|
Deep brain stimulation (or DBS) represents a well established treatment option for individuals with Parkinson’s who no longer respond to standard therapies. It involves tiny electrodes being embedded in the brain and they modulate populations of neurons that have become dysfunctional. The results of the DBS procedure can be “miraculous” for some individuals – reducing tremors and significantly improving quality of life. In up 20% of cases, however, the procedure may have little or no effect. Placement of the electrodes has been blamed for the lack of DBS response in many of these situations. But very recently researchers have discovered a new method that may aid in the better placement of electrodes. In today’s post, we will discuss what DBS is, review the new research, and explore the implications of it.
|
 Ray Kroc. Source: Medium
Ray Kroc. Source: Medium
It is said that Ray Kroc – the American fast-food tycoon, who purchased the ‘McDonalds’ company from the McDonald brothers in 1961 for US$2.7 million – once gave a lecture to Harvard MBA students.
At some point during his talk, Mr Kroc asked the students: “What business is McDonalds in?”
You can imagine all the different answers that probably came back: “Food, yeah hamburgers. Right?” “Restaurants!”, “Entertainment“, “Hospitality?”
 Source: Youtube
Source: Youtube
Mr Kroc simply laughed and said “No”
“Ladies and gentlemen, I’m not in the hamburger business. My business is real estate”
In other words: knowing (and owning) the right locations.
He proceeded to tell the students that big fast food corporations (like McDonalds, Burger King, Subways, Starbucks) spend much of their capital on identifying and buying new locations where they think there will be the opportunity for growth.
I think I’ve got the wrong blog. What on Earth does this have to do with Parkinson’s?
Identifying the right location is very applicable to Parkinson’s when it comes to deep brain stimulation.
What is deep brain stimulation?
Deep brain stimulation (or DBS) is a treatment method that involves embedding electrodes into the brain to help modulate the brain activity involved in movement.
First introduced in 1987, deep brain stimulation consists of three components: the pulse generator, an extension wire, and the leads (which the electrodes are attached to). All of these components are implanted inside the body. The system is turned on, programmed and turned off remotely.
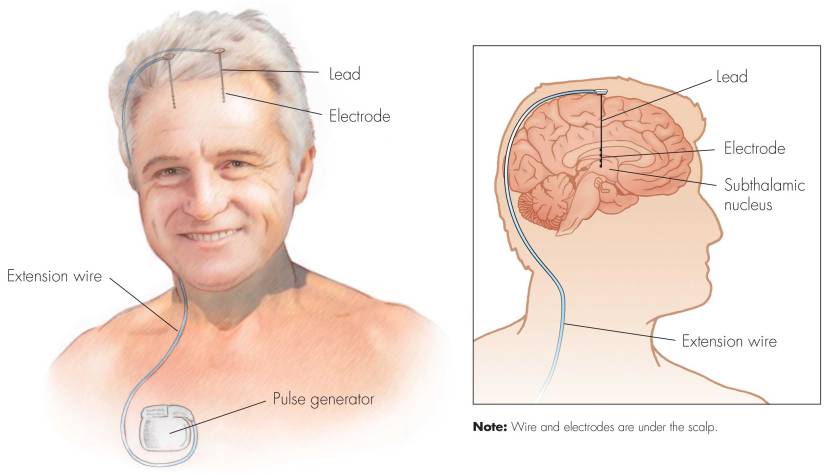
Source: Ucdmc
The electrodes that are implanted deep in the brain are tiny, and the very tip of them has small metal plates (each less than a mm in width) which provide the pulses that will help mediate the activity in the brain.

DBS electrode tip. Source: Oxford
The electrode extends up into the leads (or extension wire) which continue up and out of the brain, across the top of the skull, down the neck and to the pulse generator which is generally located on the chest.

Xray image demonstrating the leads. Source: Fineartamerica
There are many different brands/types of pulse generators, but they all largely do the same basic function. They are titanium packets containing the electronics and power supply for the leads and electrodes. They are implanted in a subcutaneous spot, usually located under the collar bone on the chest.

Pulse generators. Source: Cambridge (also a good read on DBS)
The pulse generator is programmable (which the doctor usually does several weeks post surgery). A small hand held device is used to turn on/off the generator, generally by holding the device over it.

Remote control. Source: Cambridge
For more information on the topic, click here to read about a case report of deep brain stimulation implantation.
Here is a video – kindly provided by fellow kiwi Andrew Johnson – that demonstrates the benefits of DBS:
|
RECAP #1: Deep brain stimulation involves electrodes being implanted into the brain. A pulse generator sends signals down those electrodes which allows individuals with Parkinson’s to ‘tune’ their treatment and function better.
|
So how does deep brain stimulation actually work?
I have previously discussed how we initiate movement (Click here to read that post), and I will only briefly summarise the basics here.
Movement is largely controlled by the activity in a specific group of brain regions, collectively known as the ‘Basal ganglia‘.
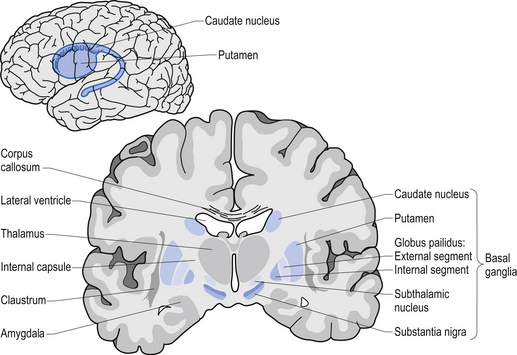
The basal ganglia structures (blue) in the human brain.
Source: iKnowledge
The basal ganglia receives signals from the overlying cerebral cortex regarding possible movements to make, and it processes that information before sending a signal on to another important participant in the regulation of movement: the thalamus.

A brain scan illustrating the location of the thalamus in the
human brain. Source: Wikipedia
The thalamus is a structure deep inside the brain that acts like the central control unit of the brain. Everything coming into the brain from the spinal cord, passes through the thalamus. And everything leaving the brain, passes through the thalamus. It is aware of most everything that is going on and it plays an important role in the regulation of movement. Importantly it is biased towards inhibiting movement – a signal from the basal ganglia has to be pretty strong, for the thalamus to give the green light and a particular movement can then be made.
Under normal circumstances, dopamine producing neurons release dopamine in the basal ganglia where it helps to mediate the local environment. It acts as a kind of lubricant for movement, the oil in the machine if you like. It helps to reduce the inhibitory bias of the basal ganglia.
Thus, with the loss of dopamine neurons in Parkinson’s, there is an increased amount of inhibitory activity. And as a result, the thalamus is kept in an overly inhibited state. And this is the reason why people with Parkinson’s have trouble initiating movement.

The workflow of the movement decision making process. Source: BJP
Now, as you can see from the image above, the globus pallidus is one of the main conduits of information into the thalamus. Given this pivotal position in the regulation of movement, the globus pallidus has been a region of major research focus for a long time.
It is also one of the sites targeted in deep brain stimulation therapy for Parkinson’s. Another target of deep brain stimulation therapy is the subthalamic nucleus which help to regulate the activity of the globus pallidus. By placing electrodes in one of these two brain regions (see image below), doctors can regulate the signal being passed from the basal ganglia to the thalamus and thus provide people with Parkinson’s relief from their motor-associated symptoms.

Two of the target sites for deep brain stimulation. Source: JAMA
But what do the electrodes do?
So as mentioned above there are two target populations of DBS for Parkinson’s: the subthalamic nucleus and the globus pallidus.
The subthalamic nucleus is a very small region of the brain (140 cubic mm), consisting of approximately 300,000 neurons – making up about 0.02% of the total volume of the brain.
In Parkinson’s, the subthalamic nucleus becomes very excited, and the cells in the region start firing at an increased rate. This over activation of the region results in dysfunction in other parts of the basal ganglia – ultimately disrupting normal movement.
The electrodes that are placed in the subthalamic nucleus during deep brain stimulation will be programmed to release tiny electrical pulses that will help regulate the firing of the surrounding cells. By modulating the firing of those cells, deep brain stimulation reduces the inhibition in the basal ganglia circuit overall, which allows individuals with Parkinson’s to move more freely.
|
RECAP #2: Deep brain stimulation allows for modulation of neuronal activity in certain regions of the brain – such as the globus pallidus and the subthalamic nucleus – which are dysfunctional in Parkinson’s. By regulating the activity of these regions, the movement-controlling parts of the brain are less inhibited, allowing affected individuals to move more freely.
|
How successful is this procedure?
DBS has sometimes been referred to as ‘the second honeymoon’ in the treatment of Parkinson’s (with L-dopa treatment being the first), but it is important to understand that while the procedure improves quality of life for people with Parkinson’s, it does not slow the progression of the condition. It can, however, significantly improve quality of life for those affected by tremor or dyskinesia when it is successful.
What do you mean “when it is successful”?
About 15% of cases are associated with limited response to the DBS treatment (Click here to read more about this).
What could cause limited response?
One of the primary reasons for no response is believed to be issues with the placement of the electrodes. Tiny variations in the networks of the brain between individuals might make a big difference in whether an electrode being implanted in a specific spot is going to have beneficial effects.
Is there any way we can improve this situation?
Hopefully yes.
Very recently, this interesting research report was published:
 Title: Integrative and Network-Specific Connectivity of the Basal Ganglia and Thalamus Defined in Individuals
Title: Integrative and Network-Specific Connectivity of the Basal Ganglia and Thalamus Defined in Individuals
Authors: Greene DJ, Marek S, Gordon EM, Siegel JS, Gratton C, Laumann TO, Gilmore AW, Berg JJ, Nguyen AL, Dierker D, Van AN, Ortega M, Newbold DJ, Hampton JM, Nielsen AN, McDermott KB, Roland JL, Norris SA, Nelson SM, Snyder AZ, Schlaggar BL, Petersen SE, Dosenbach NUF
Journal: Neuron, 2019 Dec 5. [Epub ahead of print]
PMID: 31836321
In this study, the researchers wanted to investigate the functional organisation of basal ganglia and thalamus between individuals. That is to say, they were interested in the degree of variability between individuals regarding specific functional network organisation in these two brain regions. How much difference is there between you and I?
To do this, they used data from 10 highly sampled individuals from the Midnight Scan Club dataset.
The “Midnight Scan Club”?
Yeah, that caught my attention as well.
This study was published two years ago:
 Title: Precision Functional Mapping of Individual Human Brains.
Title: Precision Functional Mapping of Individual Human Brains.
Authors: Gordon EM, Laumann TO, Gilmore AW, Newbold DJ, Greene DJ, Berg JJ, Ortega M, Hoyt-Drazen C, Gratton C, Sun H, Hampton JM, Coalson RS, Nguyen AL, McDermott KB, Shimony JS, Snyder AZ, Schlaggar BL, Petersen SE, Nelson SM, Dosenbach NUF.
Journal: Neuron. 2017 Aug 16;95(4):791-807.
PMID: 28757305 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers assembled an extremely extensive MRI dataset from 10 well characterised individuals. The extensive MRI dataset included five hours of resting-state functional connectivity MRI data, six hours of task functional MRI, multiple structural MRIs, and then (on top of all that) there was a battery of neuropsychological tests.
Sounds like a hoot, right?
And to standardise all of the data across the 10 individuals (and reduce the cost of data acquisition), all of the scanning sessions started at midnight.
Hence the name the “Midnight Scan Club” dataset.
Oh, and all of the data set is freely available/open access (Click here to read more about this).
Interesting. Quick question: What is MRI?
Magnetic resonance imaging (or MRI) is a method of scanning tissue that uses powerful magnetic fields and radio waves to produce very detailed images of the interior of the sample being imaged.
 MRI machine. Source: Medicalnewstoday
MRI machine. Source: Medicalnewstoday
Hospitals use MRI to visualise the internal organs of patients in their care.
Yes, but how does it actually work?
So you know how most of your body is made up of water? Well those molecules of water consist of hydrogen and oxygen atoms. At the very centre of each hydrogen atom is a very small particle called a proton.
Protons are sensitive to magnetic fields. They act like tiny magnets and align themselves to magnetic fields – if you lie under a powerful magnet, most of the protons in your body will line up – in the same direction – in alignment with the field being produced by the magnet. Similar to the way a magnet can pull the needle of a compass.
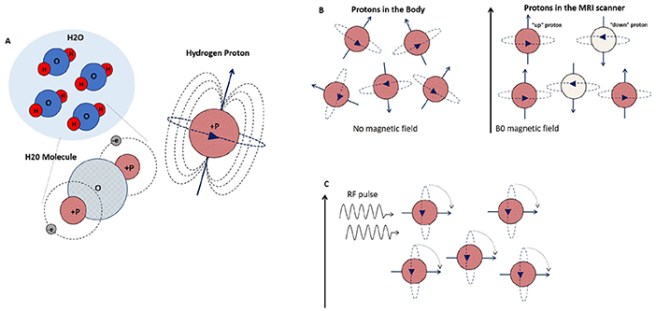 Source: Frontiers
Source: Frontiers
Now, short bursts of radio waves can be fired at the body while you are lying under that magnet and this knocks the protons out of alignment with the magnetic field. But when those radio waves stop, the protons immediately realign, and when they do this, they release energy, which can be picked up by receivers.
The signals from this energy release provide information about the exact location of the protons in the body, providing very detailed images of the internals of our bodies:
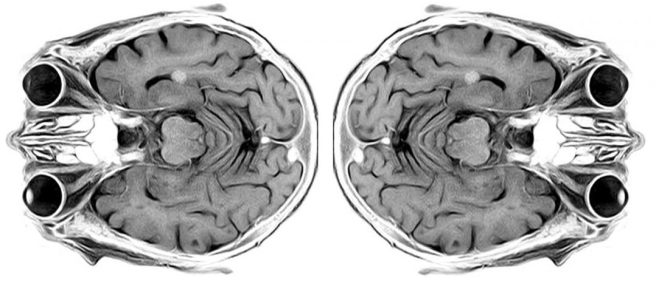 Source: Medium
Source: Medium
This video helps to explain the science behind MRI:
|
RECAP #3: Magnetic resonance imaging (or MRI) is a method of scanning tissue that uses powerful magnetic fields and radio waves to produce very detailed images of the interior of the sample being imaged. It can be used to identify structures in the brain that can be targetted for deep brain stimulation.
|
And what is functional MRI?
fMRI is an imaging method that measures brain activity by detecting changes associated with blood flow.
How..?
Our bodies use haemoglobin in red blood cells to deliver oxygen to neurons. When neuronal activity increases, there is a subsequent increase in demand for oxygen, which results in an increase in blood flow to regions of increased neural activity.
Haemoglobin has some interesting properties. For example, when oxygenated, it is diamagnetic (or repelled by a magnetic field), but when deoxygenated, it becomes paramagnetic (attracted by a magnetic field). These differences in magnetic properties allow for small differences in the MR signal to be deteched, which means that levels of neural activity can be determined based on blood flow.
The image below demonstrates the kind of images that can be generated using fMRI (in this case showing altered cerebellar connectivity in Parkinson’s patients ON and OFF L-DOPA medication):
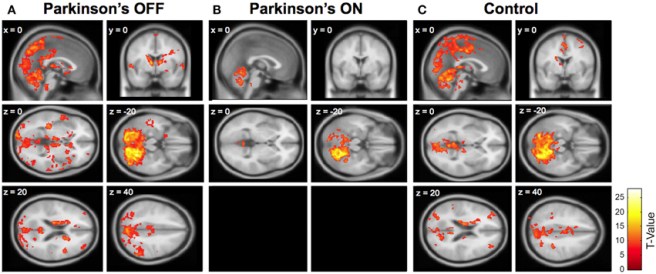 Source: Frontiers
Source: Frontiers
For those interested, this video further explains the science behind fMRI:
|
RECAP #4: Functional MRI allows for the measurement of brain activity by detecting changes associated with blood flow. When neurons become active they require more oxygen, which results in greater blood flow. This imaging method lets researchers identify functional networks in the brain.
|
So when the researchers say that they collected “five hours of resting-state functional connectivity MRI data, six hours of task functional MRI, and then multiple structural MRIs”, this means that they measured changes in blood flow during specific activities (task functional MRI) as well as during no activity periods (resting-state functional connectivity MRI). And they could overlay that on normal MRI images providing the physical structure of the brain (multiple structural MRIs).
And using all of this data from the 10 members of the “Midnight Scan Club”, the researchers sought to assess the functional organisation of basal ganglia and thalamus between these individuals.
Which brings us nicely back to the research report we are reviewing today:
 Title: Integrative and Network-Specific Connectivity of the Basal Ganglia and Thalamus Defined in Individuals
Title: Integrative and Network-Specific Connectivity of the Basal Ganglia and Thalamus Defined in Individuals
Authors: Greene DJ, Marek S, Gordon EM, Siegel JS, Gratton C, Laumann TO, Gilmore AW, Berg JJ, Nguyen AL, Dierker D, Van AN, Ortega M, Newbold DJ, Hampton JM, Nielsen AN, McDermott KB, Roland JL, Norris SA, Nelson SM, Snyder AZ, Schlaggar BL, Petersen SE, Dosenbach NUF
Journal: Neuron, 2019 Dec 5. [Epub ahead of print]
PMID: 31836321
In this study, the researchers wanted to investigate the functional organisation of basal ganglia and thalamus between individuals, so they collected data from the “Midnight Scan Club” dataset and created 3D maps color-coded by functional network for each of the 10 individual. In doing this, the investigators mapped nine functional networks in the deep-brain structures, the basal ganglia and the thalamus.
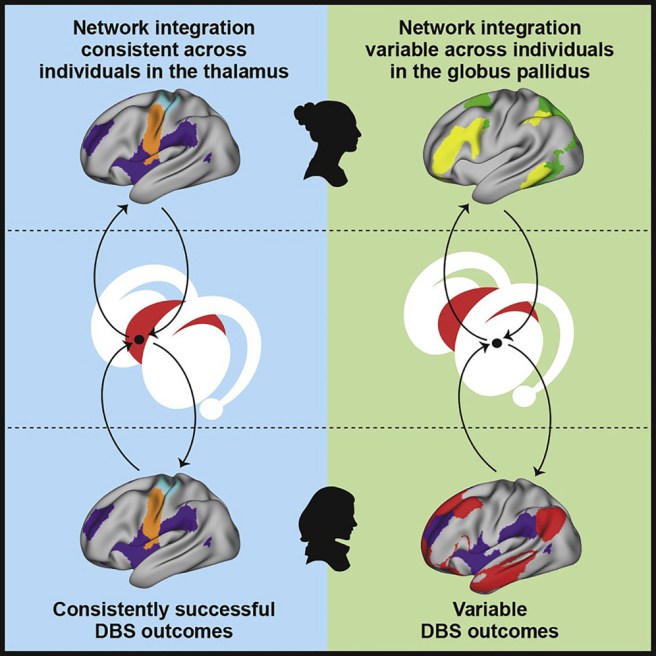
One of those functional networks was devoted to vision, three involve goal-directed behaviors, two are associated with movement, two relate to paying attention, and the final network was active when the brain is at rest. Each of these functional networks was found to blaze its own path through the deeper structures of the brain, but they would intermingle with the other networks at specific spots, called ‘integration zones’.
Some of these integration zone were located in very similar positions in all of the 10 individuals analysed, where a movement and a goal-directed network interact. The integration zone of other networks, however, varied from person to person.
Remarkably, the motor integration zones corresponded to consistently successful sites of deep brain stimulation, such as subregions of the thalamus which are associated with up to 80% tremor reductions in individuals with essential tremor (a non-progressive condition characterised by tremor).
 Source: Sciencedirect
Source: Sciencedirect
The researchers also noted that individually variable integration zones corresponded to sites associated with less successful deep brain stimulation treatment, such as the the globus pallidus when treating dystonia (only 30%–50% symptom improvement in most patients).
The investigators concluded that their data highlights the need for “precision functional mapping” before initiating any deep brain stimulation procedure, in the hope that the indentification of individualised integration zones will lead to better outcomes for DBS treatment.
So what does it all mean?
Deep brain stimulation (or DBS) has become a standardised treatment approach for individuals with Parkinson’s who no longer respond to dopaminergic therapies (such as Levodopa). More than 150,000 individuals world-wide have undergone the procedure. Considerable efforts are being made by the Parkinson’s research community to improve outcomes of deep brain stimulation therapy.
Personalising the approach will hopefully lead to better addressing individual needs and improving quality of life. The research report reviewed in today’s post suggests that identifying individual differences in functional networks in the brain may play an important role in the outcomes.
As opposed to using a “one-size-fits-all” anatomical map, the development of personalised functional maps could lead to better placement of electrodes in the exact place that would provide each patient with the most benefit. Independent replication of this data across larger cohorts will be required, but once validated this finding represents a process that could be rapidly integrated into the standard protocol for the deep brain stimulation procedure.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like!
The banner for today’s post was sourced from Creative7designs

2 thoughts on “DBS: location, location, location”