|
It feels as though novel potential therapies for Parkinson’s are being proposed with an ever increasing frequency. And just when I think there must be few other ways of attacking the condition, a new method is proposed. Recently a biotech firm called Clene Nanomedicine presented data on one such new approach. The experimental treatment is called CNM-Au8 and it involves gold. Yes, that gold. And the treatment is already being tested in a clinical trial for Parkinson’s. In today’s post, we will look at what CNM-Au8 is and what it does, we’ll discuss what data has been presented, and then we’ll outline what the clinical trial involves.
|
 Source: SFN
Source: SFN
Although I did not attend the Society for Neuroscience 2019 annual meeting in Chicago in October, I have still had a look at some of the 816 abstracts which had the keyword “Parkinson’s” attached to them (my Saturday night entertainment – sad I know!).
Those abstracts can be found online (Click here to search those abstracts).
One in particular abstract caught my attention:
 This poster was presented by research scientists from a biotech company that I had never heard of called Clene Nanomedicine:
This poster was presented by research scientists from a biotech company that I had never heard of called Clene Nanomedicine:
And the data presented focused on a novel therapy that I had never heard of which is now being targetted at Parkinson’s.
The new treatment is called CNM-Au8.
What is CNM-Au8?
CNM-Au8 is a suspension of clean-surfaced, faceted nanocrystalline gold with unique nanocatalytic properties.
….um, right…. ok…. thanks for clearing that up…. just one question: what does ANY of that actually mean?!?!?
Let’s start with the easy part of the sentence: Gold
 The shiny stuff. Source: Verdict
The shiny stuff. Source: Verdict
Everyone is familiar with gold – it’s the stuff we dig up out of the ground and then bury in a treasure chest or a bank vault as a store of value/wealth. It’s the stuff that we worship, treasure and pass on in the form of jewellery. It is what atheletes spend their lives seeking.
Interesting facts about gold: There is approximately 190,000 tonnes of gold currently in use/storage. 50% of it is used in jewelry, 40% in investments, and 10% in industry (Source).
It is also a truly remarkable element, as it is:
- resistant to most acids
- high malleability (the ability of a material to bend or change shape)
- ductility (the ability of a material to stretch thin)
- resistance to corrosion
- conductivity of electricity
- chemically inert
All of this applies, however, to gold in a bulk state – that is, large chunks/nuggets/bars of gold.
 Shiny and chemically stable. Source: Livescience
Shiny and chemically stable. Source: Livescience
A fascinating feature of gold is that on the nanoscale level, gold is actually highly catalytic – meaning that it can increase the speed of a chemical reaction. While large blocks of gold are inert and very stable (they do not corrode), at the nanoscale level gold becomes very reactive.
And given this particular property, gold nanoparticles are very useful.
In effect, gold nanoparticles can act as electron reservoirs with a large capacity to donate or receive electrons. This means that they can act as redox catalysts. And yes, I know what you are going to ask next:
What are a redox catalysts?
A redox catalyst is a compound that can change the oxidation state of other molecules.
What is oxidation?
Oxidation is the loss of electrons from a molecule, which in turn destabilises that particular molecule. It is a process that is going on all around us – even within us.
Iron rusting is the example that is usually used to explain oxidation. Rust is the oxidation of iron – in the presence of oxygen and water, iron molecules will lose electrons over time. And given enough time, this results in the complete break down of objects made of iron.

Rusting iron. Source: Thoughtco
The combustion process of fire is another example, albeit a very rapid form of oxidation.
Oxidation is one half of a process called Redox – the other half being reduction (which involves the gaining of electrons).
 The redox process. Source: Academic
The redox process. Source: Academic
Here is a video that explains the redox process:
It is important to understand, that oxidation also occurs in biology.
Molecules in your body go through the same process of losing electrons and becoming unstable. This chemical reaction leads to the production of what we call free radicals, which can then go on to damage cells. When too many free radicals are produced, it can cause oxidative stress in a cell.
What is a free radical?
A free radical is an unstable molecule – unstable because it is missing one or more electrons. They react quickly with other molecules, trying to capture the needed electron to re-gain stability. Free radicals will literally attack the nearest stable molecule, stealing an electron. This leads to the “attacked” molecule becoming a free radical itself, and thus a chain reaction is started – leading to oxidative stress inside of living cell, which can cause terrible damage, ultimately killing the cell if left unchecked.
Antioxidants are the good guys in this situation. They are molecules that neutralise free radicals by donating one of their own electrons. The antioxidant don’t become free radicals by donating an electron because by their very nature they are stable with or without that extra electron.

How free radicals and antioxidants work. Source: h2miraclewater
Redox catalysts like gold nanoparticles can be considered as having antioxidant properties, and this is why they are being explored in medicine.
|
RECAP #1: (Ooooh, new feature based on reader feedback) Large chunks of gold are chemically inert and do not corrode, whereas nanoparticles of gold are very reactive and have antioxidant properties. This makes nanoparticles of very useful.
|
If nothing I have written above makes any sense, this short video will help to explain the idea of why gold is such an interesting element:
But what exactly is CNM-Au8?
CNM-Au8 is a suspension of these gold nanoparticles.
The particles are ~13nm average diameter, and they can act as catalysts for biological reactions.
Below is an electron microscopy image of a single gold nanoparticle (or nanocrystal – Source). CNM-Au8 is a solution of these little guys:
 Source: BioRxiv
Source: BioRxiv
Given its unique properties, CNM-Au8 can help to catalyse biological reactions – meaning that it helps to increase the speed of a chemical reaction inside of cells. And this is particularly interesting with regards to the conversion of nicotinamide adenine dinucleotide (NADH) to its oxidized form (NAD+).
Que?
Which part?
The nico…tin..amide whatever part.
Nicotinamide adenine dinucleotide (or NAD) is a very important protein as it plays a critical role in a wide range of cellular reactions (we have discussed NAD in previous SoPD posts – click here to read more about this topic).
Understand two things about NAD:
- It exists in two forms: an oxidized form (NAD+) and non-oxidised (or “reduced”) form NADH.
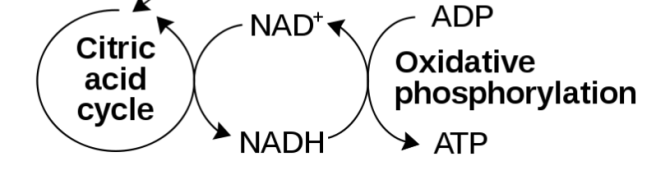
- NAD is a required co-factor in the production of Adenosine Triphosphate (or ATP)
ATP is the fuel which cells run on, and it is generated using NADH. Without NADH, the production of ATP starts to go wrong very quickly.
 Source: Wikipedia
Source: Wikipedia
NADH is present in every cell and it is essential for normal functioning. NADH is involved with converting adenosine diphosphate (or ADP) into ATP. In the process, NADH becomes NAD+ and goes looking for a new hydrogen ion (the H in NADH). Once found, NAD is recycled and can be used again.
So how does CNM-Au8 catalyse the production of ATP?
CNM-Au8 was designed to increase the conversion of NADH to NAD+, which in turn results in a greater production of ATP.
In addition to this, CNM-Au8 also has its antioxidant properties, which may help to protect cells against oxidative stress.
|
RECAP #2: Nanoparticles of gold can help boost the production of ATP in cells. ATP is the energy that cells function on. In addition, gold nanoparticles have anti-oxidant properties.
|
Interesting. Has CNM-Au8 ever been tested in any models of neurodegeneration before?
Yes, although that research has not been published in a peer-reviewed journal yet. The only data related to CNM-Au8 that is currently available (and I’m happy to be corrected on this) is a single manuscript on the preprint website BioRxiv.
This is the manuscript:
 Title: Nanocatalytic activity of clean-surfaced, faceted nanocrystalline gold enhances remyelination in animal models of multiple sclerosis
Title: Nanocatalytic activity of clean-surfaced, faceted nanocrystalline gold enhances remyelination in animal models of multiple sclerosis
Authors: Robinson AP, Zhang JZ, Titus HE, Karl M, Merzliakov M, Dorfman AR, Karlik S, Stewart MG, Watt RK, Facer BD, Facer JD, Christian ND, Ho KS, Hotchkin MT, Mortenson MG, Miller RH, Miller SD
Database: BioRxiv
DOI: https://doi.org/10.1101/712919 (This manuscript is OPEN ACCESS if you would like to read it)
In this study, the researchers treated a mouse model of Multiple Sclerosis with CNM-Au8. Multiple Sclerosis is a neurodegenerative condition associated with inflammation and demyelination of neurons (myelin being a protective coating for the branches of neurons, which helps neurons to communicate).
The researchers found that the gold nanoparticles treatment improved the motor functions of Multiple Sclerosis mice. They also reported that when they treated oligodendrocytes – the cells that produce myeline in the brain – with CNM-Au8, the maturation and functioning of the cells improved. In addition, increased levels of NAD+ and elevated levels of ATP were recorded in the cells.
Based on these findings, the investigators suggested that CNM-Au8 could “represent a novel remyelinating therapeutic for multiple sclerosis”.
Has CNM-Au8 ever been tested in models of Parkinson’s?
So this is what the Clene Nanomedicine researchers presented at the Society for Neuroscience meeting in Chicago in October (the abstract that I mentioned in the intro). They presented the results of a preclinical study investigating CNM-Au8 in models of Parkinson’s.
The researchers grew rodent dopamine neurons in culture and they found that treatment with CNM-Au8 significantly increased the cellular levels of NAD+. Dopamine neurons are a population of cells in the brain that are severely affected in Parkinson’s.
Next the investigators repeated the experiment, but this time they exposed the dopamine neurons were subjected to the neurotoxins (either MPP+ or 6-OHDA), and they found that CNM-Au8 treatment significantly preserved the branches (or neurites) of the cells and enhanced the overall survival of the neurons.
After these cell culture experiments, the researchers shifted their attention to animal models. The investigators exposed rodents with a neurotoxin (6-OHDA), and then treated the animals daily with either CNM-Au8 or a placebo treatment. The animals were divided into different groups to assess different treatment regimes. In one group of animals, the researchers started the treatment the day after the neurotoxin was delivered (Day 1), and for a second group of animals they waited 2 weeks before starting treatment (Day 14 – a delayed initiation of treatment, which is more relevant to the clinical condition).
Remarkably both the early (Day 1+) and delayed (Day 14+) treatment with CNM-Au8 resulted in better behavioural motor function in the rats at 6 week post neurotoxin (vs placebo treated animals). And this effect was superior to L-Dopa/Carbidopa treatment on one particular test. There is no mention in the meeting abstract regarding neuroprotection of the dopamine neurons in these animals – I am assuming (based on personal experience) that the abstract was probably submitted before that analysis was conducted and analysed. The researchers may have had the data available at the time of presenting their poster in Chicago, but I can only speculate here as I am publishing this post before contacting the company. For those interested, click here to read the meeting abstract.
|
RECAP #3: Nanoparticles demonstrate beneficial/neuroprotective properties in models of neurodegeneration. CNM-Au8 can boost the production of ATP in brain cells and improve motor function in models of Parkinson’s.
|
Interesting. But I suppose we’ll now have to wait for years before this new “gold drug” goes into clinical trial for Parkinson’s?
Actually no.
Clene Nanomedicine have already completed 1 study and there are 6 ongoing clinical trials involving CNM-Au8 – one of which is focused on Parkinson’s.
Really?
Really.
A “First-In-Human” Phase I study of CNM-Au8 was completed back in October 2016. It involved 86 healthy volunteers who were allocated to one of two study arms: a single ascending dose and multiple ascending dose arms. All dosing regimes were well-tolerated in this study, with treatment-emergent adverse events reported as predominantly mild. There were no serious adverse events (Click here to read more about this trial).
Due to promising preclinical data described above and the favorable toxicity profile in the Phase I trial, Clene Nanomedicine initiated a Phase II, double-blind, randomised, placebo-controlled clinical trial in Multiple Sclerosis. This study is called VISIONARY-MS, and it is currently ongoing. It involves 150 individuals with Multiple Sclerosis in Australia, who are being treated with CNM-Au8 (or placebo) for 24 weeks (Click here to read more about this trial). There is also a brain imaging clinical study investigating CNM-Au8 in individuals with Multiple Sclerosis (Click here to read more about that trial).
Clene Nanomedicine is also setting up the Phase II “RESCUE-ALS” trial for Amyotrophic Lateral Sclerosis This study will involve 42 adults in Australia (Click here to read more about this trial). There is an associated ALS study being conducted in Boston (US – click here to read more about this), and a brain imaging study in 24 individuals with ALS in Texas as well (Click here to read more about this).
But of interest to our discussion here is the REPAIR-PD study.
This is a single-center open-label pilot study being conducted at the University of Texas Southwestern Medical Center.
 It will involve 24 people with Parkinson’s (within 3 years of being diagnosed) being treated with CNM-Au8 for 12 weeks. There will be no placebo treated control group. The study will be focused on assessing the metabolic effects, safety, pharmacokinetics, and pharmacodynamics of CNM-Au8 in people with Parkinson’s (Click here to read more about this trial).
It will involve 24 people with Parkinson’s (within 3 years of being diagnosed) being treated with CNM-Au8 for 12 weeks. There will be no placebo treated control group. The study will be focused on assessing the metabolic effects, safety, pharmacokinetics, and pharmacodynamics of CNM-Au8 in people with Parkinson’s (Click here to read more about this trial).
The REPAIR-PD study was started in July 2019, and is scheduled to finish in the middle of next year (2020). If the results are encouraging, larger placebo controlled trials will need to be conducted in order to test the efficacy of CNM-Au8.
So it is early days for CNM-Au8 in term of Parkinson’s, but this will be one potential therapy to keep an eye on.
So what does it all mean?
Novel therapeutic approaches are being proposed for the treatment of Parkinson’s all the time. Many are a new angle on an old treatment, and still years from getting into the clinic. But every now and then, you read about an area of research which is not only taking a fresh approach, but also (surprise!) is further along than you might have assumed. Learning about the research being conducted by Clene Nanomedicine has been one such pleasant surprise – innovative approach and possibly heading for Phase II clinical trials soon (if the Phase I results look encouraging).
The publication of more of the preclinical and clinical data (from Clene Nanomedicine) in peer-review research journals would make life a lot easier for a poor blogger trying to learn something about their work – but this is an issue that most intellectual-property-obsessed, ‘blackbox’ biotech firms struggle with. I will be looking out for press releases from Clene Nanomedicine in the hope that they will provide more insight and the results of their clinical trial in Parkinson’s.
And when they are released, I’ll mention them here.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like!
The banner for today’s post was sourced from Madartlab


Thumbs up for the Recaps, and as per my previous comment on another post great to be reminded again about the basic science “The nico…tin..amide whatever part” and ATP
LikeLike
Hi Martin,
Thanks for your comment. Yes, I like the recaps as well.
Kind regards,
Simon
LikeLike
Fascinating! Gold salts have been a long-standing treatment for RA. I recently learned that Donald Triplett, Kanner’s first discovered case of “classic autism” from the 1940s, used gold salts for his severe juvenile RA, and many of his autism symptoms disappeared along with the arthritis!
It’s great to find such a comprehensive but readable explanation of what was going on behind the scenes… Exciting stuff!
LikeLike
Love the recaps too. Thanks.
LikeLike
Thanks I found the Recaps to be helpful in periodically re-focusing on the main point.
Mac
LikeLike
Dear Simon,
It seems that the REPAIR study results were presented at the 2021 MDS virtual congress just one week ago.
CNM-Au8 seems to improve (although not statistically significant) the metabolic profile in the brain (measured by a new SPECT 31P technique ?). It would be a first trial also showing target engagement in the brain!
Good starting point for an update in a futur post 😉 ?
LikeLike