|
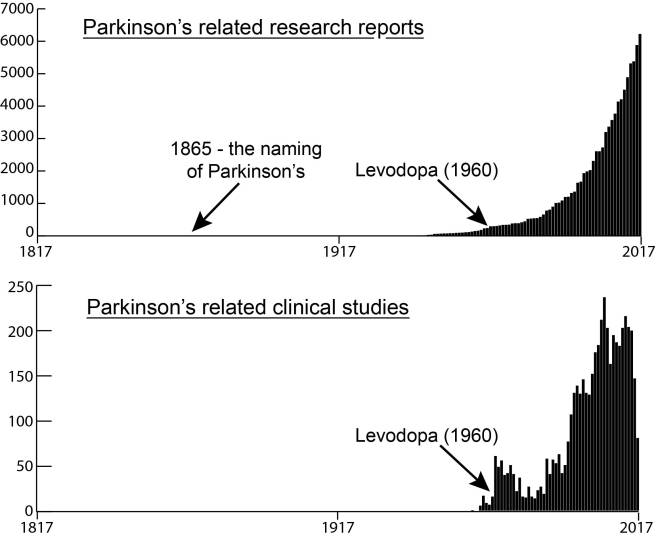
The recent documents filed with the U.S. Securities and Exchange Commission by the biotech firm Prevail Therapeutics provides interesting insight into the bold plans of this company which was only founded in 2017. Even more recent news that the U.S. Food and Drug Administration (FDA) has accepted the company’s Investigational New Drug (IND) application for its lead experimental treatment – PR001 – suggests that this company is not wasting any time. PR001 is a gene therapy approach targeting GBA-associated Parkinson’s. In today’s post, we will discuss what GBA-associated Parkinson’s is, how Prevail plans to treat this condition, and discuss what we know about PR001.
|
 Caterina Fake. Source: TwiT
Caterina Fake. Source: TwiT
The title of this post comes is from a quote by Caterina Fake (co-founder of Flickr and Hunch (now part of Ebay)), but it seemed appropriate.
This post is all about dreaming big (curing Parkinson’s), the struggle to get the research right, and to create a biotech company: Prevail Therapeutics.
What is Prevail Therapeutics?
Prevail is a gene therapy biotech firm that was founded in 2017.
 The company was founded by Dr Asa Abeliovich:
The company was founded by Dr Asa Abeliovich:
 Dr Asa Abeliovich. Source: Prevail
Dr Asa Abeliovich. Source: Prevail
It was set up in a collaborative effort with The Silverstein Foundation for Parkinson’s with GBA (Click here to read a previous SoPD post about this organisation) and OrbiMed (a healthcare-dedicated investment firm).
What does Prevail Therapeutics do?