|
Today’s post starts with more of a biology lesson than usual, but it is important to understand where in the grand scheme of things a certain type of blood cell sits. That type of blood cell has a really cool name: Natural killer cells. Recently researchers at the University of Georgia (USA) published a report suggesting that natural killer cells may be a key player in the immune system response to Parkinson’s. Specifically, they found that natural killer cell numbers are higher in disease-affected parts of the Parkinsonian brain, and that natural killer cells digest free floating alpha synuclein aggregates. In today’s post, we will discuss what natural killer cells are, review this new research report, and explore what this new finding could mean for Parkinson’s.
|
 Milestone! Source: Smilingkidsindy
Milestone! Source: Smilingkidsindy
My daughter recently lost her first tooth, and there was a bit of blood. We patched her up, but also took advantage of the moment to learn a little something about how the body works.
Me: Do you know what that red stuff is?
Little monster: Is it blood?
Me: That’s right.
Little monster: Am I going to die? (accompanied with a sudden and very concerned look on her face)
Me: No.
Extremely relieved little monster: Papa, where does blood come from?
And that was when I got all excited, and pulled out my black board.
Admittedly it took a while, but this was the answer I gave her:
 Source: Wikipedia
Source: Wikipedia
The field of hematopoiesis (or blood formation) is vast and absolutely fascinating.
You start off with a couple of small multi-potential hematopoietic stem cells (or a “Hemocytoblasts”) and – it given enough time – they can give rise to an entire blood system. And at any moment in time that blood system will contain 20-30 trillion cells (in the average human body).
And as the image above suggests, there are quite a few branches of potential cell types that these blood stem cell can generate.
This is all very interesting, but what does this have to do with Parkinson’s?
Potentially a lot.
And what we need to do today to make our way down one of those branches (in the image above) to a particular type of blood cell, which we are going to be focusing on today.
Blood accounts for approximately 7% of your body weight, and the average adult has a blood volume of roughly 5 litres (11 US pints).
There are three chief components of blood:
- Red blood cells
- White blood cells
- Plasma (92% water and 8% other stuff – think blood clotting proteins, waste, nutrients, etc)
By volume, the red blood cells constitute about 45% of whole blood, the plasma about 54.3%, and white cells about 0.7%.
 Source: KhanAcademy
Source: KhanAcademy
When it comes to the cellular parts of blood, one microliter of blood contains:
- Approximately 5 million erythrocytes, which are ‘red blood cells’ that distribute oxygen around the body. These are small, bi-concave shaped cells that lack any nucleus, but this extra space allows for more hemoglobin – the critical protein involved in transportation of oxygen.
 Source: KhanAcademy
Source: KhanAcademy
- 200,000–500,000 thrombocytes, (also known as platelets) which take part in blood clotting. Thrombocytes are are not actually cells, but rather cell fragments. They are produced when large cells called megakaryocytes which each release 2-3000 platelets in their life time. Platelets are small (2-4 micrometers), lens-shaped structures that float around in the blood system waiting to help plug a rupture.
 Source: KhanAcademy
Source: KhanAcademy
- 5,000–10,000 leukocytes, which are the white blood cells that form the body’s immune system – chewing up and removing old or rogue cells or waste, as well as attacking any foreign or infectious agents.
For the rest of this post, we are going to focus on a particular type of leukocyte.
What are leukocytes?
Leucocytes (or ‘white blood cells’) are the part of the blood system involved in protecting the body against both infectious disease and foreign invaders
They can be divided into two different groups based on what they look like under a microscope:
- granulocytes – which have tiny granules inside them
- agranulocytes – which do not have tiny granules inside them (simple enough)
This division can broken down further into five major subtypes:
 Source: KhanAcademy
Source: KhanAcademy
And while it would bring me endless pleasure to discuss the roles of each of these sub-types, I will spare you that fate and dedicate the rest of this post to the cell on the far right (of the image, not political persuasion).
That cell is a lymphocyte.
What are lymphocytes? And when does any of this actually get back to Parkinson’s?
When your is attacked by a pathogen (a disease causing agent) like the common cold virus, it will elicit what is called an immune reaction. The presence of the virus inside the body will be detected by cells in the immune system and given that the virus will be clearly determined to be ‘not self’ (or not part of your body), an immune response will be initiated.
The cells that carry out the immune response are the lymphocytes.

That big cell in the middle is a lymphocyte. Source: ASH
And for the rest of this post, we are going to focus solely on a type of lymphocyte.
There are basically three types of lymphocytes:
- B cells
- T cells
- Natural killer cells (cool name right?)
B-cells, T-cells and Natural killer cells are highly specialised blood cells that defend our body when things go wrong (which they inevitably do, it’s just the way life is!).
B-cells are bone marrow-derived cells which produce Y-shaped proteins called antibodies that are used to label the invading pathogens (such as viruses), and alert other immune cells that these unwanted pathogens need to be disposed of. By sticking to pathgens, antbodies can also block them from doing their function.

Antibodies. Source: Astrazeneca
Utterly useless pub quiz fact: The ‘B’ doesn’t come from ‘bone’ or ‘blood’, but rather it comes from the name of the place these cells were discovered: the Bursa of Fabricus. The Bursa is an organ only found in birds.
 Source: Askabiologist
Source: Askabiologist
B-cells do not kill pathogens, they just spend their short life producing antibodies which trap and neutralise them.
The killing of pathogens is left to the T-cells and natural killer cells.
Thymus-derived T-cells kill cells can be identified by the presence of a T-receptor on surface of the cell. This T-receptor identifies antigens (a molecule capable of inducing an immune response) and binds to them, which activates the T-cell. Each T-cell targets a specific antigen.
Natural killer cells are different to the T-cells.
In a nutshell: T-cells kill a specific, unique target. Natural killer cells, on the other hand, kill any thing that doesn’t look right to them.
Natural killer cells were first noticed because of their ability to kill cancer tumour cells, but what made them particularly interesting was that their approach to killing occurred without any priming or prior activation. T-cells need ‘priming’ by antigen presenting cells, but natural killer cells are simply natural killers – hence the name.
In the image below, you can see a large purple/blue cell (this is a natural killer cell) making contact with lots of other cells.
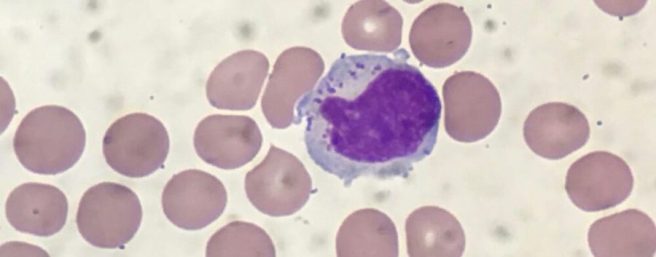 A NK cell. Source: Dana-Faber
A NK cell. Source: Dana-Faber
Natural killer cells wander around touching the surface of cells, checking that everything is ok.
The activation of a natural killer cell is controlled by a balance of signals from activation and inhibitory receptors. Inhibitory natural killer cell receptors recognise cells that are ‘self’ (or part of you) via the presence of a cluster of proteins on the surface of a cell called “MHC class 1”.
This inhibitory receptor keeps the natural killer cell in check.
If MHC class 1 is present on a cell that the natural killer cell interacts with, then the natural killer cells remains inactive and it moves on to the next cell. The presence of MHC class 1 counteracts any activating signals – a life or death balancing act (illustrated in image A. below):
 Source: BMC
Source: BMC
If HMC class 1 is not present on a target cell, however, then the natural killer tendencies of the natural killer cell are unleashed and cytotoxic granules will be released. These granules cause stress on the target cell and rupture the membrane, ultimately causing the targeted cell to die.
Note that natural killer cells do not kill by default. That is to say, when MHC class 1 inhibition is absent, natural killer cells must still be stimulated through activation receptors. They are not completely senseless killing machines.
|
RECAP #1: Natural killer cells are white blood cells (or Leucocytes), and form an important part of our immune system – how our body protects itself against outside invaders. They are part of the innate immune system, meaning that they are always present and do not really adapt in their approach to protecting the body from viruses or infection.
|
Given their innate killer tendencies, natural killer cell are not part of the ‘adaptive immune system’ (like T-cells). Natural killer cells are not activated in response antibodies or antigens, but rather by a family of chemical messengers (or cytokines) called interferons.
What are interferons?
Interferons are a group of proteins that get their name because of what they do: they interfere with the replication of viruses.
They are a family of signaling proteins, which are made and released by cells in your body in response to a viral infection. Interferons causes nearby cells to be on the alert, and to heighten their anti-viral defenses.
 Source: EBC
Source: EBC
Interferons also help to protect cells from bacterial infections and may inhibit cancer growth. But by releasing interferons, cells can also alert roving natural killer cells to the fact that they are in trouble and the natural killer cells are not inclined to showing much mercy. If they think a cell is sick, natural killer cells will simply kill it.
Once activated by these interferons, natural killer cells start releasing cytotoxic (or cell-killing) granules which then destroy an affected cells (eg. tumor or viral infected cell).
Now I appreciate that this has been a longer than usual intro/biology lesson, but it is important to know some of these “blood cell basics” and it will help in understanding the research report we are going to review today.
So we are about to get back to Parkinson’s?
Yep. With this report:
 Title: NK cells clear α-synuclein and the depletion of NK cells exacerbates synuclein pathology in a mouse model of α-synucleinopathy.
Title: NK cells clear α-synuclein and the depletion of NK cells exacerbates synuclein pathology in a mouse model of α-synucleinopathy.
Authors: Earls RH, Menees KB, Chung J, Gutekunst CA, Lee HJ, Hazim MG, Rada B, Wood LB, Lee JK.
Journal: Proc Natl Acad Sci U S A. 2020 Jan 3. [Epub ahead of print]
PMID: 31900358
In this study, the researchers were interested in the role of natural killer cells in Parkinson’s. And they began their analysis by looking at postmortem brains from people with (and without) Parkinson’s.
Specifically, the investigators went looking for natural killer cells in an area of the brain called the substantia nigra (which is where the dopamine neurons live – a population of cells that are severely affected by PD). They found that while there were very few natural killer cells present in the control brains, the Parkinsonian brains had higher levels of natural killer cells. They found that a mouse models of Parkinson’s (the PFF α-syn in M83 transgenic mouse model) also had higher levels of natural killer cells in the brain than normal untreated control mice.
Given the presence of natural killer cells in the brain of Parkinson’s patients and models of PD, the researchers next turned their attention to what those cells might actually be doing in the Parkinsonian brain. They found that the natural killer cells internalising and degrading extracellular alpha synuclein protein.
Remind me one more time: What is alpha synuclein?
As regular readers will be aware alpha synuclein is considered to be one of the main trouble makers in Parkinson’s. It may sound like a distant galaxy, but it is an extremely abundant protein in our brains – making up about 1% of all the proteins floating around in each neuron.
When alpha synuclein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure. Alone, it will look like this:
Alpha synuclein. Source: Wikipedia
By itself, alpha synuclein is considered a monomer, or a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). In Parkinson’s, alpha synuclein also aggregates to form what are called ‘fibrils’.

Microscopic images of Alpha Synuclein (AS) monomers, oligomers and fibrils. Source: Brain
These fibrils are believed to be passed between cells, which may explain the slow progression of Parkinson’s. And as these fibrils enter a cell they are thought to ‘seed’ aggregation of the protein in that cell. In this manner, alpha synuclein may have a prion-like property. There aggregations affect many aspects of cellular function.
 The effects of aggregated Alpha Synuclein protein in a neuron. Source: R&D
The effects of aggregated Alpha Synuclein protein in a neuron. Source: R&D
In many cases of Parkinson’s, alpha synuclein aggregates clump together, which is believed to lead to the appearance of Lewy bodies.
What are Lewy bodies?
Lewy bodies are dense circular clusters of alpha synuclein protein (and other proteins) that are found in specific regions of the brain in people with Parkinson’s (Click here for more on Lewy bodies).
A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
The aggregated alpha synuclein protein, however, is not limited to just the Lewy bodies. In the affected areas of the Parkinsonian brain, aggregated alpha synuclein can be seen in the branches (or neurites; see black arrow in the image below) of cells. In the image below, alpha synuclein has been stained brown on a section of brain from a person with Parkinson’s.
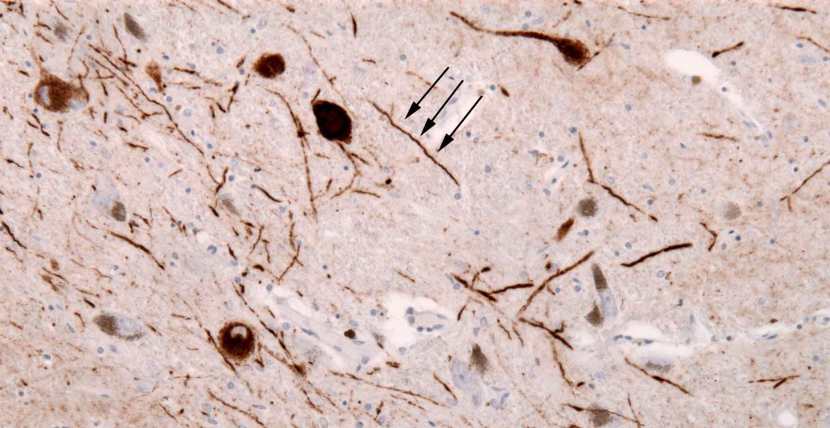
Examples of Lewy neurites (stained in brown; indicated by arrows). Source: Wikimedia
|
RECAP #2: Researchers report that the number of natural killer cells in certain regions of the Parkinsonian brain are increased. They also found that natural killer cells were internalising and degrading extracellular alpha synuclein protein.
|
Ok, so now that that is explained, what did you mean when you said that natural killer cells “were internalising and degrading extracellular alpha synuclein protein”?
I mean the researchers found that the natural killer cells were grabbing alpha synuclein protein that was floating around outside cells and chewing it up.
These cells were scavenging loose alpha synuclein that is released from neurons, and the researchers suggested that this may be critical for restraining the pathology associated with Parkinson’s. By grabbing the alpha synuclein, the natural killer cells might be slowing the spreading of Parkinson’s.
Next the investigators looked at what different types of alpha synuclein (monomers and aggregates) could do to the behaviour of natural killer cells. Curiously, they found that aggregates of alpha synuclein could reduce the ‘killer nature’ of natural killer cells.
What does this mean?
As we mentioned above, natural killers cells kill other cells by releasing toxic proteins (or cytotoxic granules) on to the target cell that they want to kill.
The investigators found that natural killer cells that digest alpha synuclein aggregates produce less of these toxic proteins. In effect, this result suggests that alpha synuclein may be reducing the role of natural killer cells.
Oooh, interesting. What did the researchers do next?
To explore what impact this phenomenon might have in Parkinson’s, the researchers next reduced levels of natural killer cells in mouse models of Parkinson’s, and they found that the symptoms of the mice got worse. The motor behaviour of the mice was worse in models of Parkinson’s with no natural killer cells.
In addition, the aggregation of alpha synuclein in the brain was worse in these mice with no natural killer cells, and the reduction in dopamine activity was more significantly worse (but curiously, the number of dopamine neurons did not change). The loss of natural killer cells also increased the levels of neuroinflammation in the mouse model of Parkinson’s.
The researchers concluded that their “data provide a role of natural killer cells in modulating synuclein pathology and motor symptoms in a preclinical mouse model of Parkinson’s, which could be developed into a therapeutic for Parkinson’s”.
|
RECAP #3: The investigators found that natural killer cells that consumed aggregated alpha synuclein released less cytokines. They also reported that reducing the number of natural killer cells in mouse models of Parkinson’s made the symptoms worse and increased the amount of alpha synuclein pathology.
|
What do they mean “could be developed into a therapeutic for Parkinson’s”?
So given their cancer cell killing ability, natural killer cells have been adapted by researchers to make them even more potent in the treatment of cancer.
In medicine, a procedure called “adoptive transfer” involves collecting natural killers cells from a patient with cancer, then activating the natural killer cells and expanding them in cell culture before re-introducing them into the patient. Natural killers cells can also be genetically adapted while they are in cell culture, to make them even more potent.
 Adoptive transfer. Source: Nature
Adoptive transfer. Source: Nature
Difference versions of this technique are being widely applied to different areas of cancer. Since natural killer cells appear to be able to reduce levels of excess alpha synuclein floating around outside of cells, one could envisage a similar adoptive transfer style approach being tested in Parkinson’s.
How?
By increasing the number of natural killer cells, perhaps we could clear more extracellular alpha synuclein?
This question needs to be addressed in models of Parkinson’s before we get too carried away with this idea. One of the weaknesses of the current study is that they only looked at what happens when you reduce the number of natural killer cells. The opposite experiment (increasing the number of natural killer cells) could have been quite informative.
Is anyone testing this idea?
One would assume that the researchers behind the study are now exploring this idea.
In addition, in 2017, the Michael J Fox foundation funded a research project exploring the idea of using immune cells to tackle Parkinson’s (Click here to read more about this). It will be interesting to see the results of this work (and other research) – hopefully this year.
Interesting. Has anyone ever looked at natural killer cells in Parkinson’s before?
Yes, there have been analyses of blood from people with Parkinson’s.
And recently, large meta-analyses of that research have been conducted:
 Title: The correlation of lymphocyte subsets, natural killer cell, and Parkinson’s disease: a meta-analysis.
Title: The correlation of lymphocyte subsets, natural killer cell, and Parkinson’s disease: a meta-analysis.
Authors: Jiang S, Gao H, Luo Q, Wang P, Yang X.
Journal: Neurol Sci. 2017 Aug;38(8):1373-1380.
PMID: 28497309
In this study, the researchers conducted a meta analysis of 21 case-control trials involving 943 cases of Parkinson’s, and they reported an increase in the number of natural killer cells was apparent in Parkinson’s.
So what does it all mean?
Our immune system is ruthlessly efficient (in most cases) at keeping us healthy. We wander through a world of potential pathogen, which invade us every moment of the day, and our immune system puts up an amazing fight to keep us functional. I hope you will agree that the complexity of that system is fascinating.
Harnessing the power of the immune system could be very useful for future therapeutic approaches targeting Parkinson’s. By using the alpha synuclein eating properties of the natural killer cells, we may be able to help slow the progression of the condition. Still a lot of work to be done in testing this idea, it is an intriguing idea.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from genengnews



Well written hopeful science, as always!
LikeLike
“This is all very interesting but I really want an Elmo bandaid” is what your daughter really said.
LikeLike
I hope your daughter also got a magical story about the tooth fairy alongside her biology lesson. Both would be important.☺
LikeLike