|
Today’s post is a recap of Day 2 at the World Parkinson’s Congress meeting in Kyoto, Japan. I will highlight some of the presentations I was able to catch and discuss some of my key take-aways from the day’s activities.
|
Early meetings meant that I arrived late to the morning session of presentations on the Day 2 (6th June) of the WPC meeting. But luckily I was in time to catch Benjamin Stecher giving his talk in the main hall.
Diagnosed at 29 years of age with young onset Parkinson’s, Ben has spent the last couple of years touring the world requesting meetings with Parkinson’s researchers, to learn more about what they do and what still needs to be done. This quest has give him a unique perspective on the state of Parkinson’s research, and has helped him in his role as an advocate.
I was looking forward to hearing him speak on the main hall stage,…
…and, like everyone else in the room, I was surprised by what he did during his talk.
He invited the other presenters from the morning session to the stage and included them as part of his presentation by asking them questions which are most relevant to the patient community.
His talk involved just 5 or 6 questions, but they were good ones.
And to their credit, the other speakers answered the questions admirably.
For example:
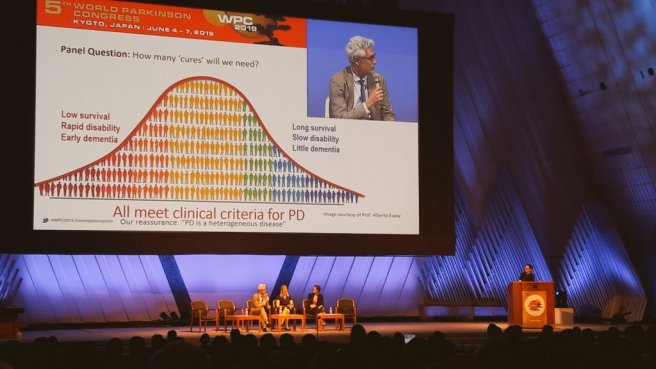
Question: How many ‘cures’ will we need?
Answer: It will depend on the type of Parkinson’s and the stage at which people are in the course of the condition.
One of Ben’s questions asked whether we should be treating each genetic form of Parkinson’s as a separate disease? And he saluted the Silverstein Foundation as a great example of this, with the recent news that a Silverstein-supported biotech company Prevail Therapeutics has just been given the green light from the US FDA to start clinically testing their novel gene therapy approach for GBA-associated Parkinson’s (Click here to read the press release… and, yes the SoPD will definitely have a post in the near future on this new clinical trial programme!).
It was an inspired talk, and the audience was fully engaged, which showed itself when Ben joined the other speakers for the Q&A session at the end of the morning presentations – the questions came thick and fast.
Questions from the audience included:
Question: Is multi-treatment approaches an option?
Answer: Yes, but safety first. And we need to build on something that works before we start combining therapies.
Question: Is privacy an issue with regards to genetic data?
Answer: Yes, but approaches like that being taken by Nebula Genomics could be a possible solution (Click here to read a previous SoPD post on Nebular Genomics).
Question: Wouldn’t a worldwide database of people with Parkinson’s be useful?
Answer: Bureaucracy & technical issues make this challenging (a missed opportunity for the panel here, they were playing safe)
It was a great morning session.
 Kiyomizu Temple. Source: JNTO
Kiyomizu Temple. Source: JNTO
After the lunch period, I wandered along to “Advancing Research, Clinical Trials and Real-World Data” session, which was being chaired by Prof Simon Lewis from Sydney. The speakers included:
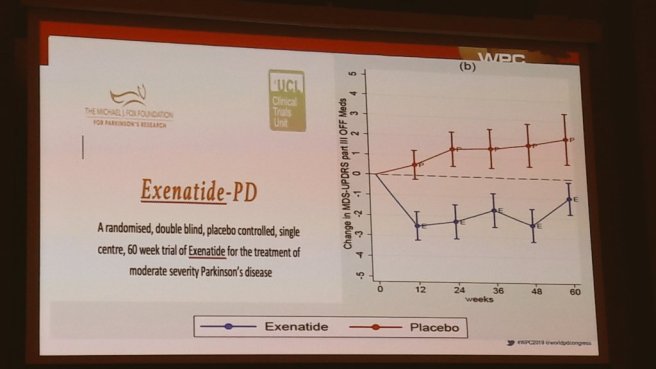
Prof Tom Foltynie – who is the principle investigator behind the Exenatide clinical trial program
Richard Windle – advocate, blogger and participant in the Transeuro study
Prof Bas Bloem – neurologist and one of the co-founders of the hugely successful ParkinsonNet.
Prof Foltynie began this session by providing an overview of current and experimental symptomatic treatments for Parkinson’s….
Before shifting on to potentially disease modifying approaches. He obviously spoke about the Exenatide work, as well as Nilotinib, immunotherapy & the GDNF trial.
And while cautioning against raising expectations too high, he finished by saying that “there is a lot of clinical trial activity going on to be hopeful about“.
For those interested in hearing Prof Foltynie discuss more about the Exenatide work, I can recommend watching this webinar (which is chaired by Prof Patrik Brundin):
Richard Windle was next to speak in this afternoon session, and his talk was titled “What do the guinea pigs really think?”.
Richard provided the audience with some very personal insights, based on his experience in the Transeuro cell transplantation trial for Parkinson’s. Anyone familiar with Richard’s website will know that he is very astute and has his finger on the pulse of Parkinson’s research. His final thoughts regarding issues that need to be addressed (see image above) are worthy of further consideration/discussion.
Next was Prof Bas Bloem who was discussing “Using real-world data as an alternative to clinical trials” – a topic he is very passionate about.
Here is a video of him (kindly provided by the good folks at Parkinson’s NZ) discussing the new PRIME-PD model – “Proactive & Integrated Management & Empowerment in Parkinson’s” – a novel 5 year collaborative effort to provide truly innovative care for people with Parkinson’s, led by Dr Emily Henderson of Bristol University, with support from the Gatsby Foundation (Click here to read more about PRIME-PD):
In his WPC talk, Prof Bloem spoke about the need for assessment to be personalised – “one size does not fit all so why do we do it?“.
He also spoke about the use of real world data, and he presented the video with the gentleman with Parkinson’s who has trouble with freezing of gait, but can ride a bike:
And this kind of approach has led to interesting clinical trials:
Click here to read more about that trial.
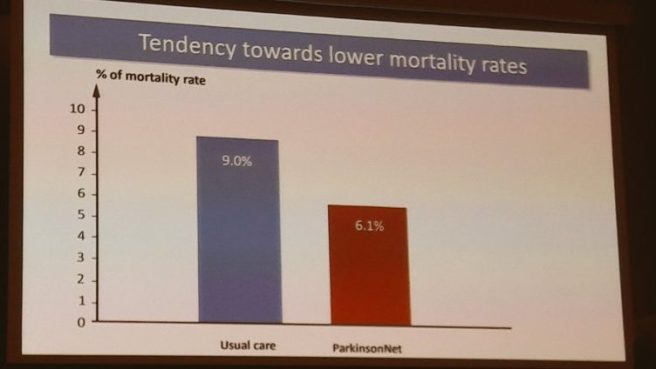
Bas also talked about how ParkinsonNet is helping to develop data (over time) of better community treatment. Shifting from doctor-driven treatment to more focus on actual patient needs.
He presented interesting examples of how analysis of medical claims databases of ParkinsonNet users and non-users in the Netherlands suggests very positive trends.
I have seem Prof Bloem speak before, but this was a really good performance.
Unfortunately I couldn’t hang around for the Q&A of this session as I was chairing a panel discussion immediately after this, on how patients can get involved in research:
 John & Sue Whipps, myself, Gary Rafaloff & Tanya Simuni
John & Sue Whipps, myself, Gary Rafaloff & Tanya Simuni
That’s me in the middle of the image above – looking like I’ve fallen asleep! A badly timed photo. I was really enjoying the discussion.
On the panel were John and Sue Whipps (advocates involved with the Peninsula Parkinson’s Interest Group – PenPRiG), Gary Rafaloff (super advocate – PDtrialtracker), and Prof Tanya Simuni (who coordinated the recent STEADY-PD III clinical trial. There was a wealth of experience, across a broad spectrum on this panel, with lots of interesting perspectives.
The session was filmed and once it is available I’ll link it here.
And that brought to an end a very long day 2 at the World Parkinson’s Congress meeting in Kyoto.
——————————————
Key take aways from Day 2. of the WPC meeting:
1. Parallel sessions – I feel like the meeting is passing by and I am missing out on a great deal. Other than recording lectures for viewing later, I’m not sure how to resolve this issue. And it is a good problem to have: there are simply too many options/too much on offer at the WPC!
2. I am continually impressed by the level of engagement from the Parkinson’s community. People keep saying we need more, but when you look at those who are already engaged, their level of knowledge and advocacy is extremely impressive!
Tune in for a summary of the third and final day in the next post.
The banner for today’s post was sourced from travelcaffeine.










2 thoughts on “WPC 2019 – Day 2”