|
The new year has started with some pleasing clinical trial news for the Parkinson’s community: The results of the “Ambroxol in Disease Modification in Parkinson Disease” (AiM-PD study) have been published. This is a clinically available drug that is used for the treatment of respiratory issues, which researchers are re-purposing for Parkinson’s based on some interesting properties the drug has. The results of the clinical trial suggest that ambroxol was safe and well tolerated in people with Parkinson’s for the length of the 6 month study. It accessed the brain and increased levels of target proteins while there. In today’s post, we will discuss what ambroxol is, what research has been conducted on it, and what the results of this study suggest.
|
The author of this blog is the deputy director of research at The Cure Parkinson’s Trust, and as such he feels that it is necessary to start this post with a very clear declaration – FULL DISCLOSURE: The Cure Parkinson’s Trust (in partnership with the Van Andel Institute) was a funder of the ambroxol clinical trial which is going to be discussed in this post.
Right. That said, let’s try and do a completely unbiased review of the ambroxol trial results 🙂
In one particular SoPD post last year we discussed the Linked Clinical Trials initiative, which is an international program that was set up 8 years ago with the goal of rapidly repurposing clinically available drugs exhibiting disease modifying potential in models of Parkinson’s (Click here to read the previous SoPD post on this topic).
What is meant by repurposing?
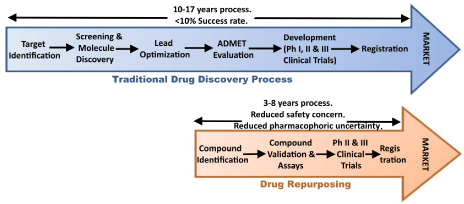
Drug repurposing (repositioning, reprofiling or re-tasking) is a strategy of identifying novel uses for clinically approved drugs that fall outside the scope of the original medical indication.
An example of this is “Viagra”.
It was originally developed as an anti-hypertensive medication, but was hugely more successful in the treatment of erectile dysfunction.
The strategy has been adopted and applied by many organisations because it allows for the by-passing of large parts of the drug discovery process, saving time and resources in getting new treatments to the clinic.
 Source: Austinpublishinggroup
Source: Austinpublishinggroup
By repurposing a clinically approved drug – for which we may know a great deal about already in terms of safety, tolerability and dose range – we can skip large parts of the clinical trial process and jump straight to testing the drug in our population of interest (in this case people with Parkinson’s).
And this is what the Linked Clinical Trials (or LCT) program was set up to do in Parkinson’s.
The first drug that was prioritised by the LCT committee for repurposing was a diabetes drug called exenatide (also known as Bydureon).
It is fair to say this LCT-initiated clinical trial program has provided interesting results thus far (Click here and here to read a SoPD post on this) and the exenatide program is now entering Phase III testing in Parkinson’s (Click here to read more about the Phase III trial).
In late 2014, the LCT committee prioritised another clinically available drug for repurposing to Parkinson’s.
That drug is called ambroxol.
What is ambroxol?