|
There is a lot of research currently being conducted that is exploring the relationship between the gastrointestinal system and Parkinson’s. A growing body of data suggests that the bacteria in our guts may be having an infuential role. Recently researchers at the University of Edinburgh in Scotland have published the results of a study in which they used microscopic roundworms to explore bacteria that can influence the pathology associated with Parkinson’s. And they announced that the bacteria that they had identified is present in many available probiotics. In today’s post, we will look at what roundworms are, what is meant by probiotic, and what this new research found.
|
 Not the PDUK committee. Source: Eagle
Not the PDUK committee. Source: Eagle
What I am about to do is utterly inappropriate, but it will hopefully provide readers with a small insight into what is happening behind the scenes in Parkinson’s research.
Several years ago I was a member of the Parkinson’s UK project grant review panel, and our job was to review applications from fellow researchers who were requesting funds to conduct large and ambitious projects. Following several weeks of reviewing the grants, we had all travelled down to London to discuss a set of applications that had been shortlisted for funding. About 30 people (both academics and PwPs) were in the room, sitting around a large square of tables, and it was our job to select the grants that should be funded.
There was a lot of pros and cons being discussed for each application… and then we came to one particular grant that I think had caught everyones eye.
It involved using C. elegans to screen for gut bacteria that may influence aspects of Parkinson’s biology.
What are C. elegans?
Caenorhabditis elegans (or simply C. elegans) are transparent nematode – also known as roundworms. They are about 1 mm in length, and they have very well characterised nervous systems (useless pub quiz fact: C. elegans have 302 neurons and 56 glial cells in total, which communicate through approximately 6400 chemical synapses, 900 gap junctions, and 1500 neuromuscular junctions – like I said, well characterised!).
 Caenorhabditis elegans – cute huh? Source: Nematode
Caenorhabditis elegans – cute huh? Source: Nematode
Given their well characterised nervous systems, C. elegans provide a useful tool for studying biology. They are easy to grow/maintain, they have an overall life span of 2-3 weeks, and researchers have developed a wide range of tools that allow for genetic manipulation to address specific questions.
But the idea of using these little guys to study the bacteria of the gut was such a novel idea. The grant funding panel sat there wondering if these creatures even had bacteria in their guts (they do – click here to read a review of the first 3 studies on this topic, all published in 2016).
I liked the novelty of the approach and thought that the high-throughput nature of the study would provide quick answers (a lot of bacteria could get studied very quickly using C. Elegans, compared to other organisms like mice).
But there was a lot of debate about this application, centred mainly around the idea of “How will any of this translate to humans?”
Source: DMM
Eventually I think curiosity won the day and the committee agree that the application should be funded. And I’m glad it was, because very recently the first results of the study have been published,… and they are really interesting.
What did the study find?
This report was published a week or so ago:
 Title: Probiotic Bacillus subtilis Protects against α-Synuclein Aggregation in C. elegans.
Title: Probiotic Bacillus subtilis Protects against α-Synuclein Aggregation in C. elegans.
Authors: Goya ME, Xue F, Sampedro-Torres-Quevedo C, Arnaouteli S, Riquelme-Dominguez L, Romanowski A, Brydon J, Ball KL, Stanley-Wall NR, Doitsidou M.
Journal: Cell Rep. 2020 Jan 14;30(2):367-380.
PMID: 31940482 (This report is OPEN ACCESS if you would like to read it)
In this study, researchers in Scotland wanted to assess the effect of gut bacteria on the clustering (or aggregation) of alpha synuclein protein, and they used a C. elegans model of Parkinson’s to do this.
Can you remind me again: What is alpha synuclein?
As regular readers will be aware alpha synuclein is considered to be one of the main trouble makers in Parkinson’s. It may sound like a distant galaxy, but it is an extremely abundant protein in our brains – making up about 1% of all the proteins floating around in each neuron.
When alpha synuclein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure. Alone, it will look like this:
Alpha synuclein. Source: Wikipedia
By itself, alpha synuclein is considered a monomer, or a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). In Parkinson’s, alpha synuclein also aggregates to form what are called ‘fibrils’.

Microscopic images of Alpha Synuclein (AS) monomers, oligomers and fibrils. Source: Brain
These fibrils are believed to be passed between cells, which may explain the slow progression of Parkinson’s. And as these fibrils enter a cell they are thought to ‘seed’ aggregation of the protein in that cell. In this manner, alpha synuclein may have a prion-like property. There aggregations affect many aspects of cellular function.
 The effects of aggregated Alpha Synuclein protein in a neuron. Source: R&D
The effects of aggregated Alpha Synuclein protein in a neuron. Source: R&D
In many cases of Parkinson’s, alpha synuclein aggregates clump together, which is believed to lead to the appearance of Lewy bodies.
What are Lewy bodies?
Lewy bodies are dense circular clusters of alpha synuclein protein (and other proteins) that are found in specific regions of the brain in people with Parkinson’s (Click here for more on Lewy bodies).
A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
The aggregated alpha synuclein protein, however, is not limited to just the Lewy bodies. In the affected areas of the Parkinsonian brain, aggregated alpha synuclein can be seen in the branches (or neurites; see black arrow in the image below) of cells. In the image below, alpha synuclein has been stained brown on a section of brain from a person with Parkinson’s.
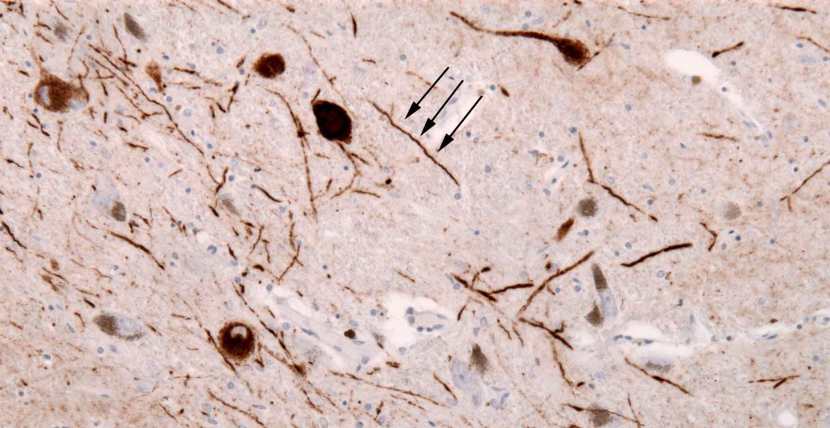
Examples of Lewy neurites (stained in brown; indicated by arrows). Source: Wikimedia
Given the importance of alpha synuclein aggregation in Parkinson’s, the researchers in Scotland wanted to use a C. elegan model of Parkinson’s to assess the influence of gut bacteria on the aggregation of alpha synuclein in neurons. So they used C. elegans that had been genetically engineered to produce high levels of human alpha synuclein.
|
RECAP #1: Microscopic worms called C. Elegans represent a rapid means of screening and idenifying agents that may influence a particular biological process. Researchers in Scotland wanted to use these organisms to identify gut-based bacteria that influence the behaviour of a protein called alpha synuclein which accumulates and aggregates in Parkinson’s.
|
But why did the researchers think that gut bacteria would influence protein aggregation in the brain?
There is a growing body of evidence suggesting that the gut and all of its constituent parts is different in people with Parkinson’s (Click here and here for some examples of previous SoPD gut posts). There is even some evidence to suggest that the condition could be starting in the gut in some cases (see the very first SoPD post to read more on that).
And there have recently published report of preclinical studies suggesting that the gut could be influencing this debilitating condition.
Such as this study:

Title: Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease
Authors: Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE, Challis C, Schretter CE, Rocha S, Gradinaru V, Chesselet MF, Keshavarzian A, Shannon KM, Krajmalnik-Brown R, Wittung-Stafshede P, Knight R, Mazmanian SK
Journal: Cell, 167 (6), 1469–1480
PMID: 27912057 (this report is OPEN ACCESS if you would like to read it)
The researchers (who have previously conducted a lot of research on the microbiota of the gut and its interactions with the host) used mice that have been genetically engineered to produce abnormal amounts of alpha synuclein.
The researchers tested these alpha synuclein producing mice and normal wild-type mice on some behavioural tasks and found that the alpha-synuclein producing mice gradually performed worse.

A lab mouse. Source: USNews
The researchers then raised a new batch of alpha-synuclein producing mice in a ‘germ free environment’ and tested them on the same behavioural tasks. ‘Germ free environment’ means that the mice have no microorganisms living within them. So these mice have a very limited microbiota in their gastrointestinal tract.
And guess what happened:
The germ-free alpha-synuclein producing mice performed as well as on the behavioural task as the normal mice. There was no difference in the performance of the two sets of mice.
Que? How could this be?
This is precisely what the researchers were wondering.
So they decided to have a look at the brains of the mice, where they found less aggregation of alpha synuclein protein in the brains of germ-free alpha-synuclein producing mice than their ‘germ-full’ alpha-synuclein producing mice.
This result suggested that the microbiota of the gut may be somehow involved with controlling the aggregation of alpha-synuclein in the brain. The researchers also noticed that the microglia – helper cells in the brain – of the germ-free alpha-synuclein producing mice looked different to their counterparts in the germ-full alpha-synuclein producing mice, indicating that in the absence of aggregating alpha synuclein the microglia were not becoming activated (a key feature in the Parkinsonian brain).
|
RECAP #2: There is evidence that bacteria in the gut may be different between people with and without Parkinson’s. There is also pre-clinical evidence that suggests these bacteria could be potentially influencing the pathology of Parkinson’s.
|
So should we get rid of the bacteria in our guts?
No.
The bacteria in our guts plays an important and symbotic (or co-operative) role for us. We would run into trouble very quickly without them. They serve many important functions.
But we could perhaps be more selective in what we hold in our bellies.
This, however, is a very difficult task.
What do we mean?
There is a universe of life in your gastrointestinal system. You should never feel alone, because you aren’t. Another interesting, but utterly useless pub quiz fact: There is approximately 2kg of bacteria in your gut, which is actually heavier by weight than your brain (1.5kg).
 Gut bacteria. Source: Biogaia
Gut bacteria. Source: Biogaia
But half of that microbial life is unknown to us (scientifically speaking). From a 30,000 foot-like view of things, gut bacteria can be broadly classified into basic, large families, but when you start to get up close and personal with individual bacterium, you discover some strange details.
For example, in amongst the thousands of different types of bacteria in our guts, there is no single type of bacteria that we all share. There is no ‘housekeeping organism’ that is present in all human guts, which could be used as a measuring stick of diversity in our bodies. This makes research into the microbiome of the gut rather difficult – there is no standard to measure by.
But this should not stop us from looking for bacteria that may have beneficial properties, which brings us back to the researchers in Scotland.
So what did they find?
The researchers fed their alpha synuclein-producing C. elegans with different batches of gut bacteria and assessed the amounts of aggregation (72 h after hatching). Among the bacterial species investigated was Bacillus subtilis (or B. subtilis), and it turned out to have a very interesting effect on the C. elegans.
While C. elegans on a normal diet exhibited aggregation, the animals fed on B. subtilis exhibited a near complete absence of aggregates. And this was despite an increase in alpha synuclein RNA levels.
Intrigued, the researchers next wanted to determine if B. subtilis could reduce levels of aggregation after the alpha synuclein protein has actually aggregated. Could shifting to a diet of B. subtilis reduce already formed alpha synuclein aggregates?
The answer?
Yes.
The researchers found that most of the aggregates cleared within 1 day after they switched the C. elegans from a normal diet to a diet including B. subtilis. And this protective effect appears to be a common feature of B. subtilis, because when the investigators tried different batches of B. subtilis they saw very similar results.
Interesting. So what did the researchers do next?
They wanted to know what the B. subtilis were doing that caused this protective effect. So they started investigting metabolites.
What are metabolites?
They are any substance formed during or necessary for metabolism.
Oh yeah, of course! Once again in English please?
They are substance made or used when the body breaks down food, drugs chemicals, or even its own tissue. These metabolites can have functions, and the researchers were assuming that after the B. subtilis were established in the gut of the C. elegans they were releasing metabolites which were acting on the roundworms to produce the aggregation reducing effect.
The researchers reported that the observed protective effect is partly mediated by DAF-16.
What on earth is DAF-16?
DAF-16 is a protein in C. elegans which eventually across aeons of evolution became the FOXO family of proteins in humans.
And what pray tell is FOXO family?
They are proteins that play various important roles in regulating cell growth, proliferation, differentiation, and longevity.
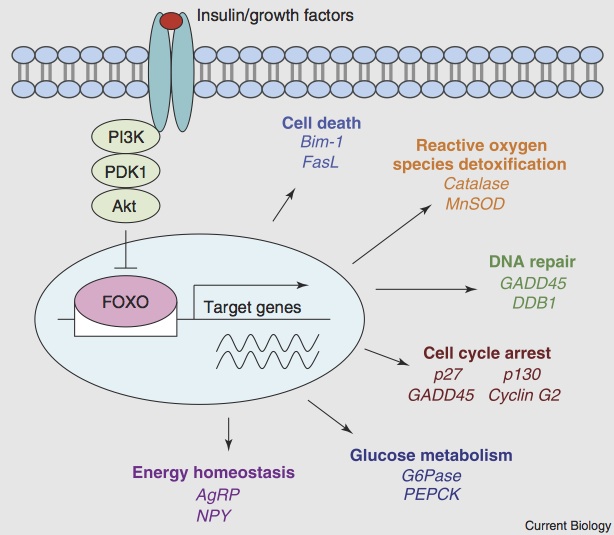 Source: Maria
Source: Maria
The researchers also found that B. subtilis was affecting sphingolipid metabolism, which is associated with Parkinson’s (Click here for a previous SoPD post on this topic). Long story short, the researchers concluded that a probiotic “that is available for human consumption, both inhibits aggregation and efficiently removes preformed aggregates in a C. elegans model with ectopic expression of human α-syn”.
Very interesting, but what is a “probiotic”?
Probiotics are “live microorganisms, which when administered in adequate amounts confer health benefits to the host” (FAO/WHO, 2002).
Based on the data presented in the current study, B. subtilis could be considered a useful probiotic for C. elegans – whether it is a useful probiotic for humans is discussed below.
|
RECAP #3: The researchers in Scotland found that a diet of B. subtilis could reduce the deveopment of alpha synuclein aggregation in C. elegan roundworms. In addition, this bacteria could also reduce pre-existing levels of aggregated alpha synuclein. They explored the biological pathways involved in this protective effect and reported proteins involved with longevity as well as lipid metabolism. They concluded that B. subtilis could be a useful probiotic for C. elegans (yet to be determined for mice or humans).
|
Is this the first time B. subtilis has demonstrated beneficial effects in C. elegans?
Actually, no.
In fact, last year this report was published:
 Title: Bacillus Subtilis Delays Neurodegeneration and Behavioral Impairment in the Alzheimer’s Disease Model Caenorhabditis Elegans.
Title: Bacillus Subtilis Delays Neurodegeneration and Behavioral Impairment in the Alzheimer’s Disease Model Caenorhabditis Elegans.
Authors: Cogliati S, Clementi V, Francisco M, Crespo C, Argañaraz F, Grau R.
Journal: J Alzheimers Dis. 2019 Dec 26. doi: 10.3233/JAD-190837. [Epub ahead of print]
PMID: 31884470
In this study, the researchers assessed B. subtilis in a C. elegan model of Alzheimer’s… and guess what?
The investigators reported that a diet of B. subtilis rescued the C. elegan model of Alzheimer’s. The lifespan of the worms were restored to normal levels, compared to the significantly shorter life span of the worms not fed with B. subtilis.
And other studies have suggested that B. subtilis could actually extend C. elegan lifespans. Such as this study:
 Title: Bacillus subtilis biofilm extends Caenorhabditis elegans longevity through downregulation of the insulin-like signalling pathway.
Title: Bacillus subtilis biofilm extends Caenorhabditis elegans longevity through downregulation of the insulin-like signalling pathway.
Authors: Donato V, Ayala FR, Cogliati S, Bauman C, Costa JG, Leñini C, Grau R.
Journal: Nat Commun. 2017 Jan 30;8:14332.
PMID: 28134244 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that C. elegans fed on B. subtilis lived longer (a ~15% increase in lifespan) than worms grown on a normal diet (and this has been demonstrated by others as well – Click here and here to read more about this). And this effect appears to be dependent on DAF-16 (or FOXO in humans) activity.
Wow! Where can I get me some of that B. subtilis stuff?
So it is really important to appreciate that all of the research discussed in today’s post has been conducted in microscopic round worms. It is not clear that the effects seens in these tiny organisms will translate to human (there are a few important differences between the species to consider).
There have been clinical trials of B. subtilis (Click here to read about one such trial), and the bacteria is reported to survive in the human gastrointestinal system and be well tolerated at the doses administered (Click here to read more about this).
But whether it is tolerated at levels required for any possible ‘anti-aggregation’ effect needs to be determined. And for the record: we do not know whether the ‘anti-aggregation’ effect occurs in higher mammals, let alone what dose. So a lot more research is required here before folks should run out start exploring probiotics.
Are there any clinical trials exploring probiotics in Parkinson’s?
There are.
Here in the UK, there is a clinical study being conducted by King’s College London which is looking at the safety and tolerability of Symprove.
 Source: Symprove
Source: Symprove
The study involves 60 people with Parkinson’s being administered with either oral probiotic or placebo for 3 months (Click here to read more about the trial details, and click here to read more about the study itself).
So what does it all mean?
Researchers in Scotland have conducted a screening study for gut bacteria that may influence the behaviour of proteins involved in Parkinson’s. They conducted their study in C. elegans – a type of microscopic roundworm that is widely used and well characterised in biological science. Their results indicate that B. subtilis could be considered a useful probiotic for C. elegans.
Whether this result will translate to mice (and perhaps ultimately humans) is yet to be determined – I’m assuming that researchers are already exploring this. But until those results are shared, the Parkinson’s community should refrain from even considering the unsupervised exploration of probiotics – we do not yet know if reducing alpha synuclein aggregation is a good thing to do. If you are determined to try, however, please consult with qualified health professionals before making any changes to your diet as this could impact your medication and potentially wellbeing.
For those interested, Parkinson’s UK (one of the funders of the study) have a great summary of this research on their medium page (Click here to read that post).
ADDENDUM – 28/1/2020:
In the last 24 hours, I have been made aware that Dr Roberto Grau of Rosario National University (Argentina) has very similar results to the Scotish researchers. At the Alzheimer’s and Parkinson’s Therapies Focus Meeting (AAT-AD/PD) in Turin (Italy) last year (March 15–18th), he presented data suggesting that B. subtilis “suppressed accumulation of α-synuclein into Lewy bodies by 75 percent, and prevented the degeneration of dopaminergic neurons” in C. elegans (Click here to read more about this).
In addition, he is planning a clinical trial to test the B. subtilis strain DG101 in people with Parkinson’s to determine if the probiotic has any potential in PD. Apparently he “plans to enroll more than 100 people in Argentina who are being treated for PD at private hospitals and follow them for six to 12 months”. I am not sure about the status of this proposed trial as I can not find it mentioned on any clinical trial registries (happy to be corrected on this).
Something to keep an eye out for.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from HealthHarvard.

What is the risk of trying a B. subtilis probiotic?
LikeLike
Hi Thomas,
Thanks for your comment. The simple answer is “unknown”. As mentioned in the post, clinical trials of B. subtilis have been conducted at certain doses in humans and it was found to be safe and well tolerated. In addition, it is widely used in foods. But what is not known, is whether the protective effect will even translate to mammals – our brains are a bit further from the gut than roundworms – and if it does, at what kind of doses.
It is a difficult question to answer.
For further reading on this topic: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3539293/
Kind regards,
Simon
LikeLike
Hi,
thanks for the interesting report!
According to the Cell Reports article the anti-aggregation effect seems to be a general property of the Bacillus subtilis species and is shared by all the strains they tested, which were at least four. I wonder why it has not been mentioned that one strain of Bacillus subtilis is contained in substantial amounts in regular food, for example Japanese natto. And I also wonder whether anything is known about the Parkinson’s risk of people who consume such food on a regular basis.
Having Parkinson’s myself and having heard rumors about the potential benefits of natto in PwP already some years ago I made natto a part of my diet, and since it counts as food I am less concerned about potential side effects one might associate with taking a supplement.
zz
LikeLike
Hi ZZ,
Thanks for the interesting comment. While I know natto, I was unaware of this connection. Very interesting!
There was a research paper titled “Amyloid-Degrading Ability of Nattokinase from Bacillus subtilis Natto” in 2009 (https://www.ncbi.nlm.nih.gov/pubmed/19117402) which looked at this. It has been cited 95 times. But I can’t find any research on alpha synuclein or PD. They refer to natto in the link in the addendum I have added to the post (https://www.alzforum.org/news/conference-coverage/do-immune-responses-promote-or-prevent-parkinsons-disease). Very curious. Thanks for sharing. Perhaps a natto clinical trial is on the cards?
Kind regards,
Simon
LikeLike
The gut and the brain are connected – well waddaya know! The first step in holistic treatment of an autoimmune/inflammatory disease is to get the gut healed and healthy. Having Hashi’s has taught me this. While I’m hardly scientifically sophisticated, I do know how my body reacts when I go off plan and have a day of junk food – sugar/carbs specifically. Everything goes off kilter, and takes days to fix. Even sinemet doesn’t work the same. I have to imagine that an anti-inflammatory diet, including necessary supplements (who gets enough sunshine in the dead of winter?) can only help those of us with PD.
LikeLike
Hi, I was wondering how they could test if this can recaptiulated in mice? What mouse models would they use and what assays etc? Thanks!
LikeLike