|
A reader recently asked me about an experimental drug called Ibudilast. It is a ‘Phosphodiesterase 4 inhibitor’. Recently there was a very interesting result in a clinical trial looking at Ibudilast in a specific neurodegenerative condition. Sadly for the reader that condition was not Parkinson’s, in fact very little research has been done on Ibudilast in Parkinson’s In today’s post we will look at what Phosphodiesterase inhibitors are, how they work, and discuss why Ibudilast may not be such a good experimental treatment for Parkinson’s. |
On April 21-27th, 2018, the American Academy of Neurology (AAN) will hold their 70th Annual Meeting in Los Angeles (California).
I will not be at the meeting, but I will definitely be keeping an eye out for any news regarding the results of one particular clinical trial. At the meeting, a biopharmaceutical company called MediciNova Inc. will be presenting data regarding one of their clinical trials.
The presentation, entitled “Ibudilast – Phosphodiesterase Type 4 Inhibitor – Bi-Modal Therapy with Riluzole in Early Cohort and Advanced Amyotrophic Lateral Sclerosis (ALS) Patients – Final Report and Future Directions“ (Source) will be presented by principal investigator of the clinical study, Dr. Benjamin Rix Brooks, of the Carolinas HealthCare System’s Neuromuscular/ALS-MDA Center at Carolinas HealthCare System Neurosciences Institute.
Dr Brooks will be presenting the results of a single-center, randomized, double-blind, placebo-controlled clnical trial which was conducted to evaluate the safety, tolerability and clinical endpoint responsiveness of a drug called Ibudilast (or MN-166) in subjects with the neurodegenerative condition, Amyotrophic Lateral Sclerosis (or ALS – also known as motor neuron disease; Click here to read a previous SoPD post about ALS and Click here to learn more about this clinical trial).
What is Ibudilast?
Ibudilast is a phosphodiesterase inhibitor.
What is a phosphodiesterase inhibitor?
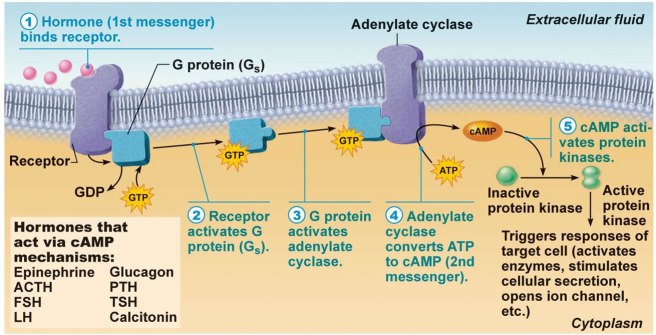
Phosphodiesterase inhibitors – as the label suggests – inhibit Phosphodiesterases.
And Phosphodiesterases are a diverse family of enzymes (there are 11 different types of Phosphodiesterases in humans) that are present inside every cell in your body. They play key roles in regulating levels of cAMP and cGMP.
And yes, I know what you are going to ask next:
Cyclic Adenosine MonoPhosphate (cAMP) and Cyclic Guanosine MonoPhosphate (cGMP) are what we call second messengers within a cell.
We have previously talked about the idea of receptors on the surface of cells (Click here to read a post on this). They function act like a ‘light switch’ – allowing for certain biological processes to be initiated or inhibited. All that a receptor requires to be activated (or blocked) is a chemical messenger – called a ligand – to come along and bind to it.
An example of a receptor on a cell. Source: Droualb
The ligand is considered a first messenger in the process of cellular signalling.
But what happens next?
After the receptor is activated, what actually happens inside the cell?
Well, this is where second messengers, like cAMP and cGMP come into the picture. cAMP and cGMP are the molecules that make stuff happen after a receptor is activated.
Source: Picmia
Second messengers, like cAMP and cGMP stimulate different biological pathways (based on which receptor has been activated). These pathways are control all sorts of cellular processes. For example, cAMP is known to reduce inflammation, reduce blood pressure, and be neuroprotective – in addition to a host of other properties:
 Some of the effects of cAMP activity. Source: Nutritionreview
Some of the effects of cAMP activity. Source: Nutritionreview
So how do Phosphodiesterases regulate levels of cAMP and cGMP?
As I mentioned above these Phosphodiesterase enzymes are involved in controlling levels of cAMP and cGMP. They do this by being involved in the enzymatic breaking down of these messengers. High levels of Phosphodiesterase = lower levels of cAMP and cGMP. And by blocking Phosphodiesterases, there is an increase in levels of cAMP and cGMP.
Thus, if you treat someone with a Phosphodiesterase inhibitor, levels of cAMP and cGMP increase.
Which results in more of that good stuff, such as neuroprotection and reduced inflammation (are you starting to see where this is all heading?).
You may recall that there are 11 different types of Phosphodiesterases. And inhibitors for some of these different types of Phosphodiesterases have been developed.
Ibudilast is primarily considered a Phosphodiesterase type 4 inhibitor.
Is Ibudilast clinically available?
Only in Japan.
Ibudilast has been marketed in Japan since the late 1980’s for the treatment of post-stroke complications and a treatment of asthma. It has exhibited both anti-inflammatory and neuroprotective properties.
Ibudilast. Source: Newdrugapprovals
It is a orally bioavailable treatment, with a very long half-life inside the body (19 hours! – Source), and few side effects (generally Ibudilast has been well tolerated with the most common side effects being cold-like symptoms and headache).
And why is Ibudilast interesting?
In 2016, Ibudilast received approval from the U.S. Food and Drug Administration (FDA) for “fast track” development as a potential treatment for progressive multiple sclerosis (Click here to read the press release). And then in late 2017, the results of a phase IIB clinical trial of progressive multiple sclerosis (called SPRINT-MS – click here to read more about the trial) were reported.
And those results were rather amazing.
Source: Neuronext
Ibudilast almost halved the amount of the brain atrophy observed in the participants receiving the drug. That is to say, the shrinkage of the brain of these participants was significantly reduced as a result of ibudilast. This indicated that ibudilast was slowing down the progression of multiple sclerosis.
Amazing result, right?
The study involved 255 people with multiple sclerosis who were randomly and double blindly placed into either a treatment group (who received ibudilast daily for 96 weeks) or a placebo group (who received a placebo for the same period). The drug was found to be safe, PLUS there was a 48% reduction in whole-brain atrophy at the end of that 96 week period (Click here to read the press release).
And that clinical trial result basically replicated the results from a previous clinical trial that took place in 2008 which involved 297 people with relapsing multiple sclerosis who had received either ibudilast or placebo for 12 months (Click here to read more about that study).
Wow! That is interesting. Has Ibudilast ever been tested in Parkinson’s?
No, the drug has never been clinically tested in Parkinson’s (that I am aware of – happy to be corrected on this). And the pre-clinical research is limited to just one report which was published last year:

Title: The phosphodiesterase inhibitor, ibudilast, attenuates neuroinflammation in the MPTP model ofParkinson’s disease.
Authors: Schwenkgrub J, Zaremba M, Joniec-Maciejak I, Cudna A, Mirowska-Guzel D, Kurkowska-Jastrzębska I.
Journal: PLoS One. 2017 Jul 28;12(7):e0182019.
PMID: 28753652 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers injected mice with ibudilast prior to administering a neurotoxin that kills dopamine neurons (MPTP). While the investigators found ibudilast had no protective effect on the survival of the dopamine neurons, they did report that it significantly reduced inflammation in the brain and an increased level of glial cell-derived neurotrophic factor (of GDNF – Click here to learn more about GDNF), which lead the researchers to speculate whether Ibudilast “may provide a better outcome in the later stages of neurodegeneration”.
The absence of neuroprotection in that study slightly contradicted a previous report using another phosphodiesterase 4 inhibitor called Rolipram:
Title: Attenuation of MPTP neurotoxicity by rolipram, a specific inhibitor of phosphodiesterase IV
Authors: Yang L, Calingasan NY, Lorenzo BJ, Beal MF
Journal: Exp Neurol. 2008 May;211(1):311-4.
PMID: 18328479 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers found that pre-treating mice with Rolipram, a specific inhibitor of the phosphodiesterase 4, protected many of the dopamine neurons in the same model of Parkinson’s as the Ibudilast study mentioned above (MPTP). The level of rescue was significant, but not great and it only occurred in a narrow treatment window.
These results do not really support the use of phosphodiesterase 4 inhibitors as a neuroprotective treatment for Parkinson’s.
Plus there is the phosphodiesterase 4 brain imaging study.
What ‘phosphodiesterase 4 brain imaging study’?
This study was published last year:
Title: Loss of phosphodiesterase 4 in Parkinson disease: Relevance to cognitive deficits.
Authors: Niccolini F, Wilson H, Pagano G, Coello C, Mehta MA, Searle GE, Gunn RN, Rabiner EA, Foltynie T, Politis M.
Journal: Neurology. 2017 Aug 8;89(6):586-593.
PMID: 28701494
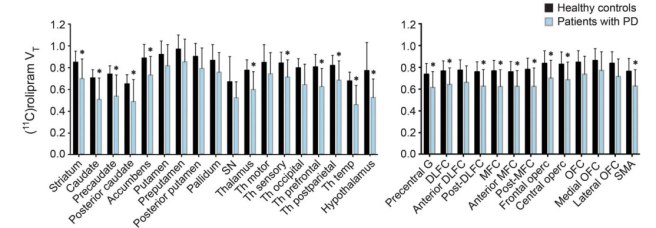
In this brain imaging study from last year, the researchers recruited 12 people with Parkinson’s with no concurrent diagnosis of mild cognitive impairment or dementia, and they imaged their brains for levels of phosphodiesterase 4 protein. The investigators then compared the data with brain images taken from 12 healthy control subjects.
The results of the study suggest that people with Parkinson’s actually have less phosphodiesterase 4 protein than healthy controls in numerous regions of the brain (see graph below), and particularly low levels of phosphodiesterase 4 protein were associated with deficits of spatial working memory.
Lower levels of phosphodiesterase 4 protein in PD. Source: Neurology
This result would suggest that phosphodiesterase 4 inhibitors (like Ibudilast) may not be ideal for Parkinson’s. If the levels of phosphodiesterase 4 protein are already lower than normal in Parkinson’s, inhibiting the protein further may not be such a good idea.
And this result in similar to what has been previously observed with another phosphodiesterase: phosphodiesterase 10A (Click here and here to read more about that research).
So perhaps the inhibition of phosphodiesterases in Parkinson’s may not be a great idea – or at the very least, each phosphodiesterase will need to be investigated individually.
Ok, so are we wasting time testing of phosphodiesterase inhibitors in Parkinson’s?
No.
While they may not rescue dopamine neurons, some of these phosphodiesterase inhibitors do have some interesting properties that could still be useful. Such as the phosphodiesterase 10A inhibitor, MR1916, which may be useful in the treatment of Ldopa-induced dyskinesias:
Title: A selective phosphodiesterase 10A inhibitor reduces l-dopa-induced dyskinesias in parkinsonian monkeys.
Authors: Beck G, Maehara S, Chang PL, Papa SM.
Journal: Mov Disord. 2018 Mar 6. doi: 10.1002/mds.27341.
PMID: 29508924
Five MPTP‐treated monkeys with advanced Parkinson’s-like features and L‐dopa‐induced dyskinesias were treated with MR1916 or a placebo control substance daily for 5 weeks. MR1916 consistently reduced L‐dopa‐induced dyskinesias, and this effect was significant with every dose of MR1916 that the investigators tested. The drug was as good, or better in some cases than Amantadine (a standard treatment for dyskinesias).
And MR1916 is not the only phosphodiesterase inhibitor that has interesting characteristics. For example, this study from last year highlights another interesting phosphodiesterase inhibitor:

Title: Protective efficacy of phosphodiesterase-1 inhibition against alpha-synuclein toxicity revealed by compound screening in LUHMES cells.
Authors: Höllerhage M, Moebius C, Melms J, Chiu WH, Goebel JN, Chakroun T, Koeglsperger T, Oertel WH, Rösler TW, Bickle M, Höglinger GU.
Journal: Sci Rep. 2017 Sep 13;7(1):11469.
PMID: 28904388 (This article is OPEN ACCESS if you would like to read it)
In this study, the investigators screened 1,600 FDA-approved drugs for their efficacy to protect dopamine cells grown in cell culture from degeneration which was caused by very high levels of alpha synuclein. When the researchers looked at the drugs that prevent degeneration the best, they identified dipyridamole (a non-selective phosphodiesterase inhibitor that is used as a medication for inhibiting blood clot formation) as the top drug.
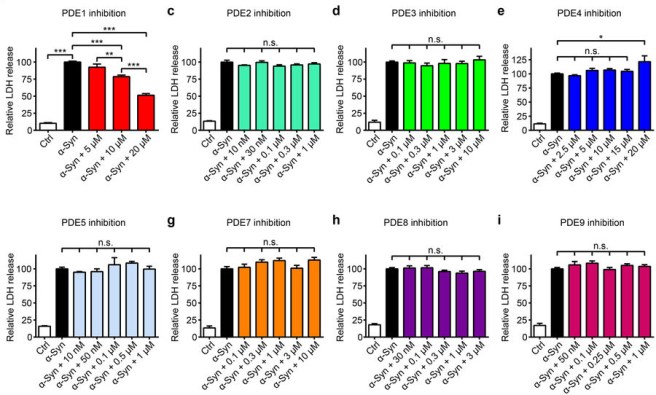
They next conducted a systematic analysis of other phosphodiesterase inhibitors, to try and identify a specific phosphodiesterase inhibitor that could most potently rescue cells from alpha synuclein toxicity. This was the result:
Only PDE1 inhibition (Vinpocetine) reduced alpha synuclein toxicity. Source: Nature
A drug called Vinpocetine (a phosphodiesterase 1 inhibitor) was the only phosphodiesterase inhibitor that reduced alpha synuclein toxicity. Vinpocetine is a synthetic alkaloid derived from the periwinkle plant. It is synthesised from a molecule called ‘vincamine‘. Vinpocetine is not FDA approved for therapeutic use.
The researchers validated Vinpocetine in cell culture conditions and found the neuroprotection was mediated by an increase of cGMP. They also found that Vinpocetine could rescued dopamine neurons in mice with very high levels of alpha synuclein.
The investigators concluded “inhibition of phosphodiesterase 1 in particular as promising target for neuroprotective treatment” for Parkinson’s.
Is Vinpocetine being clinically tested in Parkinson’s?
No, not that I am aware of.
Are any phosphodiesterase 1 inhibitors being tested in the clinic for Parkinson’s?
Yes.
A biotech company called Intra-Cellular Therapies has just initiated a clinical trial.
The company is currently recruiting 24 subjects to be involved in a Phase I/II randomised, double-blind, placebo-controlled study in people with idiopathic Parkinson’s to evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of their candidate drug, an orally administered phosphodiesterase 1 inhibitor called ITI-214 (Click here to read more about this clinical trial).
This trial is very short and we should know the results of it by the end of this year.
What does it all mean?
When readers ask me about a particular experimental agent or drug, I give them the facts of what has been published regarding that compound in models of Parkinson’s (or where possible clinical trials). Sometimes, as in the case of phosphodiesterase inhibitors (like Ibudilast, ITI-214, or Vinpocetine) there simply isn’t much evidence which makes it hard to form an opinion (let alone a post for the blog).
While phosphodiesterase inhibitors are an interesting and exciting new class of drugs being developed for a range of medical conditions, there is a great deal of further research required before any solid opinions can be given with regards to their utility in Parkinson’s.
So while I’ll be watching for news regarding Ibudilast in ALS at the American Academy of Neurology meeting later this month, I will be more interested in ITI-214 in Parkinson’s results later this year.
ADDENDUM – 25th April, 2018
On the 24th April, MediciNova presented the SPRINT-MS Phase 2b clinical trial results of MN-166 (Ibudilast) in progressive Multiple Sclerosis at the American Academy of Neurology Annual Meeting.
The study met its primary endpoint with Ibudilast treatment resulting in a 48% reduction in the rate of progression of whole brain atrophy compared to placebo. And an 80% reduction in the progression of cortical atrophy compared to placebo.
Interestingly: Ibudilast had no effect on the progression of retinal nerve fiber layer thinning.
To learn more about the result, click here to read the press release.
ADDENDUM – 23rd October, 2018
Intra-Cellular Therapies today announced the results from their Phase 1/2 clinical study evaluating ITI-214 in 40 patients with mild to moderate idiopathic Parkinson’s. The results suggest that once-daily ITI-214 treatment for 7 days was shown to be safe and generally well tolerated across a broad range of doses from 1 mg to 90 mg in subjects.The company suggested that the results “support the advancement of the ITI-214 development program in Parkinson’s” (Click here to read the press release).
The banner for today’s post was sourced from Linkedin









At work and down to an iPhone so posting in the wrong spot but interesting https://rd.springer.com/article/10.1007%2Fs10967-012-1860-5
LikeLike
Hi Joe,
That is interesting! I am waiting for the good folks at Clinicrowd to publish their results (a work in progress apparently) before doing another post on Mannitol, but that link will be very useful for that post. Thanks!
Kind regards,
Simon
LikeLike
Living in Eastern Europe as I do, Cavinton is a well known and oft prescribed treatment for improving vascular related cognitive decline. Last MRI I had identified some few age-related lesions, so my neurologist put me on a 12-week course of Vinpocetine. I really have no cognitive issues of which I am aware, BUT, in a computer class I recently took (in a foreign language, no less, where I have had concentration issues in the past when the course was not in English), I had relatively no problems at all with paying attention or understanding. And that, even the though the course materials had been translated out of English. As most IT folks know, English is the language of Computer Science, and I am usually able to follow along in a class quite well with my University German IF the materials are at least in English. However, this time, it seemed to make little difference.
Coincidence? Placebo effect? Who knows, but I’m not squawking about it.
LikeLike
Hi Gavril,
Thanks for your interesting comment. Vinpocetine is an interesting compound and your experience is intriguing. There was a report in 2003 that reviewed Vinpocetine in folks with cognitive impairment and dementia and it suggested limited effect (http://cochranelibrary-wiley.com/doi/10.1002/14651858.CD003119/abstract;jsessionid=9AB1E17DB868240F33F87E924B09F16A.f04t01). But those individuals may have been too impaired. Who knows. Maybe worthy of further investigations, particularly if it is impacting alpha synuclein toxicity. There is also suggestions that Vinpocetine could have anti-inflammatory properties (http://www.pnas.org/content/107/22/9921) which would make it even more interesting. It deserves further research.
Kind regards,
Simon
LikeLike
I was curious about Ibudilast for parkinson’s since a paper I read found a potentially high glial component of the disease.
See https://www.biorxiv.org/content/biorxiv/early/2019/01/23/528463.full.pdf
Ibudilast is an antagonist to the Toll-like receptor 4 which when blocked attenuates certain glial activity.
See: https://www.ncbi.nlm.nih.gov/pubmed/15901793
https://www.ncbi.nlm.nih.gov/pubmed/19762094
Found this page while looking for any research. It would be expected that ibudilast wouldn’t rescue cell loss via MPTP.
LikeLike