (Braak stage 1-2, Braak stage 3-4, & Braak stage 5-6). Oligodendrocyte-related genes are elevated across all of these stages (even stage 1-2, before dopamine neurons become affected by the condition).
This result kind of left the researchers befuddled and curious.
They concluded their study by writing:
“Our findings suggest that alterations in oligodendrocytes occur at an early stage of disease, which precedes neurodegeneration in the substantia nigra, arguing for a key role of this cell type in Parkinson’s disease etiology”.
|
# # RECAP #2: Analysis of gene activity across different cell types points towards non-neuronal cells having an important role in Parkinson’s. Specifically, oligodendrocyte-related genes seem to be very active across all stages of the condition. # # |
Has anyone else reported this oligodendrocyte finding?
Yes, independent researchers have reported a very similar result.
This manuscript was made available around the same time as the journal article above was published:
 Title: A human single-cell atlas of the Substantia nigra reveals novel cell-specific pathways associated with the genetic risk of Parkinson’s disease and neuropsychiatric disorders
Title: A human single-cell atlas of the Substantia nigra reveals novel cell-specific pathways associated with the genetic risk of Parkinson’s disease and neuropsychiatric disorders
Authors: Devika Agarwal, Cynthia Sandor, Viola Volpato, Tara Caffrey, Jimena Monzon-Sandoval, Rory Bowden, Javier Alegre-Abarrategui, Richard Wade-Martins, Caleb Webber
Database: bioRxiv 2020.04.29.067587 (This manuscript is available to read by clicking here)
In this study, the researchers conducted a similar study to those described above (single cell analysis and applying disease-related genetics to them) and found that “genetic risk in Parkinsons is indeed associated with dopamine neurons, but also with oligodendrocytes, giving new insights into the causes of Parkinson’s”.
And they added that “By contrast, we find no association between PD risk and microglia, suggesting that neuroinflammation is not a key causal process in Parkinson’s”. They also found that the activity of the Parkinson’s associated gene LRRK2 was significantly higher in oligodendrocyte precursor cells than any other cell type in the substantia nigra.
Interesting. But do oligodendrocytes have a close relationship with dopamine neurons?
So this is one of the really puzzling parts of all of this research pointing at oligodendrocytes:
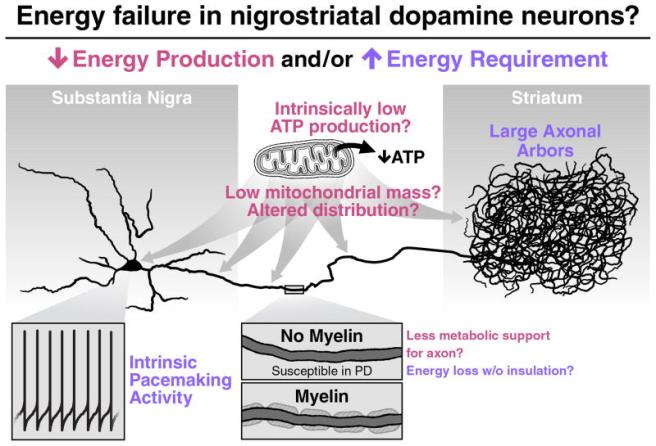
Dopamine neurons are rather sparcely insulated by myelin (Click here to read more about this).
Myelin insulation is important because it decreases the energy required by neurons for transmitting signals. The more myelin, the less energy a cell needs to use to send a signal. And as a result, poorly myelinated dopamine neurons may really be struggling and stressed over time.
Thus, dopamine neurons may be more susceptible to external stressors (like shock or pathogens) due to their increased energy demands (dopamine neurons have VERY long axons).
Source: PMC
And this weakly myelinated effect has been seen in other cell types in Parkinson’s, not just dopamine neurons. For example, this report in which the investigators looked at cardiac tissue in Parkinson’s:
 Title: Unmyelinated axons are more vulnerable to degeneration than myelinated axons of the cardiac nerve in Parkinson’s disease.
Title: Unmyelinated axons are more vulnerable to degeneration than myelinated axons of the cardiac nerve in Parkinson’s disease.
Authors: Orimo S, Uchihara T, Kanazawa T, Itoh Y, Wakabayashi K, Kakita A, Takahashi H.
Journal: Neuropathol Appl Neurobiol. 2011 Dec;37(7):791-802.
PMID: 21696416
In this study, the researchers reported that the percentage of unmyelinated axons were significantly lower in samples of cardiac tissue from people who passed away with Parkinson’s compared to those of unaffected control samples.
In addition, the investigators reported that the “overwhelming majority” of axons with alpha synuclein aggregates in these samples were unmyelinated axons. Thus, unmyelinated axons may be more vulnerable to degeneration than myelinated axons.
And all of this could explain why oligodendrocyte-related genes become more active in Parkinson’s – perhaps there is more demand for their sparce support from the ‘prima donna’ dopamine neurons early in the condition.
Obviously, more research on oligodendrocytes in Parkinson’s is required.
So what does it all mean?
Recently researchers have presented results that suggest a class of helper cells in the brain – called oligodendrocytes – may have a more important role in Parkinson’s than previously thought
It is important to appreciate that this view could have significant implications for many aspects of Parkinson’s research, from disease modelling (should we really be focusing on the cell type that demonstrates the highest levels of α-synuclein aggregates?) to drug development (should we be targeting specific types of cells therapeutically – and not necessarily dopamine neurons?). It also really get to the heart of our current understanding of what “Parkinson’s” is (“are oligodendrocytes the silent villan hiding in plain site?”).
Ten years ago everyone was focusing on dopamine neurons. Now, perhaps not so much.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from MSsoc.


4 thoughts on “Oligodendrocytes?!?”