Our first ever posting here on the SoPD dealt with the curious relationship between the gut and Parkinson’s disease (Click here to see that post). Since then, there have been a string of interesting research reports adding to the idea that the gastrointestinal system may be somehow influencing the course of Parkinson’s disease.
In today’s post we will review the most recent helpings and discuss how they affect our understanding of Parkinson’s disease.

Source: Qz
Interesting fact: The human digestive system is about 26 feet long – approximately 8 meters – from mouth to anus.
Recent research indicates that our brains are heavily influenced by the activities of this food consuming tract. Not just the nutrients that it takes in, but also by the bugs that live within those 26 feet.
Another interesting fact: The human gut hosts tens of trillions of microorganisms, including at least 1000 species of bacteria (which is a guess-timate as we are not really sure how many species there are). They make up as much as 2 kg of your total weight.
And those bacteria have influence!
In December of last year, we reviewed a study in which the researchers demonstrated that mice genetically engineered to display features of Parkinson’s disease performed as well as normal mice if they were raised with reduced levels of bacteria in their gut (either in a germ-free environment or using antibiotics). That study also showed that transplanting bacteria from the gut of people with Parkinson’s disease into mice raised in a germ-free environment resulted in those mice performing worse on the behavioural tasks than mice injected with gut samples from healthy human subjects (Click here to read that post).
Wow, so what new gut research has been reported?
A little bit of history first:
Two years ago, some Danish researchers published this research report:

Title: Vagotomy and Subsequent Risk of Parkinson’s Disease.
Authors: Svensson E, Horváth-Puhó E, Thomsen RW, Djurhuus JC, Pedersen L, Borghammer P, Sørensen HT.
Journal: Annals of Neurology, 2015, May 29. doi: 10.1002/ana.24448.
PMID: 26031848
In their report, the researchers highlighted the reduced risk of Parkinson’s disease following a truncal vagotomy.
So what’s a truncal vagotomy?
A vagotomy is a surgical procedure in which the vagus nerve is cut. It is typically due to help treat stomach ulcers.
The vagus nerve runs from the lining of the stomach to the brain stem, near the base of the brain.

A diagram illustrating the vagal nerve connection with the enteric nervous system which lines the stomach. Source: NCBI
A vagotomy comes in two forms: it can be ‘truncal‘ (in which the main nerve is cut) or ‘superselective’ (in which specific branches of the nerve are cut, which the main nerve is left in tact).

A schematic demonstrating the vagal nerve surrounding the stomach. Image A. indicates a ‘truncal’ vagotomy, where the main vagus nerves are cut above the stomach; while image B. illustrates the ‘superselective’ vagotomy, cutting specific branches of the vagus nerve connecting with the stomach. Source: Score
And what did the Danish scientists find?
Exploring the public health records, the Danish researcher found that between 1975 and 1995, 5339 individuals had a truncal vagotomy and 5870 had superselective vagotomy. Using the Danish National registry (which which stores all of Denmark’s medical information), they then looked for how many of these individuals went on to be diagnosed with Parkinson’s disease. They compared these vagotomy subjects with more than 60,000 randomly-selected, age-matched controls.
They found that subjects who had a superselective vagotomy had the same chance of developing Parkinson’s disease as anyone else in the general public (a hazard ratio (or HR) of 1 or very close to 1).
But when they looked at the number of people in the truncal vagotomy group who were later diagnosed with Parkinson’s disease, the risk had dropped by 35%. Furthermore, when they followed up the truncal group 20 years later, checking to see who had been diagnosed with Parkinson’s in 2012, they found that their rate was half that of both the superselective group and the control group (see table below; HR=0.53). The researchers concluded that a truncal vagotomy reduces the risk of developing Parkinson’s disease.

Source: Svensson et al (2015) Annals of Neurology – Table 2.
Then last year, at the meeting in Berlin, data was presented that failed to replicate the findings in a separate group of people (Sweds).

Title: Vagotomy and Parkinson’s disease risk: A Swedish register-based matched cohort study
Authors: B. Liu, F. Fang, N.L. Pedersen, A. Tillander, J.F. Ludvigsson, A. Ekbom, P. Svenningsson, H. Chen, K. Wirdefeldt
Abstract Number: 476 (click here to see the original abstract – OPEN ACCESS)
The Swedish researchers collected information regarding 8,279 individuals born in Sweden between 1880 and 1970 who underwent vagotomy between 1964 and 2010 (3,245 truncal and 5,029 selective). For each vagotomized individual, they collected medical information for 40 control subjects matched for sex and year of birth (at the date of surgery). They found that vagotomy was not associated with Parkinson’s disease risk.
Truncal vagotomy was associated with a lower risk more than five years after the surgery, but that result was not statistically significant. The researcher suggested that the findings needs to be verified in larger samples.
The results of that study have now been published (this week):

Title: Vagotomy and Parkinson disease: A Swedish register-based matched-cohort study
Authors: Liu B, Fang F, Pedersen NL, Tillander A, Ludvigsson JF, Ekbom A, Svenningsson P, Chen H, Wirdefeldt K.
Journal: Neurology. 2017 Apr 26. pii: 10.1212/WNL.0000000000003961.
PMID: 28446653 (This article is OPEN ACCESS if you would like to read it)
In this report, the researchers suggest that “there was a suggestion of lower risk among patients with truncal vagotomy” and they note that the hazard ratio (or HR) is 0.78 for this group (ranging between 0.55-1.09), compared to the HR of 0.96 (ranging between 0.78-1.17) for all of the vagotomy group combined. And they not that this trend is further apparent when the truncal vagotomy was conducted at least 5 years before Parkinson’s disease diagnosis (HR = 0.59, ranging between 0.37-0.93). These numbers are not statistically significant, so the investigators could only suggest that there was a trend towards truncal vagotomy lowering the risk of Parkinson’s disease.
What are the differences between the studies?
The Danish researcher analysed medical records between 1975 and 1995 from 5339 individuals had a truncal vagotomy and 5870 had superselective vagotomy. The Sweds on the other hand, looked over a longer period (1964 – 2010) but at a smaller sample size for the truncal group (3,245 truncal and 5,029 selective). Perhaps if the truncal group in the Swedish study was higher, the trend may have become significant.
So should we all rush out and ask our doctors for a vagotomy?
No.
That would not be advised (though I’d love to be a fly on the wall for that conversation!).
It is important to understand that a vagotomy can have very negative side-effects, such as vomiting and diarrhoea (Click here to read more on this).
Plus, while the results are interesting, we really need a much larger study for definitive conclusions to be made. You see, in the Danish study (the first report above) the number of people that received a truncal vagotomy (total = 5339) who then went on develop Parkinson’s disease 20 years later was just 10 (compared with 29 in the superselective group). And while that may seem like a big difference between those two numbers, the numbers are still too low to be truly conclusive. We really need the numbers to be in the hundreds.
Plus, it is important to determine whether this result can be replicated in other countries. Or is it simply a Scandinavian trend?
Mmm, interesting. So what does it all mean?
No, stop. We’re not summing up yet. This is one of those ‘but wait there’s more!’ moments.
It has been a very busy week for Parkinson’s gut research.
A German research group published a report about their analysis of the microbes in the gut and how they differ in Parkinson’s disease (when compared to normal healthy controls).

Microbes. Source: Youtube
Regular readers of this blog will realise that we have discussed this kind of study before in a previous post (Click here for that post).
This type of study – analysing the bacteria of the gut – has now been done not just once:

Title: Gut microbiota are related to Parkinson’s disease and clinical phenotype.
Authors: Scheperjans F, Aho V, Pereira PA, Koskinen K, Paulin L, Pekkonen E, Haapaniemi E, Kaakkola S, Eerola-Rautio J, Pohja M, Kinnunen E, Murros K, Auvinen P.
Journal: Mov Disord. 2015 Mar;30(3):350-8.
PMID: 25476529
Nor twice:

Title: Short chain fatty acids and gut microbiota differ between patients with Parkinson’s disease andage-matched controls.
Authors: Unger MM, Spiegel J, Dillmann KU, Grundmann D, Philippeit H, Bürmann J, Faßbender K, Schwiertz A, Schäfer KH.
Journal: Parkinsonism Relat Disord. 2016 Nov;32:66-72.
PMID: 27591074
Not three times:

Title: Colonic bacterial composition in Parkinson’s disease
Authors: Keshavarzian A, Green SJ, Engen PA, Voigt RM, Naqib A, Forsyth CB, Mutlu E, Shannon KM.
Journal: Mov Disord (2015) 30, 1351-1360.
PMID: 26179554
Not even four times:

Title: Intestinal Dysbiosis and Lowered Serum Lipopolysaccharide-Binding Protein in Parkinson’s Disease.
Authors: Hasegawa S, Goto S, Tsuji H, Okuno T, Asahara T, Nomoto K, Shibata A, Fujisawa Y, Minato T, Okamoto A, Ohno K, Hirayama M.
Journal: PLoS One. 2015 Nov 5;10(11):e0142164.
PMID: 26539989 (This article is OPEN ACCESS if you would like to read it)
But FIVE times now (all the results published in the 2 years):

Title: Parkinson’s disease and Parkinson’s disease medications have distinct signatures of the gut microbiome.
Authors: Hill-Burns EM, Debelius JW, Morton JT, Wissemann WT, Lewis MR, Wallen ZD, Peddada SD, Factor SA, Molho E, Zabetian CP, Knight R, Payami H.
Journal: Mov Disord. 2017 Feb 14. [Epub ahead of print]
PMID: 28195358
(And we apologies to any researchers not mentioned here – these are simply the studies we are aware of).
The researchers in the study published this week, however, did something different to these previous studies:

Title: Functional implications of microbial and viral gut metagenome changes in early stage L-DOPA-naïve Parkinson’s disease patients
Authors: Bedarf JR, Hildebrand F, Coelho LP, Sunagawa S, Bahram M, Goeser F, Bork P, Wüllner U.
Journal: Genome Med. 2017 Apr 28;9(1):39.
PMID: 28449715 (This article is OPEN ACCESS if you would like to read it)
The researchers in this study focused their analysis on 31 people with early stage Parkinson’s disease. In addition, all of those subjects were not taking any L-DOPA. The fecal samples collected from these subjects was compared with samples from 28 age-matched controls.
And what did they find?
In the early-stage, L-dopa-naïve Parkinson’s disease fecal samples, the researchers found increased levels of two families of microbes (Verrucomicrobiaceae and unclassified Firmicutes) and lower levels of two other familes (Prevotellaceae and Erysipelotrichaceae). And these differences could be used to reliably differentiate between the two groups (PD and control) to an accuracy of 84%.
In addition, the investigators found that the total virus abundance was decreased in the Parkinsonian participants. The researchers concluded that their study provides evidence of differences in the microbiome of the gut in Parkinson’s disease at a very early stage in the course of the condition, and that exploration of the Parkinson’s viral populations “is a promising avenue to follow up with more specific research” (we here at SoPD are particularly intrigued with this statement!).
So is there a a lot of consensus between the studies? Any new biomarkers?
(Big sigh) Yes….. and no on the consensus question.
The good news is that all of the studies agree that there is a difference between the abundance of different groups of bacteria in the Parkinsonian gut.
BUT only three of the six studies studies demonstrate any agreement as to which groups of bacteria. And those three studies could only agree on one family of bacteria. The recent study (Bedarf et al) agreed with the Scheperjans et al and Unger et al studies in that they all observed found reduced levels of Prevotellaceae bacteria in the gut of people with Parkinson’s disease.
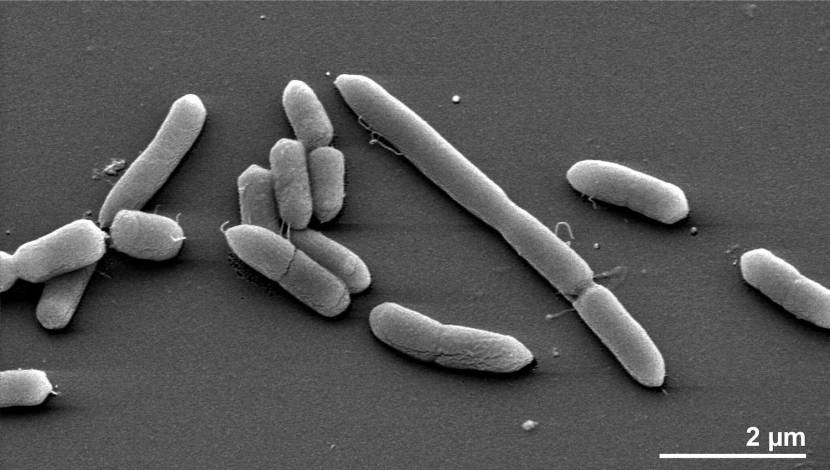
The Prevotellaceae family of bacteria. Source: MindsofMalady
Unfortunately, the reduction in abundance of this particular bacteria does not appear to be specific to Parkinson’s disease, as similar reduced levels have been observed in Japanese multiple sclerosis patients and in autistic children (Click here and here to read more about those studies).
This lack of agreement between the studies with regards to the difference in the abundance of the families of bacteria may reflect the complexity of the gut microbiome. Alternatively, it could also reflect regional differences (the Keshavarzian et al. study was conducted in Chicago, the Bedarf et al and Unger et al studies were in Germany, Scheperjans et al was in Finland, Hill-Burn et al in Alabama, and the Hasegawa et al study was in conducted in Japan).
Either way, it leaves the field lacking agreement as to which families of bacteria should be followed up in future research.
So what does it all mean?
Right, so summing up, researchers are trying to determine what role the gut may play the course of Parkinson’s disease. There is evidence that the nerves connecting the digestive organ to the brain may act as some kind of gate way for an unknown agent or simply a provocative element in the condition. Severing those nerves to the gut appears to reduce the risk of developing Parkinson’s disease.
And the bacteria populating the gut appears to be different in people with Parkinson’s disease, but there does not seem to be consistency between studies, leaving the search for biomarkers in this organ sadly lacking. Maybe it reflects regional differences, perhaps it reflects the complexity of Parkinson’s disease. Hopefully as follow up research into this particular field continues, a consensus will begin to appear. Admittedly, most of these studies are based on single fecal samples collected from individuals at just one time point. A better experimental design would be to collect multiple samples over time, allowing for variability within and between individuals to be ironed out.
Despite all of these cautionary comments, there does appear to be some smoke here. And we will be watching the gut with great interest as more research comes forward.
The banner for today’s post was sourced from the HuffingtonPost
 Me. In a skirt. Eating dirt. Not my worst moment. Source: Drqaisrani
Me. In a skirt. Eating dirt. Not my worst moment. Source: Drqaisrani Something’s missing in my life. Source: Morellisices
Something’s missing in my life. Source: Morellisices Source: Huffington Post
Source: Huffington Post