|
A lot of Parkinson’s research is focused on antioxidants – molecules that can reduce the level of stress a cell is under when it is not feeling well. One of the most widely discussed antioxidants on Parkinson’s online forums is a molecule called N–acetylcysteine (or NAC). Recently, the results of a small clinical trial – in which NAC was administered to people with Parkinson’s – have been published. The results are rather interesting. In today’s post, we will discuss what NAC is, why it is important in the context of Parkinson’s, and we will look at what the new clinical trial report suggests about this molecule.
|
 NAC. Source: Draxe
NAC. Source: Draxe
One question I get asked a lot is “What do you think of NAC?”
And I usually answer with my standard “I’m not a clinician, just an ex-research scientist. I can’t talk about medications or supplements, etc…”
But recently some interesting new data has been published regarding NAC and it’s kind of interesting.
What is NAC?
N-acetylcysteine (or NAC; also known as Acetylcysteine – commercially named Mucomyst) is a prodrug – that is a compound that undergoes a transformation when ingested by the body and then begins exhibiting pharmacological effects.
 Acetylcysteine. Source: Wikimedia
Acetylcysteine. Source: Wikimedia
Acetylcysteine serves as a prodrug to a protein called L-cysteine, and – just as L-dopa is an intermediate in the production of dopamine – L-cysteine is an intermediate in the production of another protein called glutathione.
If you remember nothing else today, remember this: Acetylcysteine allows for increased production of glutathione.
And what is glutathione?

Glutathione. Source: Wikipedia
Technically speaking, glutathione (pronounced “gloota-thigh-own”) is a tripeptide (a string of three amino acids connected by peptide bonds) containing the amino acids glycine, glutamic acid, and cysteine.
Less technically speaking, glutathione is a potent antioxidant that is produced naturally in nearly all the cells of your body.
What is an antioxidant?
An antioxidant is simply a molecule that prevents the oxidation of other molecules.
Which begs the obvious question:
What is oxidation?
Oxidation is the loss of electrons from a molecule, which in turn destabilises the molecule.
Think of iron rusting.
Rust is simply the oxidation of iron. In the presence of oxygen and water, iron molecules will lose electrons over time. Given enough time, this results in the complete break down of objects made of iron.

Rusting iron. Source: Thoughtco
And the exact same thing happens in biology. Molecules in your body go through a similar process of oxidation – losing electrons and becoming unstable. This chemical reaction leads to the production of what we call free radicals, which can then go on to damage cells.
What is a free radical?
A free radical is an unstable molecule – unstable because it is missing an electron. They react quickly with other molecules, trying to capture the needed electron to re-gain stability. Free radicals will literally attack the nearest stable molecule, stealing an electron. This leads to the “attacked” molecule becoming a free radical itself, and thus a chain reaction is started. Inside a living cell this can cause terrible damage, ultimately killing the cell.
Antioxidants are thus the good guys in this situation. They are molecules that neutralize free radicals by kindly donating one of their own electrons. The antioxidant don’t become free radicals by donating an electron because by their very nature they are stable with or without that extra electron.

How free radicals and antioxidants work. Source: h2miraclewater
When the cells in the brain are affected by free radicals or any other toxic agent, glutathione is released into the space outside of the cells to help defend them from any attack, and glutathione within the cells to employed to deal with anything that actually manages to enter them.
So to answer your question, glutathione is a naturally produced antioxidant which plays an important in removing free radicals from the brain.
Do we know anything about glutathione with regards to Parkinson’s?
Yes we do.
First, glutathione appears to be reduced in many important areas of the Parkinsonian brain. This was recognised way back in 1982:

Title: Parkinson’s disease: a disorder due to nigral glutathione deficiency?
Authors: Perry TL, Godin DV, Hansen S.
Journal: Neurosci Lett. 1982 Dec 13;33(3):305-10.
PMID: 7162692
In this study, the investigators began by analysing levels of glutathione in autopsied human brain. They found that in the brains of healthy donors glutathione content was generally significantly lower in the substantia nigra (a region severely affected by Parkinson’s) than in other brain regions.
Interestingly, when they looked at glutathione levels in the substantia nigra of people who passed away with Parkinson’s, they barely detected any glutathione at all!
And this result has been subsequently repeated by several other independent groups (Click here, here and here to see some examples). This discovery led investigators excitedly questioning whether the Parkinson’s was partly the result of a glutathione deficiency.
Unfortunately for those excited investigators, glutathione deficiency is not specific to Parkinson’s:

Title: Nigral glutathione deficiency is not specific for idiopathic Parkinson’s disease.
Authors: Fitzmaurice PS, Ang L, Guttman M, Rajput AH, Furukawa Y, Kish SJ.
Journal: Mov Disord. 2003 Sep;18(9):969-76.
PMID: 14502663
In this study, the researchers found that decreased levels of glutathione in the substantia nigra is a common feature across several neurodegenerative conditions, including progressive supranuclear palsy (PSP) and multiple system atrophy (MSA).
Interestingly they also saw a trend towards decreased levels of uric acid (another antioxidant that we have previously discussed – Click here to read that post) in the substantia nigra of all the neurodegenerative groups analysed (a decrease of -19 to -30%). In addition, previous reports have indicated that glutathione levels in the brains of people with Alzheimer’s are significantly reduced (click here for more on this). Thus, reduced levels of glutathione appears to be a common feature of neurodegenerative conditions.
Researchers have subsequently found that decreased levels of glutathione does not directly result in dopamine cell loss (Click here to read more about this), but it does make the cells more vulnerable to damaging agents (such as neurotoxins, etc – Click here and here to read more about this). This has lead investigators to ask whether administering glutathione to people with Parkinson’s would slow done the condition.
Has glutathione ever been tested in clinical studies for Parkinson’s disease?
Yes it has.
At least four times in fact. The first study was reported in 1996:

Title: Reduced intravenous glutathione in the treatment of early Parkinson’s disease.
Authors: Sechi G, Deledda MG, Bua G, Satta WM, Deiana GA, Pes GM, Rosati G.
Journal: Prog Neuropsychopharmacol Biol Psychiatry. 1996 Oct;20(7):1159-70.
PMID: 8938817
In the first clinical trial to test glutathione in Parkinson’s, the investigators recruited nine people with recently diagnosed (and untreated) Parkinson’s. Glutathione was administered intravenous twice per day (600 mg each time) for 30 days. The drug was then discontinued and the participants were followed up for 4 months.
All of the participants in this study improved significantly after glutathione treatment (an average of 42% decline in disability) and the beneficial effects lasted for 2-4 months after the treatment was stopped at the end of the study. Particularly interesting was the change in resting tremor intensity:
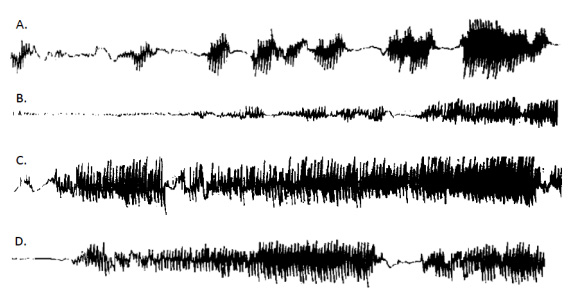
Intensity of the resting tremor of a subject with PD at baseline (A), after 30 day of glutathione treatment (B), 60 days after stopping glutathione treatment (C), and 30 days after returning to L-dopa (D). Source: Sciencedirect
Following this study, a series of videos were placed online which caused a lot of excitement within the Parkinson’s community:
A second video demonstrated some beneficial effects in three other people with Parkinson’s:
One cautionary note regarding the first glutathione study and these subsequent videos, however, is that in all of these cases, the investigators and the participants were not blinded – that is to say, they all knew who was getting the treatment and what the treatment was. Thus, investigator bias and the placebo effect could have been at work.
To deal with this possibility a randomised, double blind clinical study was organised by Dr David Perlmutter (one of the investigators who first posted the videos online):

Title: Randomized, double-blind, pilot evaluation of intravenous glutathione in Parkinson’s disease.
Authors: Hauser RA, Lyons KE, McClain T, Carter S, Perlmutter D.
Journal: Mov Disord. 2009 May 15;24(7):979-83.
PMID: 19230029
The investigators of this study randomly assigned 20 subjects with Parkinson’s to receive intravenous glutathione (1,400 mg) or a placebo treatment (10 people in each group). Both were administered three times a week for 4 weeks. The investigators found that glutathione was well tolerated and there were no withdrawals due to any adverse events of the treatment.
But at the end of the study, there were no significant differences in changes in Unified Parkinson’s Disease Rating Scale (UPDRS) scores. The UPDRS is a universally utilised method of scoring the severity of Parkinson’s disease features/symptoms. Over the 4 weeks of study, the glutathione group exhibited only a mild improvement when compared to the control. The researchers concluded by suggesting that further evaluation was required in a larger, longer study.
Intravenous delivery of glutathione is not an ideal treatment approach for any medication, particularly for a community that has movement issues. And this was taken into consideration when the next clinical trial of glutathione was conducted:

Title: A randomized, double-blind phase I/IIa study of intranasal glutathione in Parkinson’s disease.
Authors: Mischley LK, Leverenz JB, Lau RC, Polissar NL, Neradilek MB, Samii A, Standish LJ.
Journal: Mov Disord. 2015 Oct;30(12):1696-701. doi: 10.1002/mds.26351. Epub 2015 Jul 31.
PMID: 26230671 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers utilised a nasal spray approach to get glutathione into the body. The goal of the study was to test the safety and tolerability of intranasal delivery of glutathione in people with Parkinson’s. The investigators recruited 30 individuals with Parkinson’s, who were randomly assigned to either a placebo/control group (saline) or to a treatment group (two doses were tested on the treatment group – 300 mg/day or 600 mg/day of intranasal glutathione). The study was conducted over 3 months and found little difference between the groups with regards to the clinical features of Parkinson’s, but did find that intranasal glutathione was well tolerated by all of those involved in the study.
This first trial has subsequently led to a phase II study to look more carefully at the effectiveness of intranasal delivery of glutathione.

Title: Phase IIb Study of Intranasal Glutathione in Parkinson’s Disease
Authors: Mischley LK, Lau RC, Shankland EG, Wilbur TK, Padowski JM.
Journal: J Parkinsons Dis. 2017 Apr 20. doi: 10.3233/JPD-161040.
PMID: 28436395 (This article is OPEN ACCESS if you would like to read it)
Curiously in this double-blind, placebo-controlled clinical trial, all of the 45 individuals (averaging 3.5 years since diagnosis) involved in the study improved during the study (including the control group), suggesting that there was some kind of placebo effect at play within the cohort. The subjects were divided into three groups: a control group, a low dose treatment group, and a high dose treatment group for the intranasal glutathione treatment.
The high-dose group demonstrated improvement in total UPDRS score over their baseline scores, but neither treatment group improved more than the placebo control group (who also improved). It was therefore difficult for the researchers to conclude that glutathione is superior to placebo after a three month intervention (although obviously there may have been a placebo effect occurring in the control).
So glutathione treatment doesn’t work?
I would not interpret the results that way.
Firstly, none of these clinical studies have been correctly powered. That is to say, they did not recruit enough participants for major conclusions to be made of their results (except that collectively they indicate that glutathione appears to be safe for use in folks with Parkinson’s).
In addition, they have all been rather short studies (conducted over a matter of weeks or months) which makes determining changes in UPDRS scores or simply disease progression rather difficult. Parkinson’s is a slow progressive condition. Therapies that slow or halt the disease may require longer periods of time to demonstrate efficacy. Short term improvements could simply be symptomatic.
Are there any clinical trials being conducted at the moment for glutathione in Parkinson’s?
There have been quite a few clinical trials involving glutathione.
But one of the big issues with using glutathione as a treatment is that it is not absorbed very well by cells.
Generally, glutathione is made inside cells, not taken up by them.
Oh, I see. Bummer. But glutathione can still help reduce oxidation issues outside the cells?
Yes, but what we really need is molecules that can increase glutathione levels inside of cells.
And this is where we come back to where we started: NAC.
If you will recall what I said at the start, “NAC allows for increased production of glutathione”.
NAC is readily taken up by neurons and it can help to boost neuronal glutathione levels. NAC has also been shown to be neuroprotective in animal models of Parkinson’s (Click here to read more on this), which has led researchers to investigate its use as a potential treatment for Parkinson’s:

Title: N-Acetyl Cysteine May Support Dopamine Neurons in Parkinson’s Disease: Preliminary Clinical and Cell Line Data.
Authors: Monti DA, Zabrecky G, Kremens D, Liang TW, Wintering NA, Cai J, Wei X, Bazzan AJ, Zhong L, Bowen B, Intenzo CM, Iacovitti L, Newberg AB.
Journal: PLoS One. 2016 Jun 16;11(6):e0157602.
PMID: 27309537 (This article is OPEN ACCESS if you would like to read it)
The researchers in this study began their investigation by looking at the effect of NAC on the survival of dopamine neurons (made from human stem cells) in culture after exposure to a neurotoxin (rotenone; which is a pesticide/insecticide that has been associated with Parkinson’s – click here to read more on this).
They found that NAC treatment resulted in significantly more dopamine neurons surviving the exposure to rotenone when compared to cultures that received no NAC. Armed with this result, the investigators boldly moved straight into the clinic and started a clinical study (click here for the details of that trial).
They took 23 people with Parkinson’s (average time since diagnosis was approx. 3 years) and randomly assigned them to either the NAC group (12 subjects) or the control group (11 subjects). The study was not blinded, so both the investigators and the participants knew which treatment they were being administered. And the NAC was administered both intravenously and orally.
At the start of the study, all of the subjects were assessed using the UPDRS rating scale and imaging of the brain was conducted (using DaTscan). During the study, both groups continued their normal treatment for their Parkinson’s symptoms, with the experimental group also receiving NAC (50mg/kg) once per week. After approximately 90 days of NAC treatment, all of the subjects underwent a follow up evaluation, which included another UPDRS assessment and DaTscan & SERT brain images.
After the study, the researchers found that the UPDRS score of the NAC-treated group dropped from an average of 25.6 to 22.3, while the control group increased from 20.2 to 22.2. The UPDRS has a list of 55 items that are graded 0-4 depending on level of severity. A lower score indicates a more normal level of activity. Thus, any decrease in UPDRS score can be seen as a positive outcome – in this case the treated group improved by 12.9% on average.
But again, as with the glutathione studies mentioned above, we can’t read too much into this result because the study was very short and it was also open label. The clinical improvement could simply be a placebo response (a positive benefit based on no biological explanation).
What about the brain imaging?
Well, this is where the study got interesting. The NAC treated individuals exhibited a significant increase in the levels of dopamine activity in the caudate and putamen in the brain (averaging between 4.4% to 7.8%).
That might seem like a small difference, but it is rather remarkable for a 90 day study.
These results got the investigators excited and they decided to conduct a second study with the goal of gaining more data on the effect of NAC administration on Parkinson’s. And it was that second study which was published very recently:
 Title: N-Acetyl Cysteine Is Associated With Dopaminergic Improvement in Parkinson’s Disease.
Title: N-Acetyl Cysteine Is Associated With Dopaminergic Improvement in Parkinson’s Disease.
Authors: Monti DA, Zabrecky G, Kremens D, Liang TW, Wintering NA, Bazzan AJ, Zhong L, Bowens BK, Chervoneva I, Intenzo C, Newberg AB.
Journal: Clin Pharmacol Ther. 2019 Jun 17. [Epub ahead of print]
PMID: 31206613
In this study, the researchers took 42 people with Parkinson’s (average time since diagnosis being 4 years) and randomly assigned them to either administration of NAC for three months or standard care. The NAC was given both intravenously (once per week) and orally (twice per day).
The participants underwent DaTscan and SERT brain imaging at the start of the study and at the end of the study.
Hand on a second. What is DaTscan and SERT brain imaging? And why do both?
Dopamine and serotonin are two chemicals in our brains that are used for passing messages between cells. DaTscan is a method of brain imaging that allows us to look at levels of dopamine activity in a living brain. One of the issues with DaTscan, however, is that it is not very selective and it will sometimes also show serotonin activity. SERT is a brain imaging method that is used to measure serotonin activity in the brain. By conducting both DaTscan and SERT brain imaging, the researchers were hoping to get a better idea of what NAC was affecting in the brain.
At the end of the study, the researchers reported that the NAC-treatd group exhibited a significant increase in dopamine activity (based on the DaTscan) in the caudate and putamen (where the bulk of the dopamine in our brains is released). The average increase was between 3.4% to 8.3%, compared to the control group. And this was associated with significantly improved Parkinson’s symptoms (but again, remember this was an open label study!).
The investigators concluded that the “results suggest NAC may positively affect the dopaminergic
system in patients with Parkinson’s, with corresponding positive clinical effects. Larger scale studies are warranted“.
We are now waiting to hear about the follow up study. I will keep you updated as more information becomes available.
Are there no ongoing clinical trials for NAC?
There is one that I am aware of which is being conducted at the Centre Hospitalier Universitaire, in Amiens (France). But this trial is a 10 week treatment study exploring “mild-to-moderate impulse control disorders induced by dopaminergic medications in Parkinson’s” (Click here to read more about this study).
It is a large study (70 participants), but it does not appear to be investigating disease modification.
So what does it all mean?
When people ask me for an opinion on on NAC, I am still kind of stuck as to what to actually say.
 NAC. Source: Wikimedia
NAC. Source: Wikimedia
Beyond all of the ‘not a clinician’ stuff, I am still not comfortable with giving it a thumbs up or a shake of the head. We need to see a well designed, long term demonstration that this critical ingredient in the production of glutathione is a good option for folks with Parkinson’s. The evidence presented above is enticing and compelling, but it is all based on short-term, open-label trials. I completely agree with the investigators of the most recent study when they say “Larger scale studies are warranted“.
So what we need to see now are some large scale clinical trials. And these studies will need to be well structured. In the most recent report, the researchers state in the discussion that their “findings were not consistent across all subjects“. Thus, as with the exenatide trial results (Click here to read more about that), there may be some forms of Parkinson’s that respond to NAC, and others that do not. Any follow up study will need to take this into account and be sufficiently large enough so that this idea can be explored during the study – what is it about some people that makes them response to NAC and not others?
I will be very keen to see a large NAC study. Fingers crossed we hear more about this over the next 12 months.
The banner for today’s post was sourced from thyroidpharmacist

Ask Dr. Mischley to do a larger study. She’s already headed a couple and seems the most likely to get the job done
LikeLike
Hi Scott,
Thanks for your comment. You are right, she is certainly the right person for the job. But I know from speaking to her that she has tried finding the funding for that study. This is the elephant in the room with NAC: given that there is no intellectual property to provide the possibility of profits, few organisations are prepared to take it on. Hoping that this post will help to stimulate some interest.
Kind regards,
Simon
LikeLike
The NAC was administered orally and intravenously in the study – so I suppose PD patients would need to do the same if they wanted to follow the same protocol.
Pill form is easy, but not sure about how easy the intravenous administration would be for the typical individual to perform without a physician.
LikeLike
Hi rjj,
Thanks for your interesting comment. Intravenous admin is certainly not ideal. I was wondering about the nasal spray route that was used in the glutathione study. It would certainly be better for folks having issues with swallowing. I am not sure if there is a nasal spray formulation of NAC (opportunity for someone maybe?). The pill route does appear to be the least potent method of delivery though – the researchers who conducted the most recent study mention this in their discussion.
Kind regards,
Simon
LikeLike
Just noticed that NAC is available in inhaled form. Presumably not quite the same as nasal spray form, but perhaps it would be more potent than pill form. https://en.wikipedia.org/wiki/Acetylcysteine (first paragraph)
LikeLike
There should be a way of harnessing UK NHS patients using simple questions on what vitamins and supplements they are taking regularly on each visit with the Parkinson Nurse (if you are lucky enough to have access). Then correlate with disease progress.
A fair percentage of people with PD may already be taking this, partly as it is touted for so many other conditions such as dodgy joints or lung issues. Just Google NAC benefits!
It is sold both as caps and as powder – tastes fine when mixed with something sweet such as blackcurrant juice. Even better hot with rum when you have a dreadful cold.
LikeLike
Hi Linda,
Thanks for your comment. It is an interesting idea, but I’m really not sure where to begin with that hunt for data. NHS medical records are very accurate with regards to prescribed medications, but have very little on what supplemental nutriments individuals might be taking. There must be a way. Will have a bit more of a think about it.
Kind regards,
Simon
LikeLike
If the NHS wants to reduce it’s drugs bill, they are likely to be cooperative with any organisation willing to prove that substances from the supermarket health or sports supplement section are useful for some specific condition. It should be possible to distribute a very simple form asking permission to use the data, with a tick box list of possible pills and potions that they take regularly,such as multi vits, individual vits such as d3, b12 etc, cod liver oil, asprin – and NAC. Then a similar tick box list of main symptoms such as shakes, freezing, mood, bladder etc followed by a box for assesment of whether things have improved, stayed the same or got worse in the last year, 3 years etc.
Then you need a researcher to crunch the numbers.
How about the group involved in Trust me I am a doctor? There are enough people with relatives with PD for this to make a great story – and push the need for research where no one makes a profit due to lack of patents.
Also prove or debunk some of the other NAC claims.
LikeLike
Interesting, Seeing any effect of NAC in a short period suggests it is hitting the spot. BUT there is a far better way to raise glutathione synthesis and utlisation, and raise serotonin levels in the brain, and improve expression of antioxidant pathways. Vitamin D3. This is a readable review of what D3 does in neurodegenerative disease:
Click to access rstb20150434.pdf
D3 also has pronounced anti-inflammatory actions.
In the fullness of time I anticipate D3 will become a mainstay of stabilising iPD. But there’s no profit in it.so the trials to date have been too short and far too cautious on dosage. Needless to say I take lots.And on a sample size of one it seems ( in conjuction with other physilogicla agents ) to have reduced tremor and almost abolished meg muscle cramping.
LikeLike
Hi Peter,
Interesting review – thanks for sharing.
D3 is really interesting. While observational studies have suggested that (relative to healthy controls) people with Parkinson’s have lower circulating levels of D3, there does not appear to be any causal association ( https://www.ncbi.nlm.nih.gov/pubmed/28594127 ). But there was a clinical trial in 2013 that tested D3 supplementation in PD which had some interesting results ( https://www.ncbi.nlm.nih.gov/pubmed/23485413 ). They had 56 individuals with PD in the treatment group (1200 IU/d) and 58 in placebo group, for 12 months double blind testing, and the results suggest differing beneficial effects based on the vitamin D receptor Fokl genotype of the participants. Remarkably, the researchers don’t appear to have followed up this result. The other clinical studies exploring D3 in PD have been smaller and shorter (for example, https://www.ncbi.nlm.nih.gov/pubmed/30256811), but they all suggest that D3 is very safe.
I was not aware of the D3-glutathione angle (https://www.ncbi.nlm.nih.gov/pubmed/11893522), but will investigate this further.
Kind regards,
Simon
LikeLike
Hi Simon, Berridge wrote a couple of other articles on D3 and neurodegeneration emphaising its central role in anti-oxidant signalling and calcium homeostasis.eg:
https://www.researchgate.net/publication/277251978_Vitamin_D_A_custodian_of_cell_signalling_stability_in_health_and_disease/link/556ec1b408aeab777226ad9e/download
He discovered the PIP2>InsP3>Ca mobilisation signalling pathway- check out his Wiki.He is unlikely to be wrong !
Although the D3 for PD trials have not to me been a resounding success, mechanistically D3 looks very important. D3 is also involved in neuroinflammation, promoting microglial M2 phenotype over the agressive pro-inflammatory M1. However the proinflammatory facotr NFkB acts to suppress activity of the VitDReceptor. This , to me, points to the need for high dose D3 to try to overcome that feedback. I now use 12,000 IU D3 per day ( plus Vit K2 ) on the basis that it is witihn the daily max D3 synthesis rate by skin in sunlight ( ca 20,000 IU pd). Safety of such a dose is embraced in this important paper:
https://www.sciencedirect.com/science/article/abs/pii/S0960076018306228#bib0410
I could go on..and on. But you get the gist: a high dose D3 trial is badly needed ( with K2).
Best wishes,
Peter
LikeLike
Gentlemen, what are your thoughts on the role of magnesium in the function of Vit D? There seem to be a few studies that examine the interplay between these two substances, such as this one: https://doi.org/10.7556/jaoa.2018.037
LikeLike
You are SUPERMEN for me
LikeLike
Thank You
LikeLike
Good stuff. Regarding thumbs up or down, surely just as important as the result of trials is the safety aspect. If NAC or l-glutathione supplementation has little to no risk, then I see no downsides to trying it?
Regarding l-glutathione specifically, I was reading a report the other day that suggested previous studies claiming no absorption were incorrect, and more thorough analysis with Setria proves it absorbs just fine.
LikeLike
Hi, let me refresh this interesting topic. It was like 3 years ago when the latest research has been completed on NAC in PD. I wonder if there was any new study related to this subject?
cheers
MH
LikeLike
Has anyone ever looked at L-Tyrosine as a precursor to dopamine for PD? Particularly N-Acetyl L-Tyrosine to aid passing through the blood brain barrier?
Or is there a possibility of N-Acetyl Levodopa? Levadopa itself has a hard time passing the blood brain barrier but N-Acetyl seems to be a good catalyst for this.
LikeLike
Hi Simon, thanks for the article. Have there been any updates, please?
LikeLike
Why not use simply L-cysteine rather than NAC?
LikeLike