|
Speeding up the clinical development process is a shared goal across many medical conditions (not just Parkinson’s and neurodegeneration), and there are many different approaches to achieving this that are being explored. Some of these approaches could be considered to be bordering on the unethical, but there are aspects of their structure and design that are still worthy of academic discussion and consideration. One example is a crowd-funded cancer clinical trial called iCancer. In today’s post, we will discuss the iCancer project.
|
 Source: Entitymag
Source: Entitymag
Over the Christmas period, in addition to spending the required amount of time with family and friends, I fell down a rabbit hole.
Before the festive season, I had been exploring different designs of clinical trials to see what had been given serious academic consideration and thought.
I was particularly intrigued with the ‘pay-to-play’ model (in which patients pay to be part of a study). This model has fallen into disgrace due to abuse by unscrupulous individuals profiting off untested, experimental therapies being targeted towards desperate patients.
To be clear: it is utterly unethical for “for-profit” clinics to be selling access to experimental procedures if there is no proof of efficacy (and this is particularly true for the stem cell clinics).
 Source: FDA
Source: FDA
But I was interested in exploring if anyone had actually explored this type of clinical study design or aspects of it in the academic sense as a means of speeding things up.
In my role as a research co-ordinator for a Parkinson’s charity, I have been lucky enough to meet and get to know some folks who are absolute fountains of knowledge and wisdom when it comes to all things related to clinical trial design. And I just straight up asked some of these individuals if anyone had ever given serious academic thought to the ‘pay-to-play’ model?
I recieved an interesting collection of answers – all erring on the side of extreme caution, with some taking a “are you %#@&£$ serious” tone – and I suspect that any reputation I might have had with those individuals is now dented (such is the taint of pay-to-play).
But one individual – perhaps in an effort to reorient a foolish, but hungry mind – pointed me towards a possibly better approach.
It is being proposed by a group called iCancer.
What is iCancer?
Let me tell you an amazing story.
iCancer is a crowd-sourced fund raising effort that raised £2m to rescue a treatment that had shown considerable pre-clinical promise for treating neuroendocrine cancer of the pancreas.
The project was started by writer/journalist, Alexander Masters.
 Alexander Masters. Source: Guardian
Alexander Masters. Source: Guardian
In 2007, Mr Master’s close friend, Dido Davies, was diagnosed with neuroendocrine cancer of the pancreas.
 Dido Davies. Source: Times
Dido Davies. Source: Times
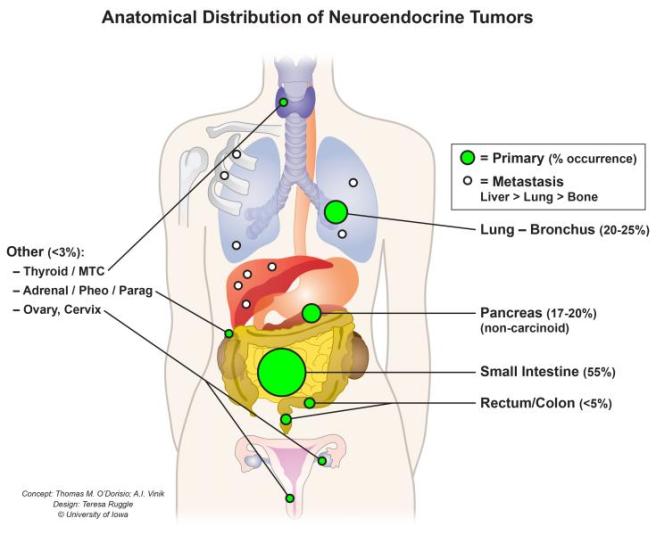
What is neuroendocrine cancer of the pancrea?
Neuroendocrine cells can be found in almost every organ of your body – they are particularly abundant in the gastrointestinal system, the gallbladder, the pancreas and the thyroid.
 Source: UIHC
Source: UIHC
These cells receive inputs from neurons, and in turn they release hormones & other messenger molecules. As a result of this position in the greater scheme of things, neuroendocrine cells are considered an integration point between the brain and the endocrine system.
Neuroendocrine cancer can begin in any of these hormone-producing cells within the neuroendocrine system.
There are different types of neuroendocrine cancers, but most can be divided into one of two main types:
- Carcinoid tumors – these are neuroendocrine cancer that most commonly arise in the digestive tract, lungs, appendix or thymus. They can also grow in the lymph nodes, brain, bone, gonads (ovaries and testes) or skin.
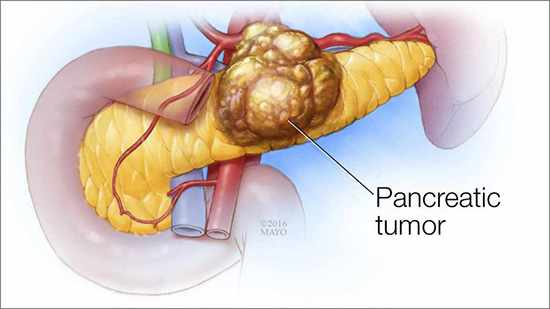
- Pancreatic neuroendocrine tumors (also called “islet cell tumors” or “pancreatic endocrine tumors”) generally arise in the pancreas, but they can also occur outside the pancreas.
 Source: Mayo
Source: Mayo
Alexander’s friend Dido was diagnosed with pancreatic neuroendocrine tumor.
It is a dangerous cancer – the 5-year survival rate for people with pancreatic neuroendocrine tumor is about 42%. The survival rate depends on a variety of factors, such as whether the tumor can be surgically removed. If a tumor can be removed, the 5-year survival rate climbe to 55% (Source).
Understand that this is the cancer that killed Steve Jobs.
 Source: Goalcast
Source: Goalcast
And it also took the Queen of Soul Arethra Franklin.
 Source: Fox
Source: Fox
Ok, so what happened with iCancer?
As Dido started with the trials of a course of chemotherapy, Alexander began desperately searching for an alternative means of curing his friend. Chemotherapy had been invented more than half a century ago, and is well known for its horrific side-effects (which can be fatal). Alexander believed that there had to be a better way.
And then after a long search he found one potential candidate.
It was called “Ad5[CgA-E1A-miR122]PTD“.
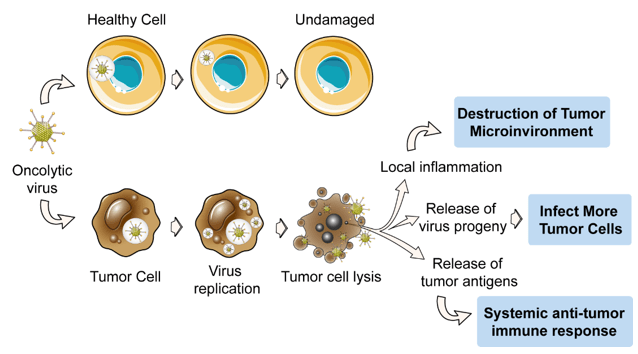
What is Ad5[CgA…whatever?
It is an ‘oncolytic virus’ – that is, a virus that specifically targets cancer cells while leaving other cells alone. This virus selectively replicates in and kills neuroendocrine cells.
 Source: Creative-biolabs
Source: Creative-biolabs
A team of researchers – led by Professor Magnus Essand at Uppsala University in Sweden – had been experimenting with it, but had struggled to get any research funding to take it forward into clinical trial.
 Professor Magnus Essand. Source: Cancerfonden
Professor Magnus Essand. Source: Cancerfonden
The researchers firstly identified this virus as a selective killer of neuroendocrine cells:
 Title: A novel chromogranin-A promoter-driven oncolytic adenovirus for midgut carcinoid therapy.
Title: A novel chromogranin-A promoter-driven oncolytic adenovirus for midgut carcinoid therapy.
Authors: Leja J, Dzojic H, Gustafson E, Oberg K, Giandomenico V, Essand M.
Journal: Clin Cancer Res. 2007 Apr 15;13(8):2455-62.
PMID: 17438105 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers discovered that a virus called Ad[CgA-E1A] could efficiently suppress fast-growing neuroendocrine cancer cells, both in cell culture and in mice. The investigators also noted that the viral genomic material was present at high levels in the neuroendocrine cancer cells, while it was not present in normal cells.
Next, the researchers attempted to improve the neuroendocrine cancer cell killing efficiency of the virus, and they published their results in this report:
 Title: Oncolytic adenovirus modified with somatostatin motifs for selective infection of neuroendocrine tumor cells.
Title: Oncolytic adenovirus modified with somatostatin motifs for selective infection of neuroendocrine tumor cells.
Authors: Leja J, Yu D, Nilsson B, Gedda L, Zieba A, Hakkarainen T, Åkerström G, Öberg K, Giandomenico V, Essand M.
Journal: Gene Ther. 2011 Nov;18(11):1052-62. doi: 10.1038/gt.2011.54. Epub 2011 Apr 14.
PMID: 21490682 (This report is OPEN ACCESS if you would like to read it).
In this study, the researchers genetically modified the Ad[CgA-E1A] virus, so it was about 3–4 times better at infecting neuroendocrine tumor cells. They called the new and improved virus Ad5[CgA-E1A-miR122].
The researchers were really excited by their research and they thought that they had a very interesting potential therapy for pancreatic neuroendocrine tumors. In addition to the obviously positive preclinical results, the virus would be cheap to produce and very easy to administer, and the potential side-effects would most likely be limited to mild flu-like symptoms.
Potentially revolutionary stuff, right?
Sounds great. So what happened next?
Nothing.
Absolutely nothing.
Despite the best efforts of Professor Essand and colleagues, they could not find funding for clinically developing this wondrous potential therapy.
And as a result, the virus was left sitting in the freezer.
What?!? WHY???
An oncologist friend once said to me, “if you ever get a cancer, try to make sure it is a common one“.
The Pharmaceutical Research and Manufacturers of America (source) estimate that for every 5000–10,000 pharmacological compounds that are screened, only 250 enter preclinical testing.
Of these, just 5 will enter Phase 1 safety clinical trials.
Only one (of those 10,000 original compounds) will eventually be approved by the Food and Drug Administration.
And just 2 in 10 approved drugs generate enough revenue to recoup their research and development costs. Thus, when a biotech company decides to fund the clinical trial program for a particular drug, they will need to determine if there is enough of a market to justify the cost of developing the therapy.
 Source: Schumpetercentre
Source: Schumpetercentre
Rare cancers account for approximately 22% of all cancers diagnosed, and they are defined as having an occurrence of less than 6 per 100,000 individuals annually. That is to say, just 6 people in every 100,000 will be diagnosed with that particular form of cancer (Click here to read more about rare cancers).
Despite the exciting preclinical results, due to the rarity of pancreatic neuroendocrine tumors, no one was prepared to provide the £2 million needed to initiate clinical trials of Ad5[CgA-E1A-miR122]PTD.
But that’s terrible. Does this happen often?
Do you want to happy version of the answer or the cold hard reality version?
Can I have the happy version please?
In a report by the Tufts Centre for the Study of Drug Development, the researchers made a rather disturbing discovery: the leading reason for phase I drug failures is commercial viability (Click here to see the report).
That is to say, the main reason a drug does not move from Phase I to Phase II in the clinical trial process, is that a business model can not be established for it. The biotech firm involved can not find investors to support it because those investors do not see any potential for return of capital (plus profit).
The Tuft report found that only 1/3 of the drugs were discontinued at Phase I because of safety issues. More disturbingly, 30% of the phase II studies they analysed failed for commercial reasons, rather than efficacy or safety. The development of the treatment was stopped because the business model didn’t add up.
As Mr Master’s wrote on the iCancer website:
“Every year, an untold number of potential new drugs or interventions, any one of which might go on to improve thousands of lives, are thrown away without being tested in humans. It is a matter of funding, not science: there is not enough money in the public or private sector to run clinical trials on every exciting proposal that comes out of research labs. Thoughtful but hurried (and often arbitrary) judgements are therefore made about which products to save – and the rest of these potentially life-saving therapies are ditched” (Source)
That’s the happy version? But… surely there’s a better way?
So this is where Alexander Masters came up with an idea.
After reading about Prof Essand’s research, he proposed:
“There are over 100,000 people in the world worth more than £20 million. According to medical statistics, between three and five people in every 100,000 get neuroendocrine cancer every year. So, three to five supremely wealthy people will have neuroendocrine cancer. For £1 million, I was going to sell one or two of these plutocrats a place on Professor Essand’s bug trial” (Source)
The central idea of Master’s plan was that “any patient who rescues a potential therapeutic agent from neglect by funding early phase clinical trials (either entirely or in large part) should be offered a place on the trial” (Source).
Clutching this idea, Mr Masters flew to Uppsala and met with Prof Magnus Essand.
 Uppsala. Source: Erasmusu
Uppsala. Source: Erasmusu
He asked Prof Essand if the £2 million they needed could be raised, would he be prepated to re-start work on his unpronounceable virus.
Prof Essand agreed.
When Alexander got back to the UK, he wrote an article about the virus for the Daily Telegraph newspaper (Click here to read the article). The last paragraph of the article read:
“To donate money to Professor Magnus Essand’s research on viral treatments for neuroendocrine cancer, send contributions to Uppsala University, The Oncolytic Virus Fund, Box 256, SE-751 05 Uppsala, Sweden, or visit www.uu.se/en/support/oncolytic. Contributions will be acknowledged in scientific publications and in association with the clinical trial. A donation of £1 million will ensure the virus is named in your honour”
And then he waited.
…
…
…
And then, just 2 weeks after publishing the article, this response was published in the Telegraph:
 Source: Telegraph
Source: Telegraph
And this is where Dominic Nutt comes into the story.
Mr Nutt was not a multi-millionaire, but having been diagnosed with a neuroendocrine tumour he was certainly motivated by the idea of the trial.
He met with Alexander and, together with Liz Scarff and Colin Midson, they started iCancer.
Here is a video of Dominic Nutt discussing the iCancer project:
To support their project, the iCancer team ran an independent peer-review process of the research behind the Ad5[CgA-E1A-miR122] virus so that they could be sure about what they were doing.
The international panel of experts all agreed that the treatment had potential.
Below is a video of Prof Alan Melcher from The Institute of Cancer Research (UK) discussing the research behind iCancer:
Following this peer-review process, the iCancer team started trying to raise the funds for the clinical trial.
So how did they do with raising the funding?
Approximately £700 000 was raised from 3,846 people in 40 different countries via crowdfunding, and the remainder (£1.3 million) came from one individuals: Vince Hamilton
Who is Vince Hamilton?
An Arizona oilman who had been diagnosed with neuroendocrine cancer.
He is one of the 100,000 people in the world worth more than £20 million. And by chance, he had read Alexander’s first article about his idea in a café in Geneva.
Alexander’s article had been published in September 2012, and all of the money was secured by the spring of 2013. Alexander wrote another article in the Daily Telegraph newspaper to celebrate the success of the project (Click here to read that article). Sadly, it was published the same day as his dear friend Dido passed away.
The researchers immediately started working on producing clinical grade virus, and discussing details of the trial with the regulators. The “AdVince”/RADNET iCancer clinical trial started at Uppsala University in 2016 (Click here to read more about the details of this study). Unfortunately, Vince Hamilton also passed away before the trial began, but his wife confirmed their commitment to funding the trial.
The “AdVince”/iCancer clinical trial is still ongoing. It was scheduled to finish in late 2020, so it will be interesting to see if we hear any news about it in 2020. Fingers crossed that the results are positive.
In addition to the trial, however, the iCancer team has been advocating for their model to be applied to other conditions. They have proposed a “Dating Agency” approach, which Alexander Masters and Dominic Nutt discussed in an article published in the British Medical Journal (Click here to read that article).
 Source: iCancer
Source: iCancer
The iCancer project is an amazing story.
One which writer Alexander Masters has written about at length – and I have used his “A plutocratic proposal” article (Click here to read the text, or click here to listen to Alex reading it) as a primary source for much that has been written here.
* * * * * * * * * * *
Some people will argue that it is unethical to provide a place in a clinical trial to a weathy individual if they are prepared to fund the trial. They will say that this is wrong – with massive potential for abuse – and we should simply stick to the way we have always done things.
To those individuals, however, it could be argued that the opposite is also unethical. Why should other potential participants in a trial be denied the chance to take part if the sole source of immediate funding is rejected? Is that ethical?
Especially in areas of medicine that are considered ‘rare’.
There is no right or wrong answer to this situation, but as we seek novel methods of speeding up the clinical trial process, this model should certainly be up for further consideration, discussion, and adaptation.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from iCancer.


2 thoughts on “A plutocratic proposal: iCancer”