|
The immune system is our main line of defense against a world full of potentially dangerous disease causing agents. It is a complicated beast that does a fantastic job of keeping us safe and well. Recently, however, there was an interesting study suggesting that a genetic risk factor for Parkinson’s may be associated with an over-reaction from the immune system in response to infection from a common human food poisoning bug. Specifically, mice who were missing the gene PINK1 literally had an ‘autoimmune reaction’ to the infection – that is the immune system began attacking healthy cells of the body – while normal mice (with intact PINK1 genes) recovered from the infection and went about their business. In today’s post, we will explore this new research and discuss why we may need to rethink PINK.
|
Source: Huffington Post
I have had a guts full of all this gut research being published about Parkinson’s.
[NOTE 1.: For the unitiated: A “guts full” – Adjective, Kiwi colloquialism. Meaning ‘Had enough of’, ‘fed up of’, ‘endured to the point of tolerance’]
[NOTE 2.: The author of this blog is a Kiwi]
I really can’t stomach anymore of it.
And my gut feeling suggests that there is only more to come. It would be nice though, to have something else… something different to digest.
So what is today’s post all about?
Gut research of course.
But this gut research has a REALLY interesting twist.
A group of researchers in Canada recently published this report:
 Title: Intestinal infection triggers Parkinson’s disease-like symptoms in Pink1−/− mice
Title: Intestinal infection triggers Parkinson’s disease-like symptoms in Pink1−/− mice
Authors: Matheoud D, Cannon T, Voisin A, Penttinen AM, Ramet L, Fahmy AM, Ducrot C, Laplante A, Bourque MJ, Zhu L, Cayrol R, Le Campion A, McBride HM, Gruenheid S, Trudeau LE, Desjardins M.
Journal: Nature, 2019 Jul 17 [Epub ahead of print].
PMID: 31316206
The Canadian researchers who conducted this study wanted to explore why genetic variations in the stretch of DNA called PINK1 increases the risk of developing Parkinson’s.
Hang on a second. What exactly is PINK1?
Approximately 10-15% of Parkinson’s cases are associated with particular variations in DNA that render people vulnerable to developing the condition (Click here to read more on the genetics of Parkinson’s). Some of these variations are in specific regions of DNA (called genes) that provide the instructions for making particular proteins.
There are currently 20+ genes that are referred to as ‘PARK genes’, as genetic variations within those genes make people more vulnerable to developing Parkinson’s.
One of those PARK genes is called PINK1 (or PARK6).
What does PINK1?
PINK1 is involved with a number of functions, but it has been most studied in the context of mitophagy.
What is mitophagy?
Mitophagy is the cellular process of disposing of old/damaged mitochondria.
For the uninitiated, mitochondria are the power house of each cell. They help to keep the lights on. Without them, the party is over and the cell dies.

Mitochondria and their location in the cell. Source: NCBI
You may remember from high school biology class that mitochondria are tiny bean-shaped objects within the cell. They convert nutrients from food into Adenosine Triphosphate (or ATP). ATP is the fuel which cells run on. Given their critical role in energy supply, mitochondria are plentiful (some cells have thousands) and highly organised within the cell, being moved around to wherever they are needed.
Like you and I and all other things in life, however, mitochondria have a use-by date.
As mitochondria get old and worn out (or damaged) with time, the cell will recycle them via a process called mitophagy (a blending of the words mitochondria and autophagy which is the waste disposal system of each cell).
What does PINK1 do in mitophagy?
PINK1 acts like a kind of handle on the surface of mitochondria. In normal, healthy mitochondria, the PINK1 protein attaches to the surface of mitochondria and it is slowly absorbed until it completely disappears from the surface and is degraded. In old or unhealthy mitochondria, however, this process is inhibited and PINK1 starts to accumulate on the outer surface of the mitochondria. Lots of handles poking out of the surface of the mitochondria.
And there is a second Parkinson’s-associated protein called PARKIN which is a flag that likes to hold onto the PINK1 handle. While exposed on the surface of mitochondria PINK1 starts grabbing the PARKIN protein. This pairing is a signal to the cell that this particular mitochondrion (singular) is not healthy and needs to be removed. The pairing start the process that leads to the development of a structure called a phagophore, which engulfs the mitochondria and eventually leads to its safe disposal.

PINK1 and PARKIN in normal (right) and unhealthy (left) situations. Source: Hindawi
In the absence of normal PINK1 or PARKIN proteins, there is no handle-flag system and old/damaged mitochondria start to pile up and exhibit mitochondrial stress. They are not disposed of appropriately and as a result the cell gets sick and ultimately dies.
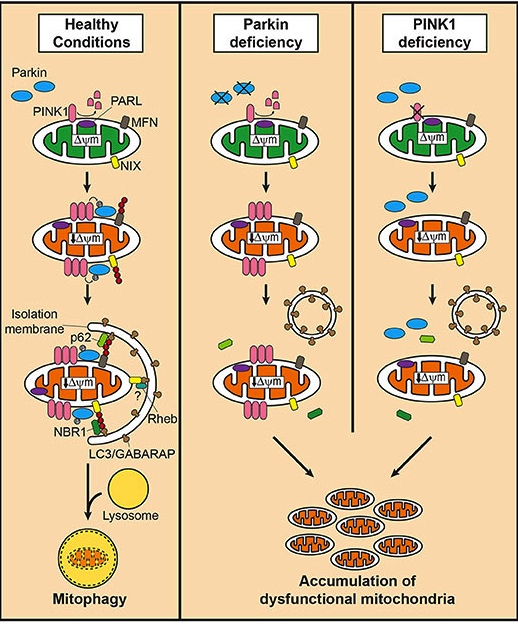 Mitophagy. Source: Frontiersin
Mitophagy. Source: Frontiersin
People with particular genetic mutations in the PINK1 or PARKIN genes are vulnerable to developing an early onset form of Parkinson’s (generally before 40 years of age – again, please note the age). It has long been believed that the dysfunctional disposal of (and accumulation of) old mitochondria is part of the reason why these individuals develop the condition at such an early age.
But recently it has been reported that mice with PINK1 or PARKIN mutations have normal basal levels of mitophagy (Click here to read more about this).
So researchers have gone back to the drawing board and re-evaluated this mitophagy idea, and started explored new possibilities.
And what have they discovered?
Well, in the study we are reviewing in today’s post, the researchers took young mice that were genetically engineered to not produce PINK1 protein gene, and they purposefully infected them with a bacterium called Citrobacter rodentium.
 Citrobacter rodentium – cute huh? Source: fineartamerica
Citrobacter rodentium – cute huh? Source: fineartamerica
Citrobacter rodentium is very similar to the common human food poisoning bug E. coli, and they caused inflammation in the gut of the mice. The researchers also infected normal mice (with a working copy of the PINK1 gene) with Citrobacter rodentium, and they were able to handle this infection – no problem.
But mice without PINK1? That was a completely different story.
And here is where the tale gets interesting:
The PINK1 mice started to slowly develop Parkinson’s-like symptoms.
Que?
Specifically, these Citrobacter rodentium infected mice (with no PINK1) displayed decreased density in the branches of the dopamine neurons in a reion of the brain called the striatum (where dopamine neurons release the bulk of their dopamine). The loss of dopamine and dopamine neuron branches in the striatum is hallmark of Parkinson’s. And it gradually leads to motor impairments (inhibition of movement) which the researchers also observed in these infected PINK1 mice.
Interestingly, these motor issues in the mice could be reversed by treatment with L-DOPA.
Are the mice permanently Parkinsonian?
No, they are not.
Curiously, the Parkinson’s-like behaviour features were only a transcient effect. The infected mice (with no PINK1) returned to normal about 12 months after the infection.
What?!? How do the researchers explain that?
To better understand why these PINK1 mice started having issues, the researchers had a good look at response of the immune system.
Why the immune system?
Because a couple of years ago, these same scientists published this report:
 Title: Parkinson’s Disease-Related Proteins PINK1 and Parkin Repress Mitochondrial Antigen Presentation.
Title: Parkinson’s Disease-Related Proteins PINK1 and Parkin Repress Mitochondrial Antigen Presentation.
Authors: Matheoud D, Sugiura A, Bellemare-Pelletier A, Laplante A, Rondeau C, Chemali M, Fazel A, Bergeron JJ, Trudeau LE, Burelle Y, Gagnon E, McBride HM, Desjardins M.
Journal: Cell, 2016 Jul 14;166(2):314-327.
PMID: 27345367 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that PINK1 – and its partner PARKIN – actively inhibit immune responses elicited by inflammation.
And in the new study, they found that while the immune system in normal mice handled the Citrobacter rodentium infection in a normal fashion, the immune cells of the mice with no PINK1 completely ‘over-reacted’.
This situation resulted in an ‘autoimmune reaction’.
What exactly is an autoimmune reaction?
An autoimmune reaction is an immune response within an organism against its own healthy cells and tissues. Many diseases result from such an immune response, and they are called autoimmune diseases.
Different types of autoimmune diseases. Source: DrJockers
Luckily for the mice (with no PINK1), as mentioned above, they gradually returned to normalcy – the immune system calmed down and there was an easing of the autoimmune reaction.
Lucky mice indeed. So does this suggest that Parkinson’s could be an autoimmune disease?
It is an interesting idea.
Some forms of Parkinson’s could well be associated with an over active immune system, and there is some evidence to support it – one recent study suggests that naturally occurring “autoantibodies” (which play an important role in clearing and blocking circulating ‘self’ proteins) are lower in people with Parkinson’s than healthy control subjects:

Title: Autoimmune antibody decline in Parkinson’s disease and Multiple System Atrophy; a step towards immunotherapeutic strategies.
Authors: Brudek T, Winge K, Folke J, Christensen S, Fog K, Pakkenberg B, Pedersen LØ.
Journal: Mol Neurodegener. 2017 Jun 7;12(1):44.
PMID: 28592329 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers collected blood samples from samples from 46 people with Parkinson’s, 18 people with Multiple System Atrophy (a condition very similar to Parkinson’s), and 41 healthy control subjects. When they analysed the blood for autoantibodies targeting the alpha synuclein protein, they found reduced levels in people with Parkinson’s when compared to healthy controls, and even more reduced in people with Multiple System Atrophy.
The researchers concluded that reduced levels of these antibodies for alpha synuclein results in more alpha synuclein floating around and causing a potentially inflammatory environment. They also proposed that the results provide a good rationale for testing immune-based therapeutic strategies directed against pathological alpha synuclein (such as the Affiris and Prothena clinical trials we have previously discussed – click here to read more about this).
A second recent report supporting a potential autoimmune association with Parkinson’s is a much larger study:

Title: Genome-wide Pleiotropy Between Parkinson Disease and Autoimmune Diseases
Authors: Witoelar A, Jansen IE, Wang Y, Desikan RS, Gibbs JR, Blauwendraat C, Thompson WK, Hernandez DG, Djurovic S, Schork AJ, Bettella F, Ellinghaus D, Franke A, Lie BA, McEvoy LK, Karlsen TH, Lesage S, Morris HR, Brice A, Wood NW, Heutink P, Hardy J, Singleton AB, Dale AM, Gasser T, Andreassen OA, Sharma M; International Parkinson’s Disease Genomics Consortium (IPDGC), North American Brain Expression Consortium (NABEC), and United Kingdom Brain Expression Consortium (UKBEC) Investigators.
Journal: JAMA Neurol. 2017 Jun 5. doi: 10.1001/jamaneurol.2017.0469.
PMID: 28586827
In this study, the researchers analysed DNA collected from 138 511 individuals of European ancestry and they identified 17 novel genetic loci shared between Parkinson and a series of autoimmune conditions (including type 1 diabetes, Crohn disease, ulcerative colitis, rheumatoid arthritis, celiac disease, psoriasis, and multiple sclerosis).
According to this study, apparently healthy individuals with a lot of these shared genetic variants which predisposes them to inflammation conditions, could also be at increased risk for developing Parkinson’s. PINK1 was not mentioned in this study, but Parkinson’s associated LRRK2 was associated with rheumatoid arthritis, ulcerative colitis and Crohn disease.
So there is evidence to suggest that some cases of Parkinson’s may be associated with an autoimmune reaction.
What is interesting in the report we have reviewed in today’s post, however, is that the mice recovered. These mice are housed in near sterile environments though, and one has to wonder what would happen to the mice with no PINK1 if they lived in the wild? Would constant exposure to environmental disease causing agents result in a chronically ‘over-reacting’ immune system? And would this ultimately lead to a more permanent Parkinsonian state?
These questions still need to be investigated.
But clinical trials are now underway exploring the use of anti-inflammatory therapies in Parkinson’s which could be useful based on the results of the report reviewed in today’s post. For example, Inzomelid which is a NLRP3 inhibitor being developed by a company named Inflazome.
This company has recently started a Phase I clinical trials of Inzomelid in healthy individuals (Click here to read more about this trial), with plans to begin testing in people with Parkinson’s in the near future (Click here to read a previous SoPD post on this topic).
So what does it all mean?
For a long time, the Parkinson’s-associated protein PINK1 has been connected to the process of removing old/damaged mitochondria (mitophagy). A great deal of effort has been made in trying to determine why genetic errors in the PINK1 gene are associated with early onset Parkinson’s, and how this could be related to the process of mitophagy.
But perhaps we have been barking up the wrong tree?
Perhaps we need to re-think PINK?
Many of the proteins in our body have multiple functions and in trying to understand diseases associated with various proteins, we need to understand all of those processes. And by re-thinking what we think we know, we are identifying new avenues to potentially treat Parkinson’s.
Not really gut research in the end I guess, but hopefully you still found it interesting.
The banner for today’s post was sourced from Goodfon


I have read quite a bit about pink and Parkin beginning about 2014 or so – interesting re the E-Coli GI connection but am more interested in epigenetic changes that might lead to identifying pink and Parkin as genetic pre disposition when an anti inflammatory diet with nutritional ketosis can lead to reversal of those epigenetic changes that might lead to improvement in disease severity and or reversal as noted in the mice without pink1 – hope that makes sense – if they were able to clear E-Coli after returning to a regular diet? – I have MSA supposedly – was on hospice for 18 months almost 6 years ago – from hospice to wheelchair to walker to – I am now walking with a cane and see many other improvements altho I am now having more bradycardia while reclined with accessory muscle and diaphragmatic weakness seen on reclined PFT’s – are you aware of ALS symptoms in Parkinsons Plus disorders with bradycardia? I do a diet against disease blog posting links to research on the biome & dybiosis and barrier disruption coupled with autoimmune disorders among other stuff on groups.yahoo.com – site posted below – I also attended a rheumatologist conference on JAK inhibitors and spoke to a group of rheumatologists about biologics etc for the treatment of neurologic/neurodegenerative disorders and was informed to my astonishment that their annual rheumatology congress had several neurologic rheumatologists present who they said were using rheumatologic therapies in theses diseases with significant success – I am unable to find studies related to this – are you aware of anything like this? Hope thats not too many questions – appreciate your posts and thanks in advance for any insight you might have!
LikeLike
Food for thought,definitely. As vit D3 or analogues are known to suppress psoriais inflammation, does the same apply to PD?
D3 modifies the ROS- NLRP3-IL1B axis in retinopathy: https://www.hindawi.com/journals/jdr/2018/8193523/
And: https://www.sciencedirect.com/science/article/pii/S2213231718309248
Very high dose D3 is being used by one of the authors for his psoriasis: https://www.sciencedirect.com/science/article/abs/pii/S0960076018306228#bib0410 Physiologucal solar synthesis of D3 is ca 20,000 IU pd.
Such doses have yet to be formally trialled AFAIK for PD
Peter
LikeLike
Hi Peter,
Thanks for your comment. Yeah, dosing is the issue. There is a great deal of research suggesting ‘this’ works or ‘that’ works in cell culture or mice, but translating to humans at some of the doses being used could be rather tricky.
Kind regards,
Simon
LikeLike
Hi Simon,
For D3 the max human phyisological dose is known and the serum levels too. No trials I have seen come close to those levels, And none have to my knowledge embraced concommitant high dose D3 with vit K2. I’ll get back to you in ten years with my own result !
Best wishes,
Peter
LikeLike
Dear Simon:
Thank you so much for your excellent summary of Parkinson’s research. It’s my favorite site.
Could you please update us as to the status of the several Ambroxol clinical trials? At least one has been completed but no data (as far as I know) has been released.
Many Thanks and keep up the great WORK!
Nate McMullen
LikeLike
The Pink idea is very popular. I heard it a long ago and already read it thousands of times. But some questions are always poke me.I can’t discover contemplates identified with this, would you say you are mindful of anything like this? I trust that is not very numerous inquiries, value your posts and thanks ahead of time for any knowledge you may have.
LikeLike