|
The results of the STEADY-PD Phase 3 clinical trial have been published. This study was evaluating the calcium channel blocker isradipine in 300 people with recently diagnosed Parkinson’s. The results suggest that this blood pressure medication did not impact the course of the disease over the 36 months of testing (based on clinical evaluations; compared to a placebo treated group). While this study did not reach its primary endpoint (the predetermined measure by which the treatment was assessed), a lot can be learned from this trial and it could be of interest to follow up this cohort long-term. In today’s post, we discuss why calcium is important in Parkinson’s, explain what isradipine is, review the results of the STEADY-PD study, and explore what still could be learnt from this study.
|
 Me and Brie. Source: Wikipedia
Me and Brie. Source: Wikipedia
When I turned 25, I realised that my body no longer accepted cheese.
It had become… (how should I put this)… ‘socially embarrassing’ for me to eat cheese.
And this was a extremely serious problem.
You see, to this very day I still really like cheese.
I mean, a bottle of red wine, a baguette and a chunk of brie – is there any better combination in life?
So obviously my body and I had a falling out about the situation. And yes, it got ugly. I wanted things to keep going the way they had always been, so I tried to make things interesting with new and exotic kinds of cheeses, which my body didn’t want to know about it and simply refused to consider. It rejected all of my efforts. And after a while, I gradually started resenting my body for not letting me be who I felt I really was.
We sought help. We tried all kinds of interventions – I was really desperate, I couldn’t live without cheese. But sadly, nothing worked.
And then things went from bad to worse: My body decided that it didn’t have room in its life for yogurt, milk or even ice cream anymore (not even ice cream!!!). Basically no dairy what so ever.
 Something’s missing in my life. Source: Morellisices
Something’s missing in my life. Source: Morellisices
OMG. How did you survive without ice cream?!?
Well, I’ll tell ye – it’s been rough.
All silliness aside though, here is what I know: It is actually very common to develop a lactase deficiency as we get older – lactase being the enzyme responsible for the digestion of whole milk. In fact, between 65-75% of the global population has a reduced ability to digest lactose after infancy (Source: NIH).
 Source: Yogurtinnutrition
Source: Yogurtinnutrition
Here is something else I know: I am not lactose intolerant (one of the few tests that I actually aced in my life), but I do have trouble digesting a particular component of dairy products – which can result in discomfort and ‘socially embarrassing’ situations (one day, over a couple of drinks, I’ll tell you the ‘cheese fondue story’). Curiously, that mystery ingredient is also present in products that have no dairy (such as mayonnaise – it absolutely kills me).
But spare me your tears, dear reader.
If one is forced to drop a particular food group, dairy is not too bad (if I am ever forced to give up wine though, I swear I’ll go postal!!!).
My biggest concern when I dropped dairy, however, was “where was I going to get my daily requirements of calcium?“.
Understand that calcium is rather important.
Why is calcium important?
You will have grown up being told that calcium (from the Latin ‘calx’, meaning lime) is important for your bones, which is correct.
 “Milk is good for your bones” Source: Yogurtinnutrition
“Milk is good for your bones” Source: Yogurtinnutrition
But above and beyond that calcium is absolutely critical for the normal functioning of your entire nervous system.
Every nerve cell in your body is surrounded by a membrane – a skin that holds together the contents of the cell. This membrane also allows the cell to consume resources from the body, and it is critical to the cells ability to communicate with the other cells in the body.
A cell can communicate with its surroundings in several ways:
- Receptors on the outer surface of the cell membrane can receive chemical messengers
- The membrane can form a small bag and release chemical messengers
But for neurons in the brain, the membrane is key to one of the most important (and fastest) methods of communication, which is called an action potential.
What is an action potential?
An action potentials is an electrical impulse that is passed down the branch (or axon) of a neuron to the axon terminals at the tip of the neurons branches, where it stimulates the release of chemical messengers to pass the signal on to the next neuron.
 Source: KhanAcademy
Source: KhanAcademy
And the cell membrane is critical for a neuron’s ability to send this action potential.
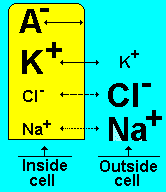
Across the cell membrane, there is a ‘concentration gradient of ions’ (an ion is a molecule with a net electric charge due to the loss or gain of one or more electrons). This gradient simply means that there are more of certain chemical elements outside the cell than inside the cell (or vice versa). You can see this in the image below:
 Source: Wikipedia
Source: Wikipedia
When a neuron is at rest, there is a high concentration of sodium (Na+) ions and chloride (Cl-) ions outside of the neuron (in the extracellular fluid) compared the situation inside the cell (the intracellular fluid) where there is a high concentration of potassium (K+) ions.
 Source: Washington
Source: Washington
During an action potential, as the impulse is moving along the axon (like a wave), this balance is reversed – sodium ions and chloride ions rush inside of the neuron, while potassium moves out. After the impulse has passed, there are active mechanisms that return the balance to normal (high concentration of sodium ions and chloride ions outside of the cell, etc).
This video will explain to you what an action potential is:
When the action potential reaches the axon terminals, it activates (voltage-dependent) calcium channels, which open up and allow calcium to come flowing into the terminal. Calcium is very important for the release of chemical messengers (like neurotransmitters) from the axon terminals. When calcium channels are blocked, neurotransmitter release is inhibited.
The critical thing to understand in the case of an action potential is that without calcium, communication between neurons becomes harder. And this is why calcium is so important in our diet.
 Foods containing high levels of calcium. Source: Animalsaustralia
Foods containing high levels of calcium. Source: Animalsaustralia
If nothing I’ve written here makes any sense, then hopefully this video will help:
|
RECAP #1: Calcium is important. More than just strengthening bones, it plays a critical role in the normal functioning of our nervous system. Without calcium, not much would happen in our brains. In addtion to other functions, calcium is involved in the passing of signals between neurons.
|
Is there any research regarding the importance of calcium in Parkinson’s?
Yes. A great deal in fact.
You see, there is a grand theory of Parkinson’s based around calcium. It goes something like this:
Dopamine neurons (the cells that are particularly vulnerable in brain of a person with Parkinson’s) have a distinct property: They are autonomously active. That is to say, they are continuously generating a low frequency of activity, even in the absence of any direct input. This ‘pacemaker’-like activity is believed to be responsible for the sustained release of dopamine in the striatum (the caudate nucleus and putamen in the image below) which is considered to be necessary for proper functioning.

The projections of the dopamine neurons up into the striatum. Source: MyBrainNotes
This spontaneous electrical ‘pacemaker’ activity is believed to be dependent on calcium channels – particularly L-type calcium channels. If you block these calcium channels, the pacemaker property of dopamine neurons disappears.
Now that is a cute trick, but it is just the start of a much larger potential involvement of calcium in Parkinson’s.
Have a look at this image below:
 A calcium channel. Source: Wikipedia
A calcium channel. Source: Wikipedia
In the image above, there is a representation of a calcium channel embedded in the cell membrane. In the top left side of the image are three word written in red (Benzothiazepine, Phenylalkylamines & Dihydropyridines). These are inhibitors of calcium channels. They represent a class of medication called calcium channel blockers, which are used to treat hypertension (or long term high blood pressure).
Eight years ago, researchers noticed something interesting about calcium channel blockers that specifically target the L-type calcium channels:
 Title: L-type calcium channel blockers and Parkinson disease in Denmark.
Title: L-type calcium channel blockers and Parkinson disease in Denmark.
Authors: Ritz B, Rhodes SL, Qian L, Schernhammer E, Olsen JH, Friis S.
Journal: Ann Neurol. 2010 May;67(5):600-6.
PMID: 20437557 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers collected the medical records of 1,931 people in Denmark who were diagnosed with Parkinson’s between 2001 and 2006. These records were age and sex matched to 9,651 records of healthy controls from the same register. After analysing all of the records, the investigators found that people prescribed with a type of medication called a L-type calcium channel blocker (dihydropyridines) were 27% less likely to develop Parkinson’s.
This finding supported a previous study that found a similar result (Click here to read more about that) and the same result was independently replicated a couple of years later (Click here to read that report).
And the result was also supported by postmortem analysis of the Parkinsonian brain:
 Title: Parkinson’s disease is associated with altered expression of CaV1 channels and calcium-binding proteins.
Title: Parkinson’s disease is associated with altered expression of CaV1 channels and calcium-binding proteins.
Authors: Hurley MJ, Brandon B, Gentleman SM, Dexter DT.
Journal: Brain. 2013 Jul;136(Pt 7):2077-97.
PMID: 23771339 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers looked at where in the human brain the L-type calcium channels were present and in what concentration. They used sections of postmortem brain to carry out this analysis, looking at both healthy specimens as well as those from people who passed away with Parkinson’s.
In the normal brains, the distribution of the L-type calcium channels and certain calcium-binding proteins was not associated with regions of the brain that are prone to the selective neurodegeneration that is seen in Parkinson’s. But the investigators observed a very different picture in the Parkinsonian brains. Increased levels of L-type calcium channels and the calcium-binding proteins were found throughout the brains of people who passed away with Parkinson’s. Even in cases of early Parkinson’s (in people who passed away shortly after being diagnosed), increased levels of L-type calcium channels were even found in the cerebral cortex – a part of the brain largely unaffected in Parkinson’s.
These findings lead the researchers to conclude that “disturbed calcium homeostasis” may be “an early feature of Parkinson’s disease and not just a compensatory consequence to the neurodegenerative process”.
|
RECAP #2: Accumulating evidence supports the idea that calcium is potentially playing a role in Parkinson’s. Nation-wide, longitudinal data suggests that use of certain calcium channel blocking medication may reduce the risk of developing Parkinson’s. In addition, calcium channel blockers (like isradipine) reduce toxicity in models of Parkinson’s.
|
But how could calcium possibly be involved with the mechanisms underlying the neurodegeneration seen in Parkinson’s?
There are actually numerous ways that calcium could be playing a role – from mitochondrial function to oxidative stress (Click here and here to read good reviews on this topic).
There have also been a number of research reports demonstrating interactions between calcium and the bad boy of Parkinson’s research: a protein called alpha synuclein.
Such as this one here:
 Title: C-terminal calcium binding of α-synuclein modulates synaptic vesicle interaction.
Title: C-terminal calcium binding of α-synuclein modulates synaptic vesicle interaction.
Authors: Lautenschläger J, Stephens AD, Fusco G, Ströhl F, Curry N, Zacharopoulou M, Michel CH, Laine R, Nespovitaya N, Fantham M, Pinotsi D, Zago W, Fraser P, Tandon A, St George-Hyslop P, Rees E, Phillips JJ, De Simone A, Kaminski CF, Schierle GSK.
Journal: Nat Commun. 2018 Feb 19;9(1):712.
PMID: 29459792 (This article is OPEN ACCESS if you would like to read it)
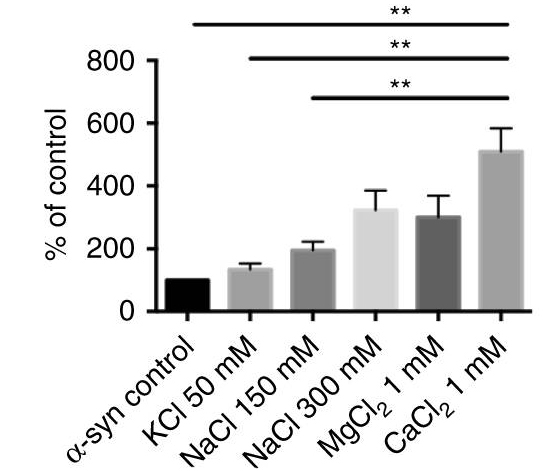
In this study, the researchers wanted to look at how calcium could be affecting the normal functioning of alpha synuclein.
Using a special technique (called ‘Chemical exchange saturation transfer-nuclear magnetic resonance‘ – please don’t ask me what that means), the researchers were able to watch what happens to the protein and calcium when they encounter each other.
When the researchers compared the binding of different ions to alpha synuclein, they found that calcium in particular had a strong affinity for alpha synuclein:
 Source: PMC
Source: PMC
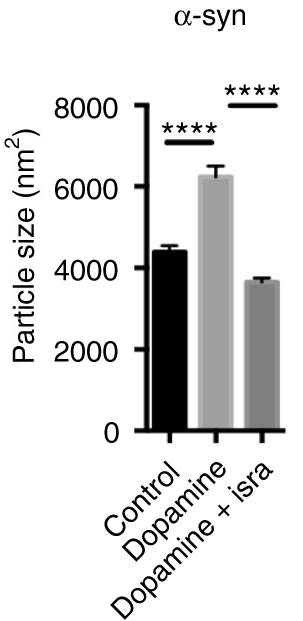
Given that their results, the researchers next asked whether this process could be involved with aspects of Parkinson’s. The investigators found that the combination of calcium and dopamine promoted the clustering (or aggregation) of alpha synuclein. Interestingly, the researchers found that these dopamine-induced changes could be reversed back to normal by treating the cells with the calcium channel blocker, isradipine (remember this drug!).
 Source: PMC
Source: PMC
The researchers also noticed that the addition of dopamine to the combination of calcium and alpha synuclein appeared to cause some toxicity effect resulting in cell death in the experiment. So they next looked at whether the removal of calcium (using the calcium channel blocker isradipine) could reduce this cell loss.
And they found that it did:
 Source: PMC
Source: PMC
There is a lot of additional independent data demonstrating that levels of calcium within a cell (in addition to oxidative stress) can promote alpha synuclein protein aggregation (Click here to read a review on this topic).
Thus, there has been ample evidence to justify a clinical trial testing if reducing calcium levels inside cells can help to slow the progression of Parkinson’s.
|
RECAP #3: Calcium interacts with Parkinson’s-associated alpha synuclein protein. Increasing levels of calcium within a cell results in more alpha synuclein aggregation. Calcium channel blocker medication, like isradipine, can reduce this aggregation effect in models of Parkinson’s. Given all of this combined evidence – nation-wide, longitudinal data as well as efficacy in models of Parkinson’s – clinical evaluation of isradipine seemed justified.
|
Interesting. Are there any clinical trials targeting calcium?
Yes, there are.
And that drug I mentioned in the previous study – isradipine – it has been tested in a series of clinical trials, called STEADY-PD (Safety, Tolerability and Efficacy Assessment of Dynacirc CR in Parkinson Disease). DynaCirc® is the brand name for isradipine.
 Isradipine. Source: Dailymed
Isradipine. Source: Dailymed
Isradipine is a calcium channel blocker of the dihydropyridine class. It is prescribed for the treatment of high blood pressure in order to reduce the risk of stroke and heart attack. Prof Frank Church over at the ‘Journey with Parkinson’s’ blog has a great page on isradipine (Click here to read that post).
The STEADY-PD program started with a pilot study, the results of which were published in 2010:
 Title: Tolerability of isradipine in early Parkinson’s disease: a pilot dose escalation study.
Title: Tolerability of isradipine in early Parkinson’s disease: a pilot dose escalation study.
Authors: Simuni T, Borushko E, Avram MJ, Miskevics S, Martel A, Zadikoff C, Videnovic A, Weaver FM, Williams K, Surmeier DJ.
Journal: Mov Disord. 2010 Dec 15;25(16):2863-6.
PMID: 20818667
In this study, the researchers wanted to assess the tolerability of isradipine in 31 individuals with recently diagnosed Parkinson’s (not requiring any dopaminergic therapy). The investigators gave participants increasing doses of the drug (from 5 to 20 mg per day) over the course of 8 weeks. The vast majority had no issues with the low dose, but almost half had issues tolerating the highest dose.
This study gave the researchers confidence as they planned a larger and longer study to assess the long term safety of isradipine.
The results of that larger study were published in 2013:
 Title: Phase II safety, tolerability, and dose selection study of isradipine as a potential disease-modifying intervention in early Parkinson’s disease (STEADY-PD).
Title: Phase II safety, tolerability, and dose selection study of isradipine as a potential disease-modifying intervention in early Parkinson’s disease (STEADY-PD).
Authors: Parkinson Study Group.
Journal: Mov Disord. 2013 Nov;28(13):1823-31.
PMID: 24123224
In this Phase 2, randomised, double-blind, parallel group study, the researcher enrolled 99 subjects to take 3 different doses (5, 10, or 20 mg) of isradipine or a matching placebo everyday for 52 weeks. The goal of the study was to assess safety and tolerability over a long period of time, and identify the best dose for a larger Phase 3 trial.
The researchers found that the tolerability of isradipine was dose dependent (this means that as the dose went up, the treatment was less well tolerated. In the placebo group, 25 of the 26 participants (96%) found the treatment tolerable, and this was comparable to the 5 mg group, where 19 of 23 (83%) participants considered the tolerable. But in the 10 mg group, 19 of 26 participants (73%) thought that the treatment was easily tolerated, and this dropped significantly in the 20 mg group, where only 9 of 24 participants (37%) found the treatment tolerable.
The investigators observed no difference in change of clinical scores (as determined by the Unified Parkinson’s Disease Rating Scale or UPDRS) across any of the groups, but the study helped to identify the maximal tolerable dose of 10 mg daily for the follow up Phase 3 trial.
So what happened with the Phase 3 study?
The double blind, Phase III STEADY-PD clinical trial was conducted at 56 research centres across the US & Canada, enrolling over 300 people with newly diagnosed Parkinson’s to take either isradipine (5 mg of immediate-release isradipine twice daily) or placebo for 36 months (participants were randomly assigned to one of the two groups – click here and here to read more about the clinical study).
Recruitment started in November 2014 and the last participant completed the 3-year treatment regime in November 2018.
On the 30th March 2019, the investigators coordinating the trial issued a press release that informed the PD community that the STEADY-PD Phase 3 trial had not met its primary endpoint – the endpoint being the predetermined measure that would determine efficacy. There was no difference between in clinical progression of Parkinson’s between the group treated with isradipine or the placebo group (click here to read the press release).
The results of the study were published this week:
 Title: Isradipine Versus Placebo in Early Parkinson Disease: A Randomized Trial.
Title: Isradipine Versus Placebo in Early Parkinson Disease: A Randomized Trial.
Authors: Parkinson Study Group STEADY-PD III Investigators.
Journal: Ann Intern Med. 2020 Mar 31. doi: 10.7326/M19-2534. [Epub ahead of print]
PMID: 32227247
The results of the study suggest that the two groups of participants were evenly randomised (170 in the isradipine group vs 166 in the placebo group). In addition, the average ages were very similar (62.1 vs 61.6, respectively), and the stage of Parkinson’s at baseline were also alike (UPDRS scores of 23.7 vs 22.6, respectively). The two groups appear to have been well balanced.
At the end of the study though, there was no difference between the two groups.
The report shows very similar rates of progression in both the isradipine and placebo groups. Deeper analyses apparently found no differences in outcome based on age, sex, race/ethnicity, or baseline Parkinson’s severity. In addition, there was no difference in the use of anti-Parkinson’s medication use between the groups.
In their concluding statement, the researchers questioned whether the dose used in the study was high enough to have a large enough effect on calcium levels. But they acknowledge that this was the prefered tolerable dose (based on the Phase 2 study). They also wondered if post diagnosis is the wrong time to be utilising this approach. Given that the epidemiological studies (the nationwide, longitudinal research discussed above) shows a reduced risk of developing Parkinson’s, perhaps isradipine would be better administered to individuals at risk of developing Parkinson’s (for example, those exhibiting symptoms of the prodromal phase of Parkinson’s).
What is the prodromal phase?
The ‘prodromal phase’ of the condition is the period of time before diagnosis. It involves some of the very early signs of the Parkinson’s (such as constipation, loss of a sense of smell, and REM sleep disorder – more on this in a moment), and the very first signs of motor/movement problems (a twitching finger, or a dragging leg).
 Source: Guidelinesinpractice
Source: Guidelinesinpractice
The investigators concluded their report by saying “that treatment with 5 mg of immediate release isradipine twice daily did not slow the clinical progression of early-stage PD, as measured by the change in the UPDRS score and various other measures and instruments”.
|
RECAP #3: Despite supportive preclinical evidence, treatment with the calcium channel blocker isradipine resulted in no difference in the clinical progression of Parkinson’s (when compared with a placebo treatment) over 3 years of assessment in a large Phase 3 clinical trial. The investigators question (based on the epidemiological evidence) if calcium channel blockers could be tested on individuals exhibiting prodromal symptoms of Parkinson’s.
|
So is that the end of calcium channel blockers for Parkinson’s?
It could be easy to come to that conclusion, but this is lazy thinking.
As the investigators discussed in the conclusion of their report, there are a lot of factors that could have played a role in the STEADY-PD study not meeting its primary endpoint. It will be necessary for researchers/statisticians to do a deep dive into the data and see what kind of new ideas post-hoc analysis can generate.
In addition, it would be interesting to follow up the participants of the STEADY-PD study to see if this treatment has any impact on the onset of dyskinesias.
What are dyskinesias?
Dyskinesias (from Greek: dys – abnormal; and kinēsis – motion, movement) are a category of movement disorders that are characterised by involuntary muscle movements. And they are certainly not specific to Parkinson’s.
But in the case of Parkinson’s, dyskinesias have generally been believed to be associated with long-term use of Levodopa (also known as Sinemet or Madopar).

Sinemet is Levodopa. Source: Drugs
NOTE: Long-term use of Levodopa is not a certainty for developing dyskinesias, but there is an association. It will differ from person to person.
We have previously discussed dyskinesias on the SoPD (Click here to read more about this topic).
Ok. But why would it be interesting to follow up the STEADY-PD cohort regarding dyskinesias?
Because it has previously been demonstrated that calcium channel blockers – such as isradipine – can also reduce dyskinesias in models of Parkinson’s (for example click here and here to read more about this).
And there is also this report from last year:
 Title: Genetic silencing of striatal CaV1.3 prevents and ameliorates levodopa dyskinesia
Title: Genetic silencing of striatal CaV1.3 prevents and ameliorates levodopa dyskinesia
Authors: Steece-Collier K, Stancati JA, Collier NJ, Sandoval IM, Mercado NM, Sortwell CE, Collier TJ, Manfredsson FP.
Journal: Mov Disord. 2019 Apr 19. [Epub ahead of print]
PMID: 31002755 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in using gene therapy to prevent the development of Levodopa-induced dyskinesias and reduce already established dyskinesias.
What is gene therapy?
Gene therapy is an experimental treatment approach that involves treating medical conditions with DNA rather than drugs.
Gene therapy basically involves introducing a new piece of DNA or replacing a faulty piece of DNA within a population of cells. DNA, as you may remember from high school science class provides the instructions for making proteins in the a cell and these proteins are the bits that actually do stuff.
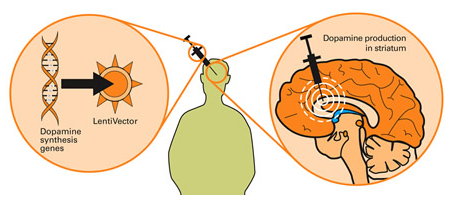
Gene therapy for Parkinson’s disease. Source: Wiki.Epfl
The researchers started their experiment by injecting rats with the gene therapy that would reduce levels of the calcium channel CaV1.3. The virus was injected into a region of the brain called the striatum, which is where dopamine neurons release much of their dopamine. Next they induced a model of Parkinson’s using a neurotoxin (6-OHDA) and then they started treating the animals with high doses of Levodopa to cause Levodopa induced dyskinesias (see panel A in the image below for a schematic of this study design).
The researchers found that rats who were injected the CaV1.3 gene therapy did not develop dyskinensias… at all!
If you look at panel B in the image below, you can see a black line of the control rats (no gene therapy, but model of Parkinson’s with Levodopa induced dyskinesias) rapidly develop the dyskinesias with increasing severity as the dose of Levodopa is increased. The red line bouncing along the bottom of the graph are the gene therapy treated animals with no dyskinesias.
 Source: Wiley
Source: Wiley
This was a really impressive result.
But the researchers wanted to take this finding one step further and see if they can’t stop established dyskinesias – that is to say, can the gene therapy approach work after dyskinesias have already developed?
So the investigators repeated the experiment, except this time they induced a model of Parkinson’s using a neurotoxin (6-OHDA) and treating the animals with high doses of Levodopa to cause Levodopa induced dyskinesias before they treated some of the animals with the gene therapy treatment (see panel A in the image below for a schematic of this study design).
As you can see from panel B of the image below, the red line of the gene therapy treated animals starts to reduce from about 2 weeks post-gene therapy treatment. The gene therapy treament resulted in a significant reversal of established, severe Levodopa induced dyskinesias.
 Source: Wiley
Source: Wiley
And remarkably, the researchers demonstrated that this CaV1.3 reducing gene therapy did not interfere with the normal motor response to Levodopa. That is to say, Levodopa retained its ability to improve motor function in the rodent models of Parkinson’s despite the gene therapy treatment.
If this experimental gene therapy treatment can be translated into humans, it could represent an impressive new method of treating Levodopa-induced dyskinesias.
But my point regarding the follow up of the STEADY-PD isradipine-treated cohort is that it would be interesting to see if they (as a group) have a delayed onset of dyskinesias.
So what does it all mean?
The results of a major Phase 3 clinical trial for disease modification in Parkinson’s have recently been published. Unfortunately, despite a strong preclinical case for support, the calcium channel blocker isradipine was found to have no effect (compared to placebo) on the course of Parkinson’s over a 3 year study.
My greatest fear when a disappointing result is published is that the biology behind the trial gets tainted and dumped on the waste pile of ‘failed’ ideas. It would be wrong to shut the door on calcium channels and move on. There certainly appears to be something happening between calcium and Parkinson’s, and it would advance the field significantly if we can establish exactly what that is.
The aftermath of a trial should also be a period of reflection and future planning – what was done well, what could be improved. When an airliner has an accident, a small army of individuals examine every aspect of the plane and this results in improvements to the design of the aircraft – the same mentality should be applied to clinical trials (Click here to read a previous SoPD rant on this topic).
In a recent SoPD post, we discussed the importance of following up clinical trial cohorts and how important new piece of data can be collected (even in studies that did not reach their primary endpoint – click here to read that SoPD post). I hope that the isradipine cohort are followed up over time. They (and their families) have dedicated a significant period of time and effort to getting this study finished and if further information can be gleamed from their amazing contribution, that would be a good thing.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from STEADY-PD.

Fascinating and brilliant explanation as always, thank you. I read about the Isradipine study in 2015 when diagnosed with PD and hypertension the same week. Seemed logical to take Isradipine as part of my antihypertensive drug regime and I’ve taken it ever since – I’m keeping the door open on my study of one!
LikeLike