|
In this end-of-year post, we review the Parkinson’s research that caught our attention at SoPD HQ in 2019. Month-by-month we will briefly discuss some of the major pieces of research/announcements that have defined the year and advanced our understanding of Parkinson’s. The list is based on nothing more than the author’s personal opinion – apologies to any researchers who feel left out – and the contents should certainly not be considered definitive or exhaustive. It was just some of the stuff that made me say “wow” in 2019. And in the next post, we will conduct our annual horizon scan and consider what 2020 may have in store for us.
|
 Source: a-star
Source: a-star
2019 was a productive year for the Parkinson’s research community.
Wait a minute. Hold your horses. What is that statement based on?
If we use number of research report published in 2019 as our measure, there was a total of 8094 articles added to the Pubmed website with the tag word “Parkinson’s” attached (compared to 7672 for all of 2018 and 7675 for 2017). That sounds rather productive.
In addition, there were a host of new clinical trials initiatiated, many of which are exploring entirely new experimental therapies. These include:
- UDCA (aka Ursodeoxycholic acid) – A bial acid therapy used for reducing gall stones that may improve mitochondrial function entered Phase II testing for Parkinson’s (Click here to read a SoPD post on the topic).
- PR001 – A gene therapy targetting GBA-associated Parkinson’s (Click here to read a SoPD post about this).
- CNM-Au8 – Gold nanoparticles entered Phase II testing for Parkinson’s (Click here to read an SoPD post about this research).
- Terazosin – This prostatic hyperplasia and hypertension drug was found to enhance Phosphoglycerate kinase 1 (Pgk1) activation & a Phase II trial was immediately initiatiated (Click here to read an SoPD post on this topic).
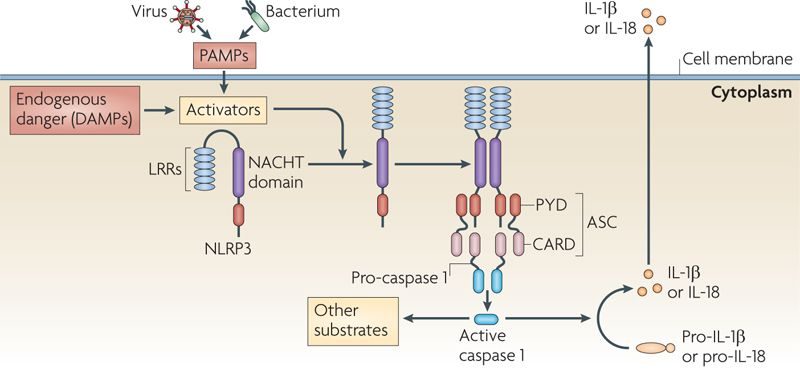
- Inzomelid – An NLRP3 inhibitor from Inflazome began Phase I testing (Click here to read a SoPD post on this topic).
On top of all of this, numerous novel potenially therapeutic pathways were proposed, such as:
- Farnesyltransferase inhibition (Click here to read a SoPD post on the topic)
- Miro1 degradation (Click here to read an SoPD post on the topic).
- CD22 inhibition (Click here to read a SoPD post on this topic).
- Felodipine – Researchers discovered that this L-type calcium channel blocker & anti-hypertensive drug boosts waste disposal (or autophagy) in mouse brains (Click here to read an SoPD post on the topic).
Plus, there were a number of major Parkinson’s research organisations launched, including the Australian Parkinson’s Mission (Click here to read more about this), Aligning Science Across Parkinson’s (ASAP – click here to read more about this), the Accelerating Medicines Partnership for Parkinson’s disease (or AMP-PD) initiative (Click here to read more about this), and the Chan Zuckerberg Initiative.
Based on all of this, I think it is safe to say that 2019 was a productive year for Parkinson’s research.
Ok, all of that sounds great, but what does that mean for someone living with the condition?
Fair point.
Towards the end of 2019, in the writing of this blog, I have come to appreciate that “hope fatigue” is a very real thing. And as a result, I am now trying to be more careful about what I write (and how I write about it) here on the SoPD.
Yes, no disease modifying therapies have been clinically approved for Parkinson’s during 2019, but a great deal of research was conducted during the last 12 months and there was some immediately applicable research.
For example, researchers in the Netherlands published the results of a double-blind, randomised controlled clinical trial (the PARKI-in-Shape study) which explored the effectiveness of at home aerobic exercise on Parkinson’s. And they found that the exercise group had a statistically significant slower rate of disease progression compared to a non-treatment control group (Click here to read a SoPD post on this topic).
In addition, during 2019 there were a number of new symptomatic treatments that were approved for clinical use by the regulators (such as Inbrija or CVT-301 – an inhaled form of levodopa by Acorda Therapeutics).
Having said this, one could certainly make an easy argument for there being an urgent need for more immediately applicable research. We will hopefully find some time to explore this idea in a future post in the new year, but there is a world of opportunity for someone (researcher, funding body, or member of the affected community) to focus on research that has ready application. And such patient relevant investigations could certainly inform our understanding of the condition and influence the wide world of Parkinson’s research.
Enough ranting, let’s begin:
Below we will review the year that was for Parkinson’s research, starting with:
Without any doubt the most exciting bit of Parkinson’s research news in January was the Australian Government announcing the initiation of a $30+ million, 5 year clinical trial programme that will be focused around large multi-arm clinic trials of potentially disease modifying therapies for Parkinson’s (Click here to read the press release, click here to visit the APM website, and click here to read a SoPD post on this topic).
Researchers also reported the use of brain-derived exosome-based biomarkers as objective measures of target engagement using serum from the exenatide Phase II clinical trial in Parkinson’s. Basically the researchers analysed ‘exosomes’ – these are small brain cell-derived sacks or ‘vesicles’ – found in the blood samples of Parkinson’s participants in the Exenatide Phase II clinical study, and they found that the treatment (the diabetes drug exenatide) had augmented insulin signaling inside the exosome. The implications of this data for the research community could be huge, as it could provide a potentially objective measure of target engagement in the brain from a simple blood sample – and this would have implications well beynd the world of just Parkinson’s research. It could be applied to any clinical trial exploring a neurological treatment (Click here to read more about this, and click here to read a SoPD post on this topic).
In January, a biotech firm called Cortexyme & international collaborators reported porphyromonas gingivalis – a bacteria involved with inflammation of the gums – is present in the brain of people with Alzheimer’s. Mice infected with P. gingivalis began to exhibit Alzheimer’s features, and a bacterial protease inhibitor (called COR388) rescued the mouse model (Click here to read more about this). Interestingly, COR388 has already completed a Phase I clinical trial (Click here to read more about this and click here to read an SoPD post on this topic). It could be interesting to explore this bacterial idea in Parkinson’s.
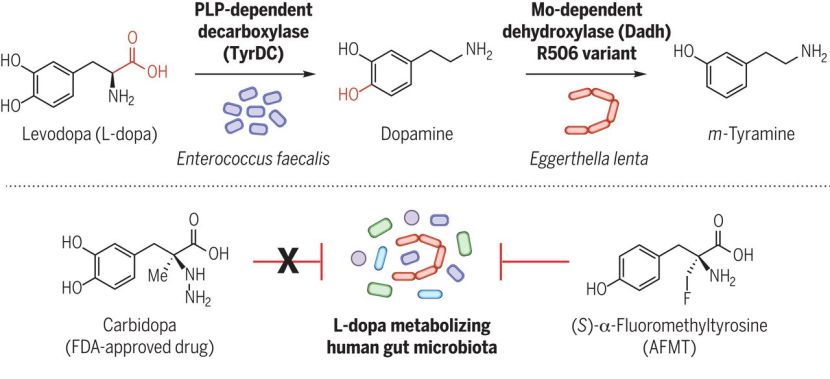
Speaking of bacteria, in January researchers reported that gut bacteria produce an enzyme called tyrosine decarboxylase. This enzyme efficiently converts levodopa – the treatment of Parkinson’s – into dopamine, even in the presence of tyrosine (a competitive substrate) and decarboxylase inhibitors. The investigators also found that high levels of this enzyme compromised levels of levodopa entering the blood system of people with Parkinson’s. The results bring into question the role of microbial metabolism in drug availability in Parkinson’s (Click here to read more about this and click here to read a SoPD post on this topic).
And finally, in January an analysis of a retrospective cohort study of 111 individuals with autopsy-confirmed Parkinson’s suggested that clinical subtypes at the time of diagnosis could be used to estimate disease course, which may be useful in providing a more accurate prognosis in individual patients in the clinical setting (Click here to read more about this).
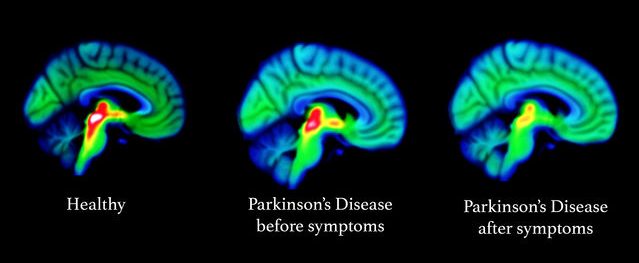
The major news in February was the publication of the Phase II GDNF in Parkinson’s clinical trial results. This was a clinical trial exploring the potential of the neurotrophic factor glial cell line-derived neurotrophic factor (GDNF), a protein that our brains naturally produce which supports neurons. We already knew the study did not meet its primary endpoint (there appears to have been a placebo response), but there were some interesting additional findings in the data, such as the brain imaging (F-DOPA PET) results (Click here and here to read the research reports of the study results and click here to read a SoPD post on this topic).
In February we also saw the initiation of a new clinical trial for Parkinson’s kicking off in Sheffield & London. UDCA (aka Ursodeoxycholic acid or ursodiol) a medication for the treatment of gallstones is being repurposed for Parkinson’s. Precinical data suggests the treatment has mitochondrial benefits in models of Parkinson’s (Click here to read the press release and click here to read a SoPD post on the topic).
A new theory of Parkinson’s was proposed in February which proposed that some parts of the human brain (such as those affected by Parkinson’s) have been “left behind” by evolution as other parts of the brain have expanded. This expansion has placed greater burden on the ‘left behind’ regions which communicate with larger, newer areas, particularly as humans have started to live longer. This new theory could explain why the condition is associated with aging. It also “suggests that disease‐modifying therapies for Parkinson’s are most likely to come from a better understanding of the unique features of the human brain that drive pathogenesis, like axonal biology” (Click here to read more about this and click here to read a SoPD post on this topic).
In February, the Silverstein foundation and the Michael J Fox Foundation announced that they were collaboratively awarding nearly US$3 million in research grants to fund studies investigating an enzyme called beta glucocerebrosidase (or GCase). Genetic variations in the DNA that provides the instructions for making the GCase enzyme are some of the most common genetic risk factors associated with Parkinson’s. One can not help being impressed with the breadth of the areas being covered for just this one subtype of Parkinson’s. The projects cover everything from the genetics underlying the condtion, all the way through to new potential therapeutics. (Click here to read the press release and click here to read a SoPD post on the topic).
A 25 year follow up study of the genetic contribution to Parkinson’s risk in a large population‐based twin study was published in February, and it found that genetic heritability was 0.27 overall – that means that in this study 27% of pairs both developed Parkinson’s. Parkinson’s developed in 83% of twin pairs when one twin was diagnosed under the age of 50 years, and in 19% of twin pairs when one twin was diagnosed over the age of 50 years. The results reenforce a strong genetic component to young onset Parkinson’s, and suggest environmental influences in later forms of the condition (Click here to read more about this).
REM sleep behaviour disorder (RBD) is considered an early sign of Parkinson’s. In a MASSIVE multi-centre study, 1280 people with RBD were recruited & followed over time. The results of the study suggest that the overall conversion rate from RBD to a neurodegenerative syndrome was 6.3% per year, with the risk of phenoconversion after 12-year of 73.5%. The study also found that there was no significant predictive value of sex, daytime sleepiness, insomnia, restless legs, sleep apnoea, urinary dysfunction, orthostatic symptoms, depression, anxiety, or hyperechogenicity on substantia nigra ultrasound. Actual rate of phenoconversion was significantly increased with features like abnormal quantitative motor testing, objective motor examination, olfactory deficit, mild cognitive impairment, erectile dysfunction, motor symptoms, an abnormal DAT scan, colour vision abnormalities, constipation, REM atonia loss, and age (Click here to read more about this).
A very popular topic of Parkinson’s research in 2019 was the influence of the gastrointestinal system. In February, researchers reported that some of the bacteria in our gut secrete a molecule called nitric oxide (NOx). Their research (in worms at least) suggested that this gut derived NOx can spread from the gut to the other areas of the host, where it promotes alterations (S-nitrosylation) of the host proteins, which ultimately affects the activity of those proteins. Given the recent focus on the influence that the gut may be having on Parkinson’s (Click here for a SoPD post on this topic), this research could have important implications if if is found that gut derived NOx can spread from the gut to other areas of larger creatures (like humans – click here to read more about this and click here for the press release).
Two months down, 10 to go:
Several years ago, Joy Milne from Scotland took part in a small pilot study that proved that she could detect differences in the body odour of people with Parkinson’s. Since then, a tremendous amount of research has been conducted on the ‘smell of Parkinson’s’, including the training of dogs to sniff out PD. March saw the publication of some of the research Joy had been involved with on the chemical components of the odour of Parkinson’s (Click here to read more about this and click here to read a SoPD post on the topic).
Melanin is a pigment that the body produces which gives skin and hair their colour. There is also a form of melanin in the human brain, called neuromelanin, which accumulates with age (age being a risk factor for Parkinson’s). In March, researchers reported that they have generatated human-like neuromelanin in rodent dopamine neurons (which do not naturally generate neuromelanin). By doing this, the researchers observed Parkinson’s-like features in the animals overtime (movement issues and neurodegeneration). They also found that boosting the waste disposal system of the cells could rescue the effect (Click here to read more about this and click here to read a SoPD post on this topic)
March also saw research suggesting that a previously “undruggable” protein is in fact “druggable”. NURR1 is a protein that has been shown to be neuroprotective for dopamine neurons in the brain – dopamine neurons being a population of cells badly affected by Parkinson’s. Extensive efforts, however, have failed to identify any compounds that could stimulate NURR1 activity… until March 2019, when researchers from the University of California SF reported that a dopamine metabolite (5,6-dihydroxyindole or just DHI) binds directly to NURR1 and stimulates its activity. DHI is too unstable to be considered a potential therapy for Parkinson’s, but the researchers hope that the findings will suggest avenues for developing synthetic NURR1 stimulators (Click here to read more about this, and click here to read the press release).
In March, researchers reported that they have engineered gut bacteria that continually produce glucagon‐like peptide‐1 (or GLP‐1). This is the protein that the drug Exenatide is based on – Exenatide being the diabetes treatment that has given interesting results in a Phase II clinical trial for Parkinson’s (Click here to read more about this). The researchers report that treatment with the bacteria significantly reduces motor impairments and dopamine cell loss in a model of Parkinson’s (Click here to read more about this).
Researchers also announced in March that they have discovered that 5′-deoxyadenosylcobalamin (AdoCbl) – a physiological form of the essential micronutrient vitamin B12 – modulates Parkinson’s-associated LRRK2 kinase activity (via allosteric regulation) and this activity confers neuroprotection in worm, fly & mouse PD models (Click here to read more about this and click here to read a SoPD post on this topic).
One very cruious report in March involved a post mortem examination of Parkinson’s brains & found a reduction in the number & volume of mitochondria. But here’s the curious twist: deep brain stimulation of subthalamic nucleus reversed the effect (Click here to read more about this and click here to read a SoPD post on the topic).
Roll on:
In April, researchers reported that the L-type calcium channel blocker & anti-hypertensive drug, Felodipine, boosts waste disposal (or autophagy) in mouse brains. It clears mutant Parkinson’s α-synuclein in mouse brains at concentrations similar to those used in humans. The findings raise the possibility that this clinically available drug could be re-purposed for Parkinson’s (Click here to read more about this and click here to read a SoPD post about this).
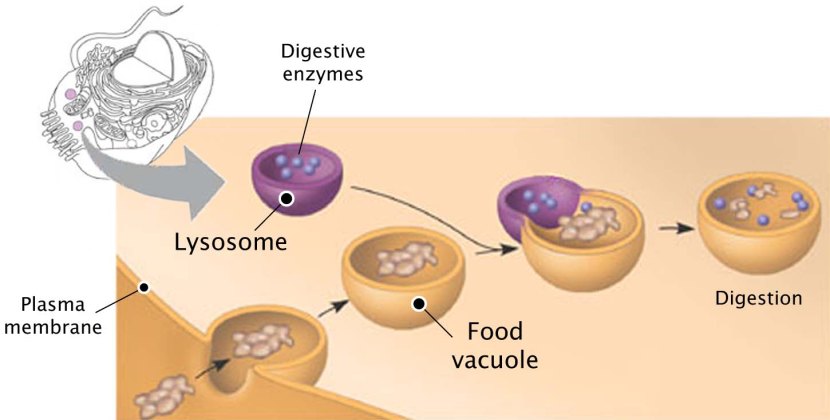
Microglial cells are the resident immune cells in the brain and they play an important role in maintaining homeostasis in the brain, by cleaning up waste (via a process called phagocytosis). As we age, this ability to maintain homeostasis becomes impaired. Genetic mutation screening experiments of age-related modifiers of microglial phagocytosis have identified CD22 (a canonical B cell receptor) as a negative regulator of phagocytosis. Inhibition of CD22 promoted the clearance of Parkinson’s-associated α-synuclein fibrils in mice. Inhibition of CD22 also promoted the clearance of Alzheimer’s-associated amyloid-β oligomers in mice. Long-term delivery of a CD22 antibody appears to reprogram microglia towards a homeostatic transcriptional state & improves cognitive function in aged mice (Click here to read more about this and click here to read a SoPD post on this topic).
In April, researchers presented the discovery of a novel quantum method of detecting & quantifying low levels of dopamine, which could potentially form the basis of better future detection & monitoring of Parkinson’s (Click here to read more about this and click here to read the press release).
Mutations in PARKIN (PARK2) are among the most common genetic risk factors associated with early onset Parkinson’s. In March, researchers reported the development of a framework for the classification of 51 PARKIN variants based on clinical & functional data. Interestingly, only a minority of PARKIN variants, even among those previously associated with Parkinson’s, were found to actually disrupt PARKIN function, and “a few of these naturally occurring PARKIN variants actually enhanced mitophagy” (Click here to read more about this).
Glial cell-line derived neurotrophic factor (or GDNF) has been a topic of heated discussion in the Parkinson’s community for a long time. Most recently due to the announcement of the results of the Phase II Bristol GDNF clinical trial results (mentioned above in February), which did not meet the primary end points of the study. At the annual American Association of Neurological Surgeons conference that was held in San Diego in April, the results of another GDNF clinical trial in Parkinson’s – a gene therapy approach – were presented. The results suggested that the treatment was safe and well tolerated, and the coordinators are now setting up a larger Phase II clinical trial (Click here to read the meeting abstract and click here to read a SoPD post on the results).
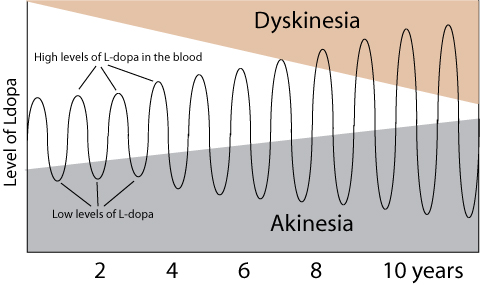
In April, researchers also demonstrated that that genetic silencing of striatal CaV1.3 calcium channels could have the potential to transform the treatment of individuals with Parkinson’s associated Levodopa induced dyskinesias (Click here to read more about this and click here to read a SoPD post on this topic).
And finally in April, the Michael J Fox Foundation & the genetic company 23andMe launched the “Fox Insight Data Exploration Network” (Fox DEN) which contains patient-reported outcomes & de-identified genetic data from the largest cohort in Parkinson’s research & useful data exploration tools (Click here to read more about this).
Spring is in full bloom now with the onset of:
May began with researchers demonstrating the importance of Parkinson’s-associated LRRK2 in lysosomal biology (a novel interaction between LRRK2 & vATPase a1 subunit), and that an anti-fungal treatment called clioquinol could be potentially re-purposed for Parkinson’s (Click here to read more about this).
The results of the STEADY-PD III clinical trial evaluating Isradipine in Parkinson’s were also announced in May. Isradipine is calcium channel blocker that is used in the treatment of hypertension, and the results of this large 3 year study suggest that the drug has no impact on the course of Parkinson’s (Click here to read more about this).
An analysis of the Taiwan National Health Insurance Research Database reported that individuals with bipolar disorder have an increased risk of developing Parkinson’s (HR=6.78, 95% confidence interval 5.74–8.02) (Click here to read more about this and click here to read the press release).
In May, researchers also reported that scalp electroencephalogram (or EEG) measures of beta beta waveform shape over the sensorimotor cortex distinguishes between people with Parkinson’s on & off medication, as well as PwPs off medication from controls. Could this represent a safe, non-invasive biomarker for PD? (Click here to read more about this).
And further fodder for the β2AR agonist/antagonist debate was published in May. An analysis of Danish medical recordes found that β2AR agonist use was associated with reduced Parkinson’s risk (and antagonist use increased PD risk). BUT the association of agonists mediated by smoking. “The association of β2AR antagonists indicates reverse causation, with Parkinson’s symptoms triggering their prescription rather than β2AR antagonists causing PD. Thus, current epidemiologic data do not support a causal link between β2AR agonists & antagonists and PD risk” (Click here to read more about this).
June saw the World Parkinson’s Congress held in Kyoto (Japan) at the start of this month. It was a 4 day meeting that brought together not only patients and researchers, but also Eastern & Western cultures – a truly unique event. Over the course of the conference, there was a continuous stream of presentations, workshops and discussions about all aspects of Parkinson’s.
One of the presentations from the WPC2019 meeting that will stay with me for a long time was given by Dr Linda K Olson – triple amputee & Parkinson’s advocate – an amazing & truly inspiring talk (take a couple of minutes and watch this remarkable woman speak):
It was my first time at the WPC meeting and I felt extremely grateful for the opportunity to attend and take part in the event. So much so that I tried to provide a blow-by-blow account of the presentations and activities of the meeting. Click on the links below for a daily summary:
June also saw the publication of the results from the Phase I glial cell line‐derived neurotrophic factor (or GDNF) gene therapy clinical trial (which I mentioned above in April). The treatment in 13 people with advanced Parkinson’s was safe & well tolerated. Increased [18F]FDOPA uptake (a measure of dopamine activity) in the putamen was also reported (Click here to read more about this and click here to read a SoPD post on the topic).
There were several other clinical trial announcements in June. For example, the biotech firm Collaborative Medicinal Development reported the results of a 24 weeks, Phase I, non-placebo controlled, dose-finding study of treatment with CuATSM in 18 people with Parkinson’s. Cu-ATSM is an orally administered, blood-brain barrier permeable synthetic molecule that contains copper. It has traditionally been used as an imaging agent for hypoxic tissues, but preclinical evidence has been accumulating that it could be useful in the treatment of neurodegenerative conditions. The results of this trial suggest that the treatment is safe & well tolerated. In addition, there were indications that clinical symptoms improved (based on total UPDRS score) and participants reported improvements in quality of life scores. It is important to note that this was an open label trial (there was no control group), but the researchers behind the study are planning a larger trial to properly test the efficacy of the compound (Click here to read more about this).
In another set of clinical trial results, researchers reported that 3 months of N‐acetyl‐cysteine (NAC) significantly increased DAT binding in the caudate & putamen (3.4% to 8.3%) compared to controls & significantly improved Parkinson’s symptoms. This was also an open label study – using a combination of oral & intravenous NAC. 42 participants were involved in the study: 28 in treatment group & 14 standard care (Click here to read more about this and click here to read a SoPD post about this study).
In addition to clinical trial results, June also saw the initiation of some ambitious new clinical studies. On the 4th June, 2019, Prevail Therapeutics announced that the U.S. Food and Drug Administration (FDA) has accepted the company’s Investigational New Drug (IND) application for PR001 (a gene therapy approach for GBA-associated Parkinson’s – click here to read more about this). This news gives the company a green light to “initiate a Phase I/II clinical trial that will investigate the safety and tolerability of PR001, and will also measure key biomarkers and exploratory efficacy endpoints, in patients with PD-GBA. The Company plans to begin dosing patients in the trial this year” (Source).
In June, researchers reported the presence of serotonergic pathology in people with a rare genetic mutation, which makes them vulnerable to Parkinson’s (A53T SNCA). Importantly, these issues were apparent before signs of dopaminergic pathology or motor symptoms. The “findings provide evidence that molecular imaging of serotonin transporters could be used to visualise premotor pathology of Parkinson’s”. It will be interesting to see if this applies to wider Parkinson’s community. If so, serotonin transporter imaging could be a useful tool for screening & monitoring progression for individuals at risk of developing PD or as a marker of PD burden in clinical trials (Click here to read more about this, click here to read a media report on the study).
June saw a string of interesting gut related studies published as well. Researchers reported that microbial L-dopa decarboxylase can be inactivated by (S)-α-fluoromethyltyrosine (AFMT), raising possibilities of developing combinations of Parkinson’s drugs that circumvent microbial inactivation (Click here to read more about this, click here to read the editorial, and click here to read the press release). This new research independently validated and built on the results published in January of this year.
Researchers also reported previously detected difference in gut microbiota between people with Parkinson’s and controls, actually persist after 2 years. There was also some support in the study for decreased abundance of Prevotella bacteria in faster-progressing individuals (Click here to read more about this). And a week after this report was published, an independent grup of researchers published an analysis of two independent conhorts (200 subjects) reveals altered gut microbiota in Parkinson’s. They reported correlations with clinical phenotypes & severity, and altered plasma cytokine profiles associated with gut microbiome composition alterations (Click here to read more about this).
(I have had a guts full of all this gut research)
In further gut related research, an analysis of health claims data from the largest German health insurer of 228 485 individuals (aged >50 years) found that gastrointestinal infections are associated with an increased risk of Parkinson’s (HR=1.42; 95% CI 1.33 to 1.52). “Our results point to the missing link of what may cause alpha-synuclein pathology in the enteric nervous system: bacterial & viral pathogens, which breach the mucosal lining of the gastrointestinal tract (GI) during GI infections, may trigger aggregation of alpha-synuclein in enteric neurons”. I have questions regarding the definitions of GI infections (eg “infectious gastroenteritis & colitis of unspecified origin”?), but interesting result – needing independent validation & further investigation (Click here to read more about this).
(I really can’t stomach anymore of this)
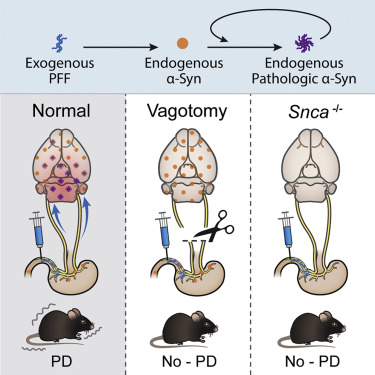
And finally, animal models added further weight to the idea that alpha-synuclein aggregation can spread from the gut to the brain, and possibly play a role in Parkinson’s pathogenesis. Interestingly, vagotomy or the genetic removal of alpha synuclein protected the mice against the development of pathology (Click here to read more about this and click here to read a SoPD post about this research).
And just days after that report was published, an independent group of researchers presented evidence for bi-directional & trans-synaptic parasympathetic & sympathetic propagation of alpha-synuclein in rodent (Click here to read more about this).
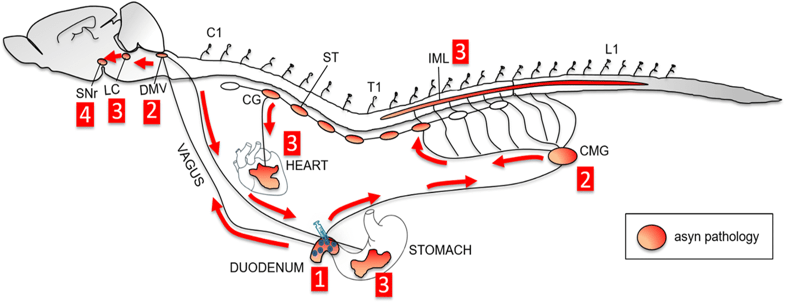 (Apparently “what happens in Vagus” doesn’t stay in Vagus – ok, enough of the silly puns!)
(Apparently “what happens in Vagus” doesn’t stay in Vagus – ok, enough of the silly puns!)
Making way for:
 In July of 2019, researchers demonstrated that when young mice who lack the Parkinson’s-associated PINK1 gene are infected with a particular bacteria, it caused inflammation in the gut, which led to the mice slowly developing Parkinson’s-like symptoms (normal mice recovered fine from the infection). These Parkinson’s feature reduce over time, but the results further add to a growing pile of data suggesting that the gut and environmental factors may be more influential in Parkinson’s than previously thought (Click here to read more about this and click here to read a SoPD post about this topic)
In July of 2019, researchers demonstrated that when young mice who lack the Parkinson’s-associated PINK1 gene are infected with a particular bacteria, it caused inflammation in the gut, which led to the mice slowly developing Parkinson’s-like symptoms (normal mice recovered fine from the infection). These Parkinson’s feature reduce over time, but the results further add to a growing pile of data suggesting that the gut and environmental factors may be more influential in Parkinson’s than previously thought (Click here to read more about this and click here to read a SoPD post about this topic)
July also produced some interesting dyskinesia research. Dyskinesias are involuntary movements mainly associated with the long term use of Levodopa. A new research paper highlights clinical features of early Parkinson’s which are associated with increased risk of developing dyskinesias. The study involved the Oxford Discovery cohort of 734 people with Parkinson’s who were followed for up to 10 years since their diagnosis. Curiously, low mood and anxiety were predictive features of developing dyskinesias (Click here to read more about this).
In addition to dyskinesias, researchers reported using data from longitudinal gait analysis (using wearables) to help predict conversion to Parkinson’s. The TREND study reported that wearable technology assessing gait can discriminate future PD converters from unaffected individuals up to 4 years prior to diagnosis (Click here to read more about this).
In July, researchers also reported using big data in a study assessing the association of marketed medications with risk of parkinsonism. The study used 4 large US claims databases (2181 drugs & 117,015,066 people). Reduced risk of parkinsonism was associated with Armodafinil (56% reduction), Modafinil (54%), Methylphenidate (39%) & the β-agonist Albuterol (17%). Of the β-blockers, only propranolol was associated with increased risk (32% increased risk). “Isradipine was associated with a decreased risk, but the results were heterogeneous & the sample sizes were small. When this heterogeneity was incorporated, the combined estimate showed no statistically significant association between the use of isradipine & parkinsonism” A guide for Parkinson’s drug discovery? ( to read more about this).
In August, the 2019 Linked Clinical Trials (LCT) meeting was held in Grand Rapids (Michigan). This is a major drug repurposing programmed co-ordinated by The Cure Parkinson’s Trust in partnership with the Van Andel Institute. Chair of the LCT committee Prof Patrik Bundin (Van Andel Institute) has provided a video explaining how the LCT initiative is trying to get new potentially disease modifying therapies for Parkinson’s:
The LCT initiative currently has 16 drugs in 17 clinical trials (and is hoping to increase this to 25 over the next 12 months – click here and here to read more about the Linked Clinical Trials process and click here to read a SoPD post about this topic.
In August, we also had the results of the Enterin Phase 2a multicenter, dose-escalating RASMET study. This was a clinical trial to evaluate the safety, tolerability & efficacy of orally administered ENT-01 for the treatment of constipation in Parkinson’s. This was an open label trial, but 80%+ participants achieved the primary efficacy endpoint (with the mean number of complete spontaneous bowel movement per week increasing from 1.2 at baseline to 3.6 during fixed dosing. But systemic absorption was <0.3%, so effect is very local ( to read the results and click here for the press release).
In addition to interesting preclinical research, some preclinical data was also published. MEDI1341 (a α-syn-targetting monoclonal antibody developed by MedImmune, & being clinically tested for Parkinson’s by Astra Zeneca & Takeda Pharmaceuticals) was reported to enter the brain, and bind to monomeric & aggregated forms of α-syn. This therapy is currently in clinical testing and we should see the first results in late 2020 ( to read more about this).
In other preclinical results, data from the new & improved ‘Prosavin’ gene therapy viral vector (OXB-102/AXO-Lenti-PD) in non-uman primates was published in August. The results ‘strongly support’ the ongoing clinical trial in Parkinson’s ( to read more about this and click here to read a SoPD post about this topic).
Moving on to: In September, researchers in the Netherlands published the results of a double-blind, randomised controlled clinical trial (the PARKI-in-Shape study) exploring effectiveness of at home aerobic exercise on Parkinson’s,… and they have a positive result! They report an OFF-state UPDRS motor score between-group difference of 4·2 points (p=0·0020) in favour of aerobic exercise (after 6 months of 30–45 min training 3x per week). Conclusion: At home aerobic exercise by folks with Parkinson‘s can help attenuate OFF-state motor signs ( to read more about this, click here to read the press release, and click here to read a SoPD post on this topic).
In September, researchers in the Netherlands published the results of a double-blind, randomised controlled clinical trial (the PARKI-in-Shape study) exploring effectiveness of at home aerobic exercise on Parkinson’s,… and they have a positive result! They report an OFF-state UPDRS motor score between-group difference of 4·2 points (p=0·0020) in favour of aerobic exercise (after 6 months of 30–45 min training 3x per week). Conclusion: At home aerobic exercise by folks with Parkinson‘s can help attenuate OFF-state motor signs ( to read more about this, click here to read the press release, and click here to read a SoPD post on this topic).
In September, researchers also reported that the protein LRRK2 can alter the course of bacterial and viral infections. Mice with the Parkinson’s–p.G2019S LRRK2 genetic variant showed reduced replication of a bacterial pathogen, but worse outcome to viral infection. Mice carrying the kinase-dead LRRK2 genetic variant, p.D1994S, exhibited improved survival to viral infection, suggesting that the kinase region is involved. Female LRRK2-deficient mice did not control infection as well as males. This is a fascinating study, with implications for not only Parkinson’s, but also leprosy & Crohn’s disease (Click here to read more about this, click here to read the press release and click here to read a SoPD post on this topic).
September also brought news that Chinese and US researchers had discovered that the prostatic hyperplasia and hypertension drug, terazosin may be useful for repurposing for Parkinson’s. Multiple PD models & human medical database evidence were presented in their report. They found that Terazosin activates phosphoglycerate kinase 1 (PGK1), one of the first enzymes in ATP production (the energy that cells run on). Treatment with terazosin rescued multiple neurotoxin & genetic models of Parkinson’s, and human dopamine neurons from LRRK2-G2019S patients that were grown in cell culture. MarketScan medical records database analysis found 2880 Parkinson’s patients with PD taking Terazosin (or similar) “had fewer PD-related diagnoses” than 15,409 individuals with PD + no Terazosin. PD-free individuals on Terazosin had reduced frequency of PD (0.62; P < 0.0001). A Phase II clinical trial for Terazosin in Parkinson’s is already being organised. It is a double blind, placebo controlled,12 weeks treatment in 20 people with PD (1:1 randomisation). Safety is the primary outcome, and it will be reporting in early 2020 ( to read the report, click here to read more about the clinical trial, click here to read the press release, click here for a good overview of this research, and click here to read an SoPD post on this topic).
 In other exciting news in September, researchers identifed Miro1 as a protein accumulating on depolarized (stressed) mitochondria. They found that the failure to effectively remove Miro1 was found in a large proportion of cells collected from both people with Parkinson’s and those at high risk of developing the condition – raising the possibility of a biomarker. But then the researchers conducted a drug screen and identified a small molecule that promotes Miro1 degradation. They found that this drug could rescue mulitple Parkinson’s models. Novel biomarker and treatment? (Click here to read more about this, click here to read the press release and click here to read a SoPD post on this topic).
In other exciting news in September, researchers identifed Miro1 as a protein accumulating on depolarized (stressed) mitochondria. They found that the failure to effectively remove Miro1 was found in a large proportion of cells collected from both people with Parkinson’s and those at high risk of developing the condition – raising the possibility of a biomarker. But then the researchers conducted a drug screen and identified a small molecule that promotes Miro1 degradation. They found that this drug could rescue mulitple Parkinson’s models. Novel biomarker and treatment? (Click here to read more about this, click here to read the press release and click here to read a SoPD post on this topic).
An overview of the Aligning Science Across Parkinson’s (or ASAP) initiative was published in September. This is an exciting new project focused on basic research for Parkinson’s. It is being coordinated by a Nobel Prize winning research scientist and funded by the significant resources of the Sergey Brin (Google founder) family foundation. Expect to hear a lot more about this project going forward (Click here to read more about this and click here to read a SoPD post on this topic).
Autumn is now in full effect with:
In October, researchers reported a new small-molecule modulator of glucocerebrosidase (GCase) called S-181 which improves pathogenic phenotypes in human dopaminergic neuronal models & transgenic GBA mouse models of Parkinson’s. S-181 increased WT GCase activity in iPSC-derived dopaminergic neurons from sporadic PD, as well as carriers of GBA1, LRRK2, DJ-1, & PARKIN variants. S-181 treatment of these PD cells partially restored lysosomal function, lowered oxidized dopamine, glucosylceramide & α-syn. A new company Surmount Bio has been set up to explore & develop S-181 further. (Click here to read more about this and click here to read the press release).
At the same time other researchers reported that stress-induced cellular clearance of waste in cells is mediated by the SNARE Protein ykt6, which is inhibited by Parkinson’s-associated α-synuclein (leading to disruption of the lysosomal stress response). They found that farnesyltransferase inhibitors restores activity and protect the cells (Click here to read more about this and click here to read a SoPD post on the topic)
In October, researchers also reported that a brain-penetrant LRRK2 kinase inhibitor (PF-360) improved endosomal maturation & lysosomal function, but also prevented rotenone-induced neurodegeneration in Parkinson’s models (Click here to read more about this). LRRK2 inhibition is currently being clinically tested in Parkinson’s (Click here to read an old SoPD post on this topic).
More on LRRK2 in October: Researchers reported that PPM1H acts as a key modulator of Parkinson’s-associated LRRK2 signaling. They provide compelling evidence that PPM1H acts to dephosphorylate Rab proteins, which counteracts LRRK2 signalling. “Targeting PPM1H to increase its activity or expression in order to promote Rab protein dephosphorylation could be explored as a therapeutic strategy for preventing &/or treating LRRK2-mediated Parkinson’s” (Click here to read more about this and click here to read the press release).
And yet more on LRRK2 in October: Data from the Michael J Fox Foundation supported PPMI project found evidence of subtle motor & non-motor signs of Parkinson’s in non-manifesting carriers of LRRK2 genetic variants (compared with healthy controls) that can precede DAT deficit. Longitudinal data now essential to confirm these findings (Click here to read more about this).
October also saw the launch of the first research funding round for the Aligning Science Across Parkinson’s (ASAP) initiative, in partnership with Michael J Fox Foundation, which seeks to support multi-disciplinary/-institutional research teams to address key knowledge gaps regarding Parkinson’s (Click here to read more about this and click here to read an SoPD post on this topic).
The Chan Zuckerberg Initiative also launched a new funding opportunity for innovative approaches to fighting neurodegenerative conditions like Parkinson’s (Click here to read more about this).
In November, a manuscript was posted on the OPEN ACCESS preprint database BioRxiv by the biotech firms ionis Pharma & Biogen, which demonstrates antisense oligonucleotides targeting alpha synuclein mRNA reduces the production of the protein and reduces pathology in pre-formed fibril models of Parkinson’s (cells, mice & primates). Could this data be signalling a move by these two companies to clinically test this approach? (click here to read more about this).
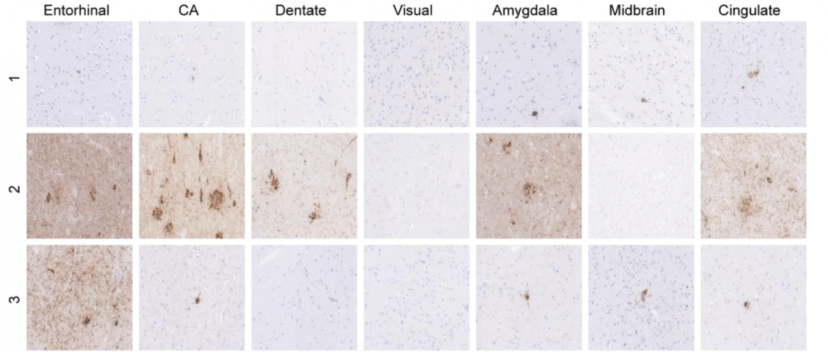
In November, researchers also published a study examining proteins associated with neurodegeneration – α-synuclein, tau, & β amyloid – in 12 postmortem brains from individuals who carried a LRRK2 genetic mutation. They found α-synuclein pathology in 63.6% of cases, but tau pathology in 100% of cases (abundant in 91%). Abundant β amyloid also be found (Click here to read more about this).
Researchers reported in November that that loss of NLRP3 inflammasome function reduces tau hyperphosphorylation & aggregation (by regulating tau kinases & phosphatases) in models of Alzheimer’s – further highlighting this therapeutic target for neurodegenerative conditions. They also found that Tau activates the inflammasome (Click here to read more about this, click here to read the press release and click here for a SoPD post exploring this topic).
In other inflammasome news in November, Korean researchers identify N,N′-diacetyl-p-phenylenediamine (DAPPD) as capable of restoring microglial dysfunction & improving cognitive function in Alzheimer’s mouse models via suppression of NLRP3 (Click here to read more about this and click here to read an interesting discussion)
In November, researchers reported a longitudinal metabolome analysis on 30 drug-naive, de novo Parkinson’s patients (& 30 matched controls) and revealed PD-specific patterns in microbial-host sulfur co-metabolism that may contribute to PD severity. Key results: (1) longitudinal trajectory of certain metabolites differed between Parkinson’s & controls; (2) PD medication showed strong lipidomic signatures; (3) taurine-conjugated bile acids correlated with the severity of motor symptoms, while low levels of sulfated taurolithocholate were associated with Parkinson’s incidence in the general population; & (4) computational modeling predicted changes in sulfur metabolism, driven by A. muciniphila & B. wadsworthia, which is consistent with the changed metabolome (Click here to read more about this).
And finally, in November a homegrown drug for treating Alzheimer’s was approved by China’s National Medical Products Administration. Oligomannate (aka GV-971 – derived from brown algae) demonstrated statistically improved cognitive function among people with AD. Green Valley, a Shanghai-based pharmaceutical company, plans to launch Phase III clinical trials in the US & Europe in early 2020. Little is known about the mechanism of action, but the researchers behind the drug suggest that epidemiological data drew them to it. The Phase 3 Chinese trial was a multicenter, randomized, double-blind, placebo-controlled, parallel-group 36 week study led by Peking Union Hospital & Shanghai Jiaotong University Medical School Mental Health Center (conducted in 34 Tier-1 hospitals across China). A total of 818 patients with mild to moderate Alzheimer’s completed the study. Unpublished results suggest a mean difference between Oligomannate & placebo groups in ADAS-Cog12 Score of 2.54 (p< 0.0001), sustained from first month of treatment to the end of 9 months of treatment (Click here to read the press release and click here to read a previous research report on this drug, and click here to read some…um, questioning commentary).
Which brings us finally to:
December started off with a BANG! with the announcement of the ASAP-supported Global Parkinson’s Genetics Program (GP2). GP2 will be a study that “will engage existing global consortia & cohorts to expand genetic analysis efforts with samples from more than 150K people, including those with Parkinson’s, people at risk for PD, & control volunteers”. “GP2 will use cutting-edge techniques to analyze samples from people around the world living in or with ancestors from Africa, Asia, Europe, & Central+South America. These efforts will transform understanding of the genetic architecture of Parkinson’s across populations”. Importantly, the project will be focusing on “those currently underserved in research”. GP2 is a huge five-year study (Click here to read more about this, click here to read an editorial, and click here to read a previous SoPD post about ASAP).
Researchers also reported precision functional mapping identifies subcortical integration zones in individuals. Integration zones map onto variably effective deep brain stimulation sites. This research if replicated on a larger scale could have major implications for more personalised approaches to deep brain stimulation for Parkinson’s (Click here to read more about this, click here for the press release, and click here to read a SoPD post on this research).
In December there was also a lot of alpha synuclein data – Researchers demonstrated that inoculation of transgenic mice with different strains of recombinant or Parkinson’s brain-derived α-synuclein aggregates produces clinically & pathologically distinct diseases. “Strain-specific clinical, pathological & biochemical differences were faithfully maintained after serial passaging, which implies that α-synuclein propagates via prion-like conformational templating” (Click here to read more about this and click here to read the press release).
Researchers also amplified α-synuclein aggregates from Parkinson’s & Multiple System Atrophy (MSA) brain extracts & analyzed their conformational properties. They found greater structural heterogeneity among α-synuclein fibrils from the PD brains. PMCA used to amplify a-syn aggregates from brain extracts of 10 patients (5 PD & 5 MSA). Amplyfication of samples from 1 control subject did not exhibit any a-syn aggregates. The a-syn fibrils amplified from patient brain extracts were structurally different to ALL of the in vitro polymorphs analyzed. Highlights potential differences between in vitro & in vivo aggregates. Also, a-syn fibrils propagated from Parkinson’s & MSA brains do not exhibit distinctive structural properties. So no to the “one disease-one strain” hypothesis? (Click here to read more about this and click here to read the press release).
In addition, researchers reported a master regulatory mechanism of Parkinson’s-assocaited α-synuclein function & aggregation, extending the repertoire of molecular chaperones & highlighting new therapeutic avenues (Click here to read more about this).
Microglia are the resident immune cells of the brain. In December, researchers published a study in which they conducted single cell analysis of microglia from 10 different species (covering more than 450 million years of evolution). They reported that microglia are only heterogeneous in humans, not other species. They also found that Parkinson’s genes are conserved only in primates (Click here to read more about this).
And finally for 2019, in December the results of the Georgetown Nilotinib clinical trial in Parkinson’s were published. Drug appeared to be reasonably safe. A couple of the exploratory biomarkers were altered in response to Nilotinib. But “no significant differences were seen in motor and nonmotor outcomes between the nilotinib groups and the placebo group”, but the authors point out that “the study was underpowered for this analysis” (Click here to read more about this).
The NILO-PD Steering Committee also announced that although safe & tolerable, Nilotinib does not demonstrate any change in clinical symptoms or biological measures in the Phase II Nilotinib for Parkinson’s (NILO-PD) Trial (Click here to read more about this).
A post will be written on the SoPD about these trials once the NILO-PD results are published, but both studies found very limited amounts of Nilotinib accessed the brain (less than 1% of blood levels), which suggests that this drug may not be a worthy test of c-Abl inhibition in the brain. Other clinical trials using c-Abl inhibitors specifically designed for neurological conditions (such as SPARC’s K-0706 and Inhibikase’s IkT-148009) will be better tests of this approach.
# # # # # # #
These were some of the highlights for Parkinson’s research in 2019 (according to yours truly). In the next post, we will look at where things are heading in 2020.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE – The author of this post is an employee of the Cure Parkinson’s Trust. The Trust is supporting many of the clinical trials mentioned in this post (including the Exenatide study (including the blood-derived exosome study), the Bristol GDNF study, the UDCA study, the Nilotinib study, the Australian Parkinson’s Mission, and the Linked Clinical Trials initiative). The Trust has not requested the production of this post, but the author considered the information interesting and important to share with the Parkinson’s community.
Many of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with any of the companies or associated parties. This post has been produced for educational purposes only.
In addition, the information provided by the SoPD website is for educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from CNTraveller























































Thanks as always Simon.
LikeLike
Simon,nothing on cell replacement?
LikeLike
Hi Deepak, thanks for your comment. Nothing on cell replacement research made me say ‘wow’ in 2019 (happy to be corrected on this if anyone is aware of interesting work). We will, however, be looking at cell transplantation trials in the next post on what is ahead for 2020. See next post.
Kind regards,
Simon
LikeLike
Thanks Simon. Can your next blog post throw light on what we can expect to see in the gene therapy space in 2020, considering that Voyager AADC has RMAT designation? Also what can we expect to see from an antibody / immunotherapy standpoint . There has been no news of Affiris.
Also, this post claims that a phase 2 is underway for terazosin. As far as I know it is only phase one. Can you elucidate?
LikeLike
Hi Deepak,
The next post will briefly deal with some of the gene therapy trials, but I am primarily focusing on the disease modification trials as a review of the broader Parkinson’s research landscape is beyond possible (there is a great deal to cover). Immunotherapy gets a good mention (as many of the Phase I trials are scheduled to complete in 2020), but I have heard zip out of Affiris (nothing since 2018).
Regarding Terazosin – Quoting from the Clinicaltrials.gov website for the TZ-PD trial:
“The TZ-PD trial will be a 1:1 (active:placebo) randomized, double-blind, placebo-controlled Phase II trial to evaluate the safety and tolerability of terazosin for the treatment of PD” (https://clinicaltrials.gov/ct2/show/NCT03905811). They mention a Phase I component, but it is listed as Phase II. Let’s call it a Phase IIa trial (safety/tolerability are the primary endpoints).
Kind regards,
Simon
LikeLike
Thanks, Simon. Terazosin retrospectively has evidence, waiting for efficacy in a double blind trial.
Inhibikase,Sparc k0706 and First Biotherapeutics’ 1ST-102 (https://clinicaltrials.gov/ct2/show/NCT04165837?term=1st-102&draw=2&rank=1 ) are the Abelson kinase inhibitors in trials.
Just a minor correction to your statement regarding immunotherapy. Both prothena and Biogen are in phase 2
LikeLike
Hi Deepak,
Thanks for this. Yes, Prothena/Roche and Biogen are both Phase II (and scheduled to complete in 2021), but I was referring to Astrazeneca (MEDI1341) and Lundbeck (Lu AF82422) which are both scheduled to complete Phase I testing in 2020.
One question: Is Inhibikase’s IkT-148009 in trial? The company filed their IND for IkT-148009 in February 2019 (https://www.globenewswire.com/news-release/2019/02/26/1742368/0/en/Inhibikase-Therapeutics-Files-Two-Investigational-New-Drug-Applications-for-IkT-148009-a-Disease-Modifying-Therapy-for-Parkinson-s-Disease.html), but I have not been able to find anything regarding a subsequent trial on any trial registries. Are you aware of some news?
Kind regards,
Simon
LikeLike
Hi Simon,
Yes, I heard from Santa that a phase 1 safety study is underway in adult healthy volunteers for IKT 148009. There will be an interventional phase two in 2020 perhaps. That’s as much as I know.
They did not publicize their recruitment for the phase one for some reason but say that the results will be well publicized. These tyrosine kinase inhibitors are the most promising strategy from the standpoint of stopping the disease in its tracks.
Santa also tells me that axovant’s strategy of coding for dopamine production endogenously is a little risky because it may lead to dyskinesias. However from what I read they are only attempting to manage the off time with a tonic level of striatal dopamine. They announced on Facebook that data from their p2 will be made public in the first half of 2020.
My Santa is a reliable source
LikeLike
LikeLike
https://youtu.be/P78NdikEtQE. Fujifilm Cellular dynamics Have launched their GMP facility for manufacturing dopamine neurons among other things
LikeLike
Simon,
Is there retrospective evidence of exenatide halting/slowing PD in diabetics using the drug? Something like Terazosin when historical data suggested that BPH patients using the drug had a significant slowing down of the disease compared to people taking another category of drugs
LikeLike
Hi Deepak,
There is at least one group that I am aware of doing such work. Not sure what the results are, but hope to see them later this year.
Kind regards,
Simon
LikeLike