|
Each time a cell divides, the DNA inside the resulting pair of cells has changed slightly. These small alterations – known as genetic mutations – provide a method by which an organism can randomly determine traits that may be beneficial. New research indicates that in certain parts of the brain, post-mitotic (non-dividing) cells are taking on as many as one mutation per week across the span of our lives. This results in thousands of genetic variations accumulating in each cell by the time we eventually pass away in old age. In today’s post we will review new research and consider what this gradual build up of genetic mutations could mean for our understanding of neurodegenerative conditions, like Parkinson’s. |
Source: Pexels
Coming from the back waters of third world New Zealand, you will understand that sheep hold a very special place in my heart.
I grew up a simple country lad, and each year I had a pet lamb that I would raise and train to do silly tricks in the hope of impressing the judges at the annual agricultural/farm day at school. In addition to instilling me with a crazy fanaticism for the sport (read: religion) of rugby, my parents figured that having a pet lamb each year would teach me a sense of responsibility and a sort of discipline.
I’m not really sure how this practice has influenced my later life, but I certainly do have very fond memories of those early years (the first lamb was named ‘Woolly’, the 2nd lamb was named ‘Woolly2’, the third lamb was actually a goat – bad lambing season – which I named ‘Billy the kid’, the 4th lamb was named ‘MacGyver’,…).
Lots of happy memories.
Source: Countryliving
But as I grew into the teenage years, there was one thing that really bothered me with regards to my pet lambs.
It was that whole negative stigma associated with the ‘black sheep’.
Why, I would wonder, was it the ‘black sheep of the family’ that was the bad kid? And why was the one black sheep in every flock considered the worst of the bunch?
Source: theodysseyonline
Why was this association applied to sheep?
Why not dogs? Or cows? Why do we pick on sheep?
Ok, to be fair, black cats get a bad rap and black swans are a thing of intrigue. But the question still stuck with me: why are black sheep stigmatised.
Curiosity has led me to look into this, but the explanations are unfortunately limited.
The association may have started with Myles Coverdale‘s 1535 Bible, which was the first complete bible printed in English. It was translated from German texts and rendered Genesis 30:32 as:
‘All blacke shepe amonge the lambes’
In the original German texts, Genesis 30:32 deals with the shepherd Jacob who remove any sheep or goats with a blemish (spotted, dark, etc) from his master’s flock, in order to be able to later show that he had not stolen any of the white ones. Other bibles translated the original German text have Genesis 30:32 as ‘every speckled and spotted sheep and every black lamb’ or ‘all the browne cattell among the sheepe’. They did not pick solely on the poor black sheep.
In fact, in the original German text ‘black sheep’ were actually held in esteem rather than disrepute.
Source: Fortune
But the mix-up in an English translation does not account for the presence of the idiom being found in other languages (such as German, Hebrew, etc).
In 18th and 19th century England, the black sheep was seen as the mark of the devil. Why this is, is the stuff of debate. Supposedly a real black sheep had wool that could not be dyed and thus was worth less.
Having said that, one black sheep in a flock was actually considered good luck by shepherds in the counties of Sussex, Somerset and Kent, Derbyshire.
While I appreciate and respect your kiwi affections for sheep, what does any of this black sheep stuff have to do with Parkinson’s?
In biology, genetic mutations carry the same sort of sigma as the ‘black sheep’. If you have a mutation, it is considered a bad or negative thing – they are perceived as the cause of all problems. And yet not all genetic mutations are bad, just as not all black sheep are bad.
What are genetic mutations?
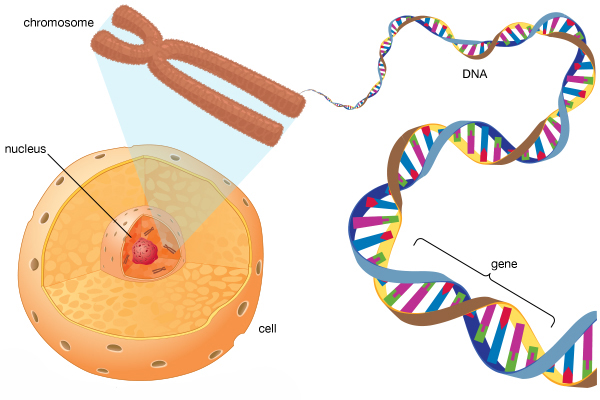
Let’s start at the beginning: In almost every cell of your body, there is a nucleus.
It is the command centre for the cell – issuing orders and receiving information concerning everything going on inside and around the cell. The nucleus is also a storage bank for the genetic blueprint that provides most of the instructions for making a physical copy of you. Those grand plans are kept bundled up in 23 pairs of chromosomes, which are densely coiled strings of a molecule called Deoxyribonucleic acid (or DNA).
DNA’s place inside the cell. Source: Kids.Britannica
DNA is the ‘double helix’ stuff that biologists get excited about (Click here for the National Institute of Health’s website explanation of DNA). The information about you is stored in DNA using a code, made up of four chemical bases: adenine(A), cytosine (C), guanine (G), and thymine (T). These As, Cs, Gs and Ts are paired up and strung together in a huge number of pairings – approximately 3 billion pairs in a single cell in fact.
And when I say ‘pairs’, I mean that these letters pair off — A with T and C with G . An easy way to remember these pairings is Apples are in the Tree and Cars are in Garages:
Source: Pinterest
These pairings are called base pairs (the A, C, G & T being referred to as bases). Each base is also attached to a sugar molecule and a phosphate molecule. Together, these three components are called a nucleotide.
The AGTCs of DNA. Source: Knowgenetics
Understand that this simple pairing hold us together by holding our DNA together. They are critical to the normal biological functions of life – without them, there is no ‘us’.
Certain long segments of these pairings in the DNA provide the instructions for making a protein. These regions are called ‘Protein coding’ regions. These segments give rise to a molecule called Ribonucleic acid (or RNA) through a process called transcription. RNA is the intermediate that is used in the production of a protein – a process called translation.
Curiously, large portions of DNA is made up of regions that do not give rise to RNA, while other regions give rise to RNA, but no protein is made using that RNA. These region are called ‘Non-coding’ regions (the RNA that is produced has other functions in those situations – Click here to read more about this).
‘Protein coding’ or ‘Non-coding’. Source: Nature
Whether an RNA-producing region of DNA is a ‘Protein coding’ or ‘Non-coding’, these regions are all collectively referred to as genes. And it’s each of these genes that provide the instructions for keeping you alive.
BUT (and it’s a big BUT), with 3 billion different base pairs, you will understand that occasionally things may go a little awry. An individual pairing may have an error or a small section of DNA may be deleted, especially during periods when the DNA is being replicated (such as when cells divide).
These errors are referred to as genetic mutations or genetic variants.
And these genetic variations come in a variety of different flavours: some can simply be a single mismatched base pair (also called a point-mutation or single nucleotide variant), while others are more complicated such as:

Lots of different types of genetic variations. Source: Nature
Ok, but these mutations are bad right?
Actually, no. In the vast majority of cases these mutation do not have any effect (we’ll come back to this later). In some situations the effects are rather mild (such as genetic variants of the melanocortin-1 receptor (MC1R) gene give us red hair).
And in other cases, they may actually be beneficial (spontaneous errors are one of the foundations in the process of evolution). A random mutation may make someone immune to certain conditions (for example, the CCR5-delta 32 genetic mutation hampers the ability of the virus that causes HIV (AIDS) to infiltrate immune cells, thus making carriers immune – click here to read more about this).
But some times these genetic variants can cause trouble.
Big trouble (think: Cancer).
And researchers have been interested in looking at how these mutations can collect over a life-time and impact on our well being. Which brings us to the research we are reviewing today. Recently researchers at Harvard University in Boston (USA) have had a look at the human brain to determine the frequency of genetic variations that collect during our lives:
Title: Aging and neurodegeneration are associated with increased mutations in single human neurons.
Authors: Lodato MA, Rodin RE, Bohrson CL, Coulter ME, Barton AR, Kwon M, Sherman MA, Vitzthum CM, Luquette LJ, Yandava C, Yang P, Chittenden TW, Hatem NE, Ryu SC, Woodworth MB, Park PJ, Walsh CA.
Journal: Science. 201 8, 359, 555-559
PMID: 29217584
In this study, the researchers used single-cell, whole-genome sequencing to look for somatic single-nucleotide variants in the DNA from 161 single neurons that were collected from two regions of the brain (the prefrontal cortex and hippocampus) from 15 normal individuals (aged 4 months to 82 years), as well as 9 individuals affected by genetic conditions that result in early-onset neurodegenerative conditions (Cockayne syndrome and xeroderma pigmentosum).
Ok, what does any of that actually mean? You’re going to have to explain some of it more clearly. What is ‘whole-genome sequencing’?
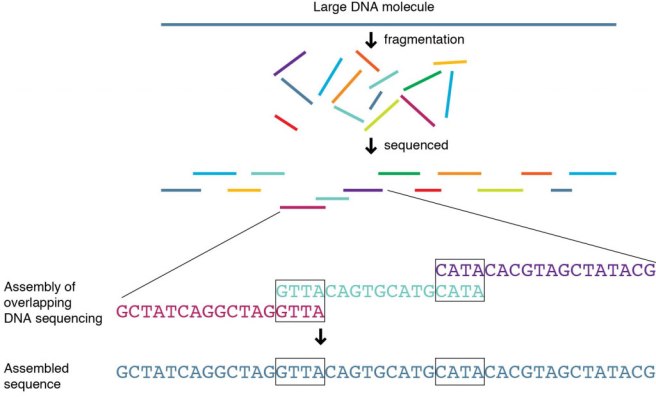
Whole genome sequencing is simply a method of determining of the actual start-to-finish sequence of As, Gs, Ts, & Cs in your DNA. Your ‘genome’ includes all three billion plus base pairs of your DNA. Samples of your DNA (extracted from blood or saliva) are chopped up and put into a machine that can identify each base pair in a section of DNA. Once the sequence of each section is determined, the sequences of many sections are pooled together, allowing for overlapping sections to be combined – ultimately resulting in a complete readout of your DNA.
Source: knowgenetics
In this study, the researchers conducted whole genome sequencing – the reading of the DNA sequence – on 161 individual cells collected from two different regions of the human brain.
And what are ‘somatic single-nucleotide variants in the DNA’?
Most of the genetic variants that we have which define who we are, we have had probably had since conception. These are called ‘germ line’ mutations. They are inherited from our parents. But we also have other genetic mutations which we pick up during life and that are usually specific to particular cells or tissues in the body, and they are called ‘somatic’ mutations.
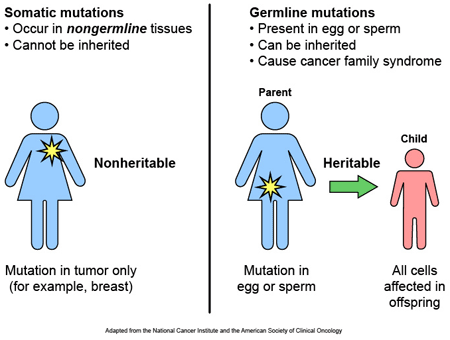
Somatic vs Germline mutations. Source: AutismScienceFoundation
And these somatic variants usually occur at single-nucleotide locations (they are also called point-mutations). A single-nucleotide variant is where an alteration occurs in a single base (or nucleotide). For example: the genetic sequence ATG could become CTG as a single-nucleotide variation occurs with the A becoming a C.
The researchers in this study conducted whole genome sequencing on 161 individual cells and they have been looking for small single-nucleotide somatic mutations. The cells have been collected from two different regions of the brain: the prefrontal cortex and hippocampus.
What are the prefrontal cortex and hippocampus?
The prefrontal cortex sits at the front of your brain (see the red area in the image below). It is where you make all of your ‘executive’ decisions, like “what should I have for dinner tonight?” or “should I go left or right at the next street?” or “should I keep reading this gibberish?”.
It is a region of the brain where the vast bulk of the cells are considered ‘post-mitotic‘ or non-dividing. Mitosis is a process by which cells divide, once cells become post-mitotic they cease to be able to divide anymore. Given that genetic mutations occur a lot during cell division, one would assume that being post-mitotic should reduce the number of variations that can occur over time.
The prefrontal cortex. Source: Neuropsychotherapist
The hippocampus, on the other hand is a region of the adult brain where cell division is still occurring even in late age. It is a banana shaped area, that lies on both sides of our brain. As the image below suggests, the hippocampus (in red) is deep inside the brain. It is believed to be involved with the formation of new memories.
The hippocampus. Source: Psychologytoday
So the researchers collected cells from these brain regions and then sequenced the DNA from each cell? And what did they find?
The researchers collected and analysed 93 neurons from the postmortem prefrontal cortex of 15 normal individuals. These individuals varied in age, from 4 months to 82 years. The investigators also collected and examined 26 neurons from the hippocampus of six of these individuals, since the hippocampus is an area of the brain where cells are still dividing. And for comparative sake, the researchers also collected and analysed 42 prefrontal cortex neurons from 9 individuals who where diagnosed with Cockayne syndrome or xeroderma pigmentosum during their lives. Both of these conditions are inherited disorders associated with DNA repair problems. When DNA is damaged, there are mechanisms within our cells that help to repair the damage. In both Cockayne syndrome and xeroderma pigmentosum, these DNA repair mechanisms do not function correctly. The researchers were interested to see what impact losing the DNA repair mechanism may have on the accumulation of somatic variants over time.
When the researchers looked at the results of the DNA analysis from all of the neurons from healthy subjects, they found that the number of somatic single-nucleotide variants correlated with age – that is to say: as age went up, so too did the number of somatic single-nucleotide variants.
This trend resulted in 1000s of variants per neuron over a human life span.
And this increase varied between the two regions analysed, from approximately 40 somatic single-nucleotide variants per year in the hippocampus (that is almost 1 per week!), to approximately 23 per year in the prefrontal cortex.
When the investigators looked at the cells from the cases of Cockayne syndrome and xeroderma pigmentosum, they found a 2.5-fold increase in the number of somatic single-nucleotide variants compared to the expected age-adjusted normal rate, suggesting that in the absence of DNA repair mechanisms the accumulation of genetic variants is greater.
The researchers concluded that their analysis revealed that somatic SNVs accumulate very slowly but continuously over time in normal human brain (a phenomenon that they have termed genosenium). Already at one year after birth, non-dividing neurons have collected between 300 to 900 somatic SNVs. The researchers suggest that this ‘genosenium’ phenomenon may be important in other human age-associated conditions, such as neurodegenerative conditions.
How would mutations in individual neurons affect a condition like Parkinson’s?
Well, the researchers were indicating that mutations in just one neuron may not have a big impact, but the accumulation of mutation across all neurons may result in a divergence of genomes across the brain which could potentially start to affect function. And the question then becomes: could this be possible in situations where an individual is born with a genetic variant that makes them vulnerable to developing a particular condition, such as Parkinson’s.
This is really bad right?
Well, no, not necessarily.
And here we come back to the poor old black sheep situation.
You see, less than 25% of your DNA is actually functional (Source). That is to say, 75% of the DNA in your cells has no meaningful purpose what-so-ever (as far as we can tell). It may be left-over evolutionary material, but it provides no discernible instructions for making stuff, and thus has no benefit or negative impact.
It does nothing.
Zip. Zero, zilch, nada.
Soooo, most of the somatic variants that we collect over our lives will occur within this region of DNA we politely refer to as ‘junk DNA‘ and this will (as far as we can tell) have no impact on neural function. So if each neuron collects 4000 somatic mutations over our lives, only 1000 will be in any areas that are important.
In addition, there are an estimated 19,000-20,000 protein-coding genes in the human genome, and perhaps another 40, 000 non-coding genes (which result in no protein). Thus, if 1000 genetic variants are spread over 60,000+ genes, one would have to be extremely unlucky for one of those genes to be critical for the normal functioning of a neuron.
As a result of this, most of those 1000 variants will probably have little or no impact on normal functioning, while some may actually have a beneficial effect on something like cell survival (I am speculating wildly here). But then, of course, there will be a small percentage of that could have negative consequences and this may result in dysfunction or the cell actually dying. But this is likely to be a very small number of cells.
So what does it all mean?
Harvard researchers have determined an approximate frequency of somatic single nucleotide variants in individual neurons in the human brain. While the results are very interesting and perhaps larger than expected, it is still a large leap to suggest that this phenomenon could be involved in the development of neurodegenerative conditions (if a mutation occurs in a critical gene in an individual neuron, that cell will die, but this does not mean the person will develop Parkinson’s).
Having said that, it would be interesting to repeat this study on a larger scale and assess what the situation is in the brains of individuals who passed away with neurodegenerative conditions, particularly those with genetic vulnerability for their condition.
Regardless, I hope that this post has given you a fresh perspective on genetic mutations… and black sheep.
The banner for today’s post was sourced from Study










To add to your great sheep picture collection
LikeLike
Thanks DKDC – all these pictures of sheep are making me home sick!
LikeLike