The word ‘Kainos‘ comes from ancient Greek, meaning ‘new’ or ‘fresh’.
A company in South Korea has chosen to use this word as their name.
Why?
In today’s post we will discuss a clinical trial that started this week that is taking a ‘new and fresh’ approach to treating Parkinson’s disease.

Enchanting country. Source: Eoasia
South Korea is an amazing place, with a long and proud history of innovation and technological development. This week a biotech company there called Kainos Medicine has added itself to that history by initiating a clinical trial that takes a new approach to treating Parkinson’s disease.
As Kainos Medicine points out on their website, the current treatment options for Parkinson’s disease function by alleviating symptoms, for example L-dopa simply replaces the lost dopamine rather than treating the underlying disease. Kainos’s new experimental treatment, called KM-819, is trying to help in a different way: it is trying to slow down the cell death that is associated with Parkinson’s.
How does it do that?
KM-819 is an inhibitor of Fas Associated Factor 1 (or FAF1).
And what is FAF1?
Fas Associated Factor 1 is a protein that interacts with and enhances the activity of a protein on the surface of cells with the ominous name: Fas Cell Surface Death Receptor…and yes, the use of the word ‘death’ in that name should give you some indication as to the function of this protein. When Fas Cell Surface Death Receptor gets activated on any given cell, things have definitely taken a turn for the worse for that particular cell.
Fas Cell Surface Death Receptor (also called CD95) is an initiator of apoptosis.
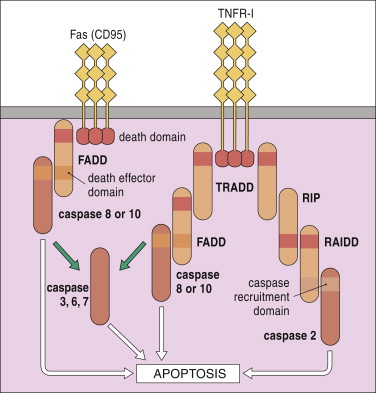
FasSource: Sciencedirect
What is apoptosis?
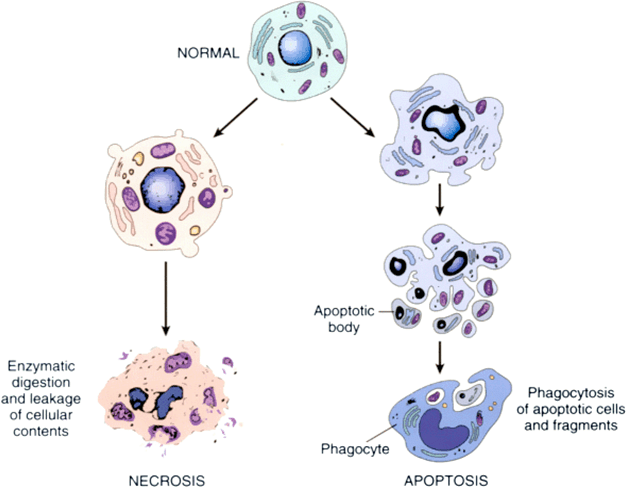
Apoptosis (from Ancient Greek for “falling off”) is the process of programmed cell death – a cell initiates a sequence of events that result in the cell shutting down and dying.
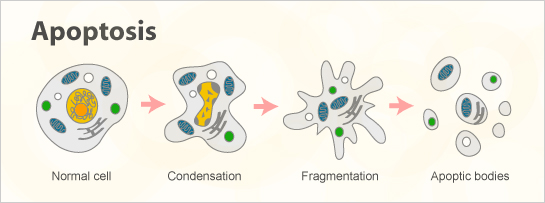
The process of apoptosis. Source: Abnova
Apoptosis is a very clean and organise process of a cell being removed from the body, with it eventually being broken down into small units (called apoptotic bodies) which are consumed by other cells.
Sounds interesting, but what research has been done on FAF1 and Parkinson’s disease?
Back in 2008, this research report was published:
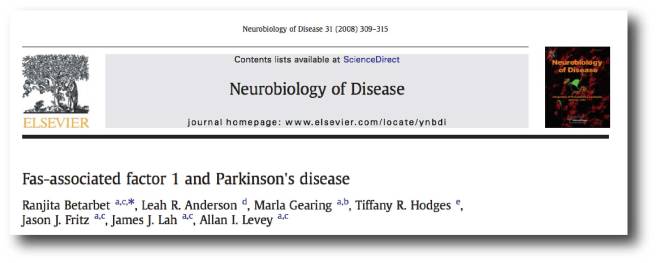
Title: Fas-associated factor 1 and Parkinson’s disease.
Authors: Betarbet R, Anderson LR, Gearing M, Hodges TR, Fritz JJ, Lah JJ, Levey AI.
Journal: Neurobiol Dis. 2008 Sep;31(3):309-15.
PMID: 18573343 (This article is OPEN ACCESS if you would like to read it)
The researcher who conducted this study noticed that the FAF1 gene was located in the ‘PARK 10’ region of chromosome 1. PARK regions are areas of our DNA where mutations (or disruptions to the sequence of DNA) can result in increased vulnerability to Parkinson’s disease (there are currently at least 20 PARK regions). PARK 10 is a region of DNA in which mutations have been associated with late-onset Parkinson’s disease. The scientists thought this was interesting and investigated FAF1 in the context of Parkinson’s disease.
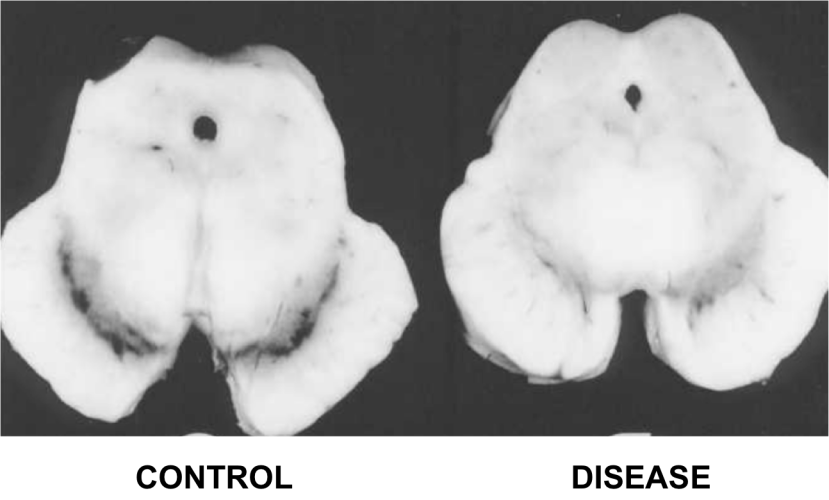
When they looked at postmortem brains, the researchers found that FAF1 levels were significantly increased in brains from people with Parkinson’s disease when compared to brains from healthy control cases. In addition, increased levels of FAF1 exaggerated the cell death observed in different cell culture models of Parkinson’s disease, suggesting an important role for FAF1 in sporadic Parkinson’s disease.
NOTE: More recently, a closer analysis of the PARK10 region resulted in a shrinking of the area which resulted in FAF1 falling outside the PARK10 domain (click here and here to see that research). We are currently not sure if genetic variations in the FAF1 gene infer vulnerability to PD.
This initial work led others to researching FAF1 in the context of Parkinson’s disease and in 2013 this research report was published:

Title: Accumulation of the parkin substrate, FAF1, plays a key role in the dopaminergic neurodegeneration.
Authors: Sul JW, Park MY, Shin J, Kim YR, Yoo SE, Kong YY, Kwon KS, Lee YH, Kim E.
Journal: Hum Mol Genet. 2013 Apr 15;22(8):1558-73.
PMID: 23307929
These researchers found that Parkinson’s associated protein, Parkin (which we have briefly discussed in a previous post) labels FAF1 for disposal. And they found in the absence of Parkin there was a build up of FAF1, making the cells more vulnerable to apoptosis. They followed this finding up by demonstrating that FAF1-mediated cell death was rescued by re-introducing the normal parkin protein. Interestingly, there was no rescue when the mutant parkin protein was re-introduced. These results suggest that normal Parkin acts as an inhibitor FAF1.
To further investigate this finding, the researchers next modelled Parkinson’s disease in genetically engineered mice which had the FAF1 gene removed. They found that the behaviour motor problems and loss of dopamine cells in the brain was significantly reduced in the FAF1 mutant mice, indicating that the FAF1 pathway could be a worthy target for future Parkinson’s disease treatment.
And this and other research has led those same researchers to the clinical trial started in Korea by Kainos Medicine.
So what is the clinical trial all about?
The company will be conducting a phase 1 dose-escalation clinical trial in South Korea, which will evaluate the safety, tolerability, and biochemical properties of their drug KM-819 in 48 healthy adults (click here to read more about the trial).
This is the very first step in the clinical trial process.
The study is split in two parts: Part A is a single dose of KM-819 or a placebo given in ascending doses to participants. And Part B is the same except that multiple ascending doses of the compound will be given to the participants.
The trial will last around six weeks, and – according to the press release – the first subject has just been dosed.
What does it all mean?
Parkinson’s disease is a neurodegenerative condition, which means that certain cells in the brain are dying. Medication that could block that cell death from occurring represents an interesting way of treating the disease and this is what Kainos are attempting to do.
Blocking or slowing cell death is a tricky business, however, because in other parts of the body, cell death is a very necessary biological process. In some areas of our body, cells are born, conduct a particular function and die off relatively quickly. By slowing that cell death in the brain which may be a good thing, we may be causing issues elsewhere in the body, which would be bad.
In addition there has recently been concerns raised about the clinical use of apoptosis inhibitors, such as this study:

Title: Caspase Inhibition Prevents Tumor Necrosis Factor-α-Induced Apoptosis and Promotes Necrotic CellDeath in Mouse Hepatocytes in Vivo and in Vitro.
Authors: Ni HM, McGill MR, Chao X, Woolbright BL, Jaeschke H, Ding WX.
Journal: Am J Pathol. 2016 Oct;186(10):2623-36.
PMID: 27616656
The researchers who conducted this study found that using apoptosis inhibitors on a mouse model of liver disease did stop apoptosis from occurring, but this didn’t save the cells which eventually died via another cell death mechanism called necrosis (from the Greek meaning “death, the act of killing” – lots of Greek in this post!). In necrosis, rather than breaking down in a systematic and organised fashion (apoptosis), a cell will simply rupture and fall apart. Very messy.
Thus there is the possibility with the Kainos drug, KM-819, will protect cells in the Parkinsonian brain from dying via apoptosis, but as the disease continues to progress those cells may become more ill and eventually disappear as a result of necrosis. That said, if the drug can slow down Parkinson’s disease, it would still represent a major step forward in our treatment of the condition!
The connection with Parkin is also very interesting.
It would be wise for future phase 2 and 3 trials – which will test efficacy – to include (or specifically recruit) people with Parkinson’s disease who have mutations in the Parkin gene. This is a very small proportion of the overall Parkinson’s community (approx. 20% of people with early onset PD have a Parkin mutation – click here to read more on this), but if the drug is going to be effective, these would be the best people to initially test it in.
This will be a very interesting set of clinical trials to watch. The phase 1 safety trial will be very quick (6 weeks), and hopefully Kainos Medicine will be able to progress rapidly to a phase 2 efficacy trial. Fingers crossed for positive results.
The banner for today’s post was sourced from Koreabizwire