|
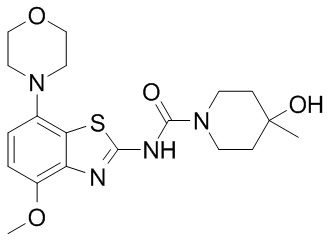
This week Denali Therapeutics released the results of a phase I clinical trial of their primary product, called DNL-201. DNL-201 is a LRRK2 inhibitor that the company is attempting to take to the clinic for Parkinson’s disease. In today’s post we will look at what LRRK2 is, how an inhibitor might help in Parkinson’s, and what the results of the trial actually mean. |

Denali. Source: Wikipedia
Denali (Koyukon for “the high one”; also known as Mount McKinley) in Alaska is the highest mountain peak in North America, with a summit elevation of 20,310 feet (6,190 m) above sea level. The first verified ascent to Denali’s summit occurred on June 7, 1913, by four climbers Hudson Stuck, Harry Karstens, Walter Harper, and Robert Tatum.
Tatum (left), Karstens (middle), and Harper (right). Source: Gutenberg
Robert Tatum later commented, “The view from the top of Mount McKinley is like looking out the windows of Heaven!”
More recently another adventurous group associated with ‘Denali’ have been trying to scale lofty heights, but of a completely different sort from the mountaineering kind.
Continue reading “Inhibiting LRRK2: The Denali Phase I results”