|
There has been a lot of discussion on this site (and elsewhere on the web) regarding the need for more objective systems of measuring Parkinson’s – particularly in the setting of clinical trials. Yes, subjective reports of patient experience are important, but they can easily be biased by ‘placebo responses’. Thus, measures that are beyond the clinical trial participants conscious control – and focused on biological outcomes – are needed. In today’s post, we will consider one possible approach: Smart pills. We will discuss what they are, how they work, and how they could be applied to Parkinson’s research. |
Source: Chicagotribune
In order to encourage a growing discussion regarding objective measures of Parkinson’s (and to follow up on previous rants – Click here and here for examples), I have decided to regularly (once a month) highlight new technologies that could provide the sort of unbiased methods of data collection that are required for assessing whether a treatment is having an impact on Parkinson’s.
Today, we will look at smart pills.
What is a smart pill?
Smart pills (or digital pills) are being viewed as an exciting new area of future medicine. They are pills that can be consumed and then they monitor aspects of the gastrointestinal tract and report that information back to the outside world, providing data that can be analysed and assessed over time.
Wow. Cool. How does this technology work?
There a numerous companies developing smart pill technology, and the pills differ from company to company. One of the products that has advanced the most in clinical availability is from a company called Proteus Digital Health.
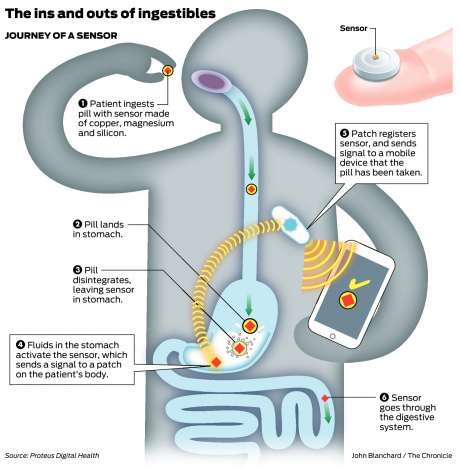
They have engineered a sensor that people can consume with their usual medication. The 1mm sensor – which can be embedded in pills – provides a method of insuring that individuals are adhering to their treatment regime – that is to say, they have taken their medication as prescribed (approximately 50% of people fail to take medication as prescribed – Source).
The Proteus pill with sensor visible in the centre. Source: Medgadget
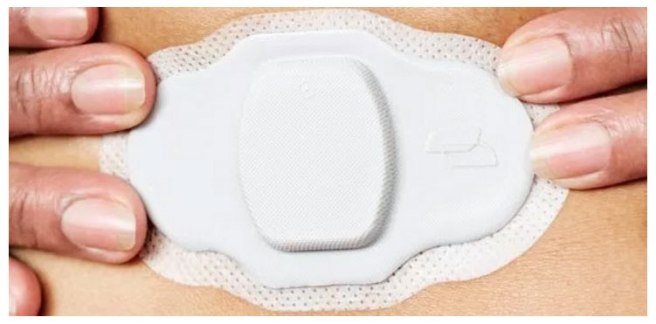
The sensor is made up of tiny silicon sensors covered in magnesium and copper. The magnesium and copper are important, because when the pill that the sensor is attached to reaches a person’s stomach, these two elements can generate enough power – through chemically reacting with the stomach acid – to power the sensor and allow it to communicate a unique time-stamped signal to a patch that is attached to the subject’s torso. And the patch only needs to be replaced every seven days.
Proteus patch. Source: Healthtechinsider
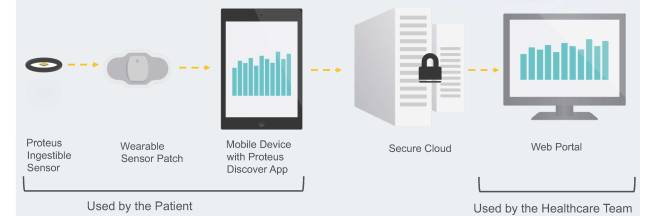
The wearable patch then transmits the data to a smart phone app. And the patch continuously collects additional information – such as steps, activity, rest and heart rate – which can also be viewed via the mobile app. If the app does not receive a signal indicating the person has taken their medication at the prescribed time, it can alert the person with a reminder on the smart phone.
Source: Proteus
Thus, the Proteus smart pill system work something like this:
Source: sfchronicle
In addition to the smartphone app that participants can view, physicians and caregivers can also see the information being collected by the patch using a web-based portal. ‘Big brother’ you say? Yes, there are concerns about privacy surrounding the smart pill approach (Click here to read more about that), but the ends hopefully outweigh the means in this situation.
Smart pill data sharing. Source: jmir
A great deal of information can be collected by this Proteus system, and the good news is that in November 2017, the system was approved by the FDA for clinical use in the Aripiprazole pills (under the label “Abilify MyCite” – Click here to read more about this). Aripiprazole is a drug that treats schizophrenia and bipolar disorder.
If you haven’t understood anything I have written, this video – a CBC news article about the FDA approval – explains the system very well:
That’s really amazing. So how could we use it for Parkinson’s?
Before we move on to that, we should have a look at the smart pills being developed by other companies, in particular Medtronic.
Medtronic has also been developing a smart pill which – while not yet approved by the FDA for clinical use – will be able to do a lot more than the current Proteus smart pill.
Medtronic’s product is called the SmartPill™ motility testing system:
Medtronic SmartPill. Source: Emilysstomach
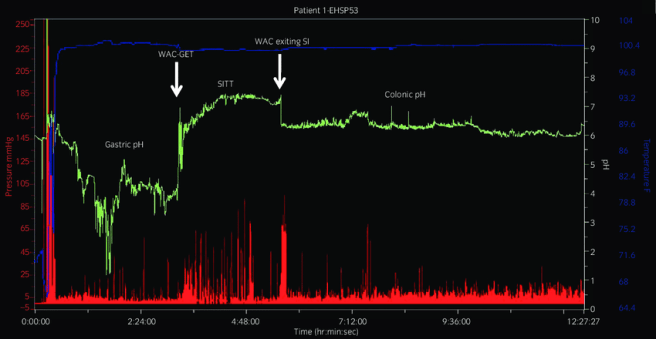
As the SmartPill™ motility capsule travels through a person’s gastrointestinal tract, it measures pressure, acidity and temperature – providing valuable information, in addition to total gastrointestinal transit time.
An example of some of the information collected. Source: Researchgate
A lot of information could be collected with this Medtronic smart pill system, and compared with other participants in a clinical study. My only concern is that the pill is currently quite large:
A tough pill to swallow? Source: Emilysstomach
Dysphasia (trouble swallowing) is a common feature of Parkinson’s, so Medtronic may need to shrink this particular pill down a little in order for it to be practical for Parkinson’s.
Ok, all of this is interesting, but how can a smart pill that monitors the activity of the gut help us with assessing a neurodegenerative condition like Parkinson’s?
A desirable requirement of any objective measure would be its specificity.
Ideally, each measure will assess a very particular function of a specific organ without interfering with that function (or any other functions). These pills would provide a very useful objective assessment of adherence to drug treatment regime (the Proteus system) and measures of gut function (the Medtronic system).
Yeah, but the gut?!?
Beyond any theories that the gut may be influential in the development or course of the condition (Click here to read previous posts about this), the gastrointestinal tract is affected by Parkinson’s and this presents itself in several ways. Constipation, in particular, is a common issue in Parkinson’s and using a smart pill – which can provide a quantitative measure of gastrointestinal transit – could provide a useful method of assessing whether a drug is improving gut function in Parkinson’s.
And there are ongoing clinical trials for drugs targeting constipation in Parkinson’s.
For example, this week a biotech company called Enterin Inc. announced that they have completed enrolled for their RASMET study: a Phase 1/2a randomised, controlled, multi-center clinical trial evaluating a synthetic version of squalamine (called ENT-01) in people with Parkinson’s (Click here to read the press release).
Source: Onemednews
We have previously discussed squalamine/ENT-01 (Click here to read that post). Squalamine is an interesting compound that was originally extracted from dogfish sharks.
Dogfish shark. Source: Discovery
Squalamine is a steroid with a wide range of antimicrobial activity. Steroids are used as a treatment for certain inflammatory conditions, but the recent research suggests that Squalamine can also block the Parkinson’s associated protein alpha synuclein from clustering (or aggregating).
This property of Squalamine may sound great for Parkinson’s, as the aggregation of alpha synuclein is widely considered to be one of the major influencers involved in the development of Parkinson’s. But there is just one small problem:
Squalamine doesn’t cross the blood-brain barrier
(click here to read more on this)
The blood brain barrier is a protective membrane surrounding the brain, which prevents certain chemicals (Squalamine among them) from entering the environment of the brain. This issue of brain penetrance has not stopped Enterin, however, who are seeking to clinically test ENT-01 for the treatment of Parkinson’s related constipation.
The study will involve 50 patients over a 9-to-12-month period. The trial is being conducted in two stages: Phase 1 involved 10 participants and it sought to assess the safety, tolerability, and pharmacokinetics of single escalating doses of ENT-01 over a 30-60 day period. This dose-escalation study was preceded by a 2-week run in period of assessment and followed by a 2-week wash-out period with further assessments. This study has now been completed, though the results have not been released. Given that the company has announced that they have completed the recruitment for the Phase 2a part of the trial, we can make the assumption that the safety profile of the drug is acceptable.
The RASMET study is now moving on to the Phase 2a trial, which will enrol 40 people with Parkinson’s. This study will be composed of 4 periods:
- A 2-week run-in period
- A 3-5 week escalating dose period to identify a prokinetic dose in the initial set of 10 patients
- A 1-week period of randomised dosing (placebo versus the previously identified pro-kinetic dose)
- A 2-week wash-out period.
What is a prokinetic dose?
A prokinetic (also known as a gastroprokinetic or gastrokinetic agent) is a class of drug which enhances gastrointestinal motility by increasing the frequency (or strength) of contractions in the small intestine, but without actually disrupting their rhythm.
In the RASMET study, pharmacodynamics (or the effect of the drug on the body) will be assessed along with safety and tolerability. Relative outcomes will be compared within each patient and across groups. Frequency of bowel movements and other non-motor symptoms of Parkinson’s will be collected over the course of both phases.
The primary outcome measure of the study will be the number of participants with treatment-related adverse events. This is a measure of the safety of the drug.
The secondary outcome measure will be the frequency of bowel movements. I am not sure how this is being measured, whether it is just patient reports/diary or a more sophisticated method, but obviously this is where the smart pill approach could be useful – 1.) providing an objective measure of adherence to drug treatment regime (using the Proteus system) and measures of gut function/gastrointestinal transit time (the Medtronic system).
What does it all mean?
In an effort to encourage the use of more objective methods of assessment in clinical trials, we will start an effort here on the SoPD to try and discuss one method per month.
In today’s post, we have looked at the concept of smart/digital pills. Tiny electronic devices that can monitor various aspects of the gastrointestinal tract. Such tools could be very useful in the setting of a clinical trial focused on issues like constipation – a common problem in Parkinson’s. If a drug is being tested for this complaint, smart pills could provide objective methods of acquiring very accurate and unbiased data on the efficacy of the treatment.
It will be interesting to see how this technology develops in the near future.
EDITOR’S NOTE: Proteus Digital Health is a privately owned company and Medtronic is a publicly traded company. Neither company has requested this material to be produced, nor has the author had any contact with these companies or any associated parties. The material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. This post has been produced for educational purposes only and should not be considered an endorsement of the companies or their products. Information regarding the technology is merely being shared here as an example of what is possible regarding the objective measurement of chronic medical conditions.
The banner for today’s post was sourced from Consumerreports










4 thoughts on “Objective measures: Getting smart about pills”