|
# # # # One of the more interesting pieces of clinical trial news in 2020 was the publication of the results of a “basket study” for neurological conditions. This was a trial that involved a drug being tested on a selection of neurodegenerative conditions, rather than just one condition. Between December 2013 to May 2017, researchers recruited a total of 29 individuals with Alzheimer’s, 14 with progressive supranuclear palsy (PSP) and 30 with corticobasal syndrome. These participants were intravenously injected with the same drug (TPI-287 – a microtubule stabilizer) once every 3 weeks for 9 weeks (with an optional 6-week open-label extension). Although the findings of the study did not support further development of TPI-287 for tauopathies, the overall structure of the study represents an interesting example of how researchers are taking different approaches to investigating neurodegenerative conditions. In today’s post, we will discuss novel clinical trial designs (“baskets and umbrella”) and other examples of research efforts to better understand neurodegeneration as a whole. # # # # |
Source: HuffPost
It was when my daughter turned 3 years old that the psychological warfare really started.
And I remember the moment of realisation very clearly: It began with her desire for a pet dog.
Up until that point in time, she had limited experience with dogs and her negotiation strategies centred solely around crying. I think she loved “the idea” of a dog, but she was generally quite timid around them. Regardless, she gradually began applying pressure (read: lots of crying) on us to get a dog.
And said pressure began to build rapidly (read: frequent episodes of lots of crying).
 Source: Focus
Source: Focus
Now my wife is definitely not a dog person (“wet, filthy, smelly things“). I on the other hand quite like dogs, but I was utterly, utterly, utterly opposed to getting one because I know full well who will be lumped with the mid-winter late night “walkies” two years down the line: me!
The pressure from our daughter continued to increase, however, until we finally had to sit down with her and explain that we were not going to be getting a dog. On the surface, it looked like she handled this news very well (that is to say: she did not cry). She simply accepted the situation, got up and left the room, saying “Ok”.
My wife and I looked at each other and thought “problem solved”.
The next morning, however, this picture was waiting for us on the kitchen table:
I kid you not.
That’s my daughter and her pet dog (“Linguine“) on the right, and I’m the big, cross-eyed, bad guy on the left.
Since that time the psychological manoeuvring has only become more sophisticated (the teenage years are still a few years away, but I am already absolutely terrified!).
Amusing, but what does this have to do with Parkinson’s?
Well, before telling my daughter that we weren’t getting a dog, I did start looking at the different kinds of dogs and thinking “IF we were to get one, what type of dog would we get?“. But that process only got me thinking what a heterogenous species dogs are.
I mean, look at them! There are so many different breeds, sizes, shapes, and colours:
 Source: Bil-jac
Source: Bil-jac
The only fundamental features (the “first principles”) of the term “dog” is… I don’t know: 4 legs, a tail, and it barks? But even then, some of the barks are extremely unique or just weird:
And all of this brought me back to neurodegenerative conditions, and our efforts to differentiate and stratify them. To put them into semi-definable groups (based on some basic characteristics) and give them a particular label, like “Parkinson’s”.
But as we have said many times on this website, the border lands between many of these conditions is extremely grey areas. And in some cases, even the experts aren’t sure where the borders actually are.
What do you mean?
Many of the differences between the neurodegenerative conditions are extremely subtle, and in a lot of cases there are huge overlaps that no one likes to talk about.
Such as?
In the mid 1980’s, James Leverenz and Mark Sumi of the University of Washington School of Medicine (Seattle) made a curious observation.
After noting the high number of people with Alzheimer’s that often displayed some of the clinical features of Parkinson’s, they decided to examine the postmortem brains of 40 people who had passed away with pathologically confirmed Alzheimer’s – that is, an analysis of their brains confirmed that they had the hallmarks of Alzheimer’s present in their brains.
What the two researchers found shocked them to the core:

Title: Parkinson’s disease in patients with Alzheimer’s disease.
Authors: Leverenz J, Sumi SM.
Journal: Arch Neurol. 1986 Jul;43(7):662-4.
PMID: 3729742
Of the 40 Alzheimer’s brains that they looked at nearly half of them (18 cases) had either dopamine cell loss or Lewy bodies – that characteristic feature of the Parkinsonian brain. They next went back and reviewed the clinical records of these cases and found that rigidity, with or without tremor, had been reported in 13 of those patients. According to their analysis, 11 of those patients had the pathologic changes that warranted a diagnosis of Parkinson’s. But these individuals had been labelled as Alzheimer’s and treated as such.
But the most surprising aspect of this research report?
Almost all of the follow up studies – conducted by independent investigators – found exactly the same result!
It is now generally agreed by neuropathologists (the folks who analyse sections of brain for a living) that 20% to 50% of cases of pathologically determined Alzheimer’s also have Lewy bodies.
In fact, in one analysis of 145 Alzheimer’s brains, 88 (that is 60%) had chemically verified Lewy bodies (Click here to read more about that study).
Oh, and if you are wondering whether this is just a one way street, the answer is “No sir, this phenomenon works both ways”: the features of the Alzheimer’s brain (such as the clustering of a protein called beta-amyloid) are also found in many cases of pathologically confirmed Parkinson’s (Click here and here to read more about this).
Thus, it appears that many neurodegenerative conditions will have a singular label, but the evidence in the brain suggests a shared pathology.
So, does that mean we should be treating all of these conditions the same?
It is an interesting question.
One that has recently been addressed by some researchers who conducted a “basket” clinical study.
A what?
Last year I wrote a post about efforts to get a “multi-arm, multi-stage” (or MAMS) clinical trial platform up and running for Parkinson’s (Click here to read more about this). A MAMS trial is a study that allows for the assessment of several potential therapies at the same time in a large group of individuals with a specific medical condition, like Parkinson’s.
 MAMS clinical trial design. Source: iospress
MAMS clinical trial design. Source: iospress
It is a more dynamic approach when compared to traditional clinical trials as it allows for the shifting of participants seamlessly from a Phase II (safety and efficacy) study to a Phase III trial, by regular within study measures (or interim analyses). Regular assessments of the data as the trial is being conducted mean that decisions can be made about which therapies should be carried forward into Phase 3, and which should be stopped (allowing for new experimental treatments to be added and tested).
There are now numerous examples of MAMS studies for other conditions, but this type of study structure is not the only novel clinical trial design being evaluated by researchers. In fact, there are some really radical ideas currently being evaluated.
Such as?
Well, basket and umbrella studies are good examples.
 Baskets and umbrellas. Source: Amazon
Baskets and umbrellas. Source: Amazon
What are they?
A basket clinical trial takes a collection of conditions that share a similar trait (such as a specific biomarker or perhaps a particular genetic characteristic) and tests a single treatment on all of them to determine if that agent works better for one compared to the others. A schematic of a basket trial can be seen in panel B in the image below):
 Source: Researchgate
Source: Researchgate
An alternative to a basket trial is an umbrella study, which takes a single condition and tests different drugs on it (see panel A in the image above), with the drug allocation being determined by the presence of particular biomarkers in each participant. The key in both basket and umbrella studies are the biomarkers used.
The cancer research world has been leading the charge in novel clinical trial design – conducting MAMS, basket- and umbrella-style studies – and there is a lot that other medical conditions can learn from their experience.
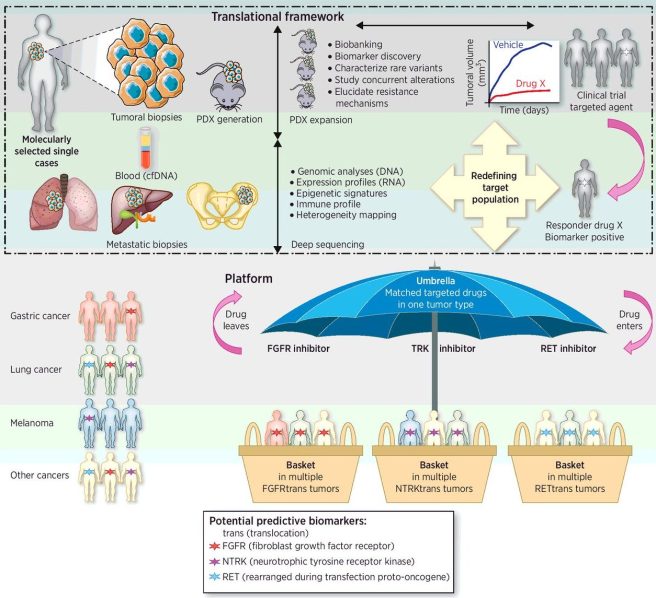 Source: AACR
Source: AACR
And as we shall see, the lessons that oncology have learnt are now being applied to the field of neurodegeneration.
|
# RECAP #1: Many medical conditions are difficult to define, and as a result they are also difficult to diagnose and treat. A lot of neurodegenerative conditions, for example, share some of the pathology in the brain. As a result of this situation, researchers have been exploring novel clinical trial designs to determine if putting several different conditions into a single “basket” and testing a therapy based on a shared characteristic could be the way forward for finding better treatments. # |
Have researchers ever conducted a “neurodegeneration basket study”?
Yes, they have.
This report was published late last year:
 Title: Reactions to Multiple Ascending Doses of the Microtubule Stabilizer TPI-287 in Patients With Alzheimer Disease, Progressive Supranuclear Palsy, and Corticobasal Syndrome: A Randomized Clinical Trial.
Title: Reactions to Multiple Ascending Doses of the Microtubule Stabilizer TPI-287 in Patients With Alzheimer Disease, Progressive Supranuclear Palsy, and Corticobasal Syndrome: A Randomized Clinical Trial.
Authors: Tsai RM, Miller Z, Koestler M, Rojas JC, Ljubenkov PA, Rosen HJ, Rabinovici GD, Fagan AM, Cobigo Y, Brown JA, Jung JI, Hare E, Geldmacher DS, Natelson-Love M, McKinley EC, Luong PN, Chuu EL, Powers R, Mumford P, Wolf A, Wang P, Shamloo M, Miller BL, Roberson ED, Boxer AL.
Journal: JAMA Neurol. 2020 Feb 1;77(2):215-224.
PMID: 31710340 (This report is OPEN ACCESS if you would like to read it)
In their study, the researchers recruited 68 participants for their study. There were 29 individuals with clinically diagnosed Alzheimer’s, 14 people with progressive supranuclear palsy, and 30 cases of corticobasal syndrome.
Three different forms of neurodegeneration, all with one common feature: Tau.
What is Tau?
Have a look at this image:
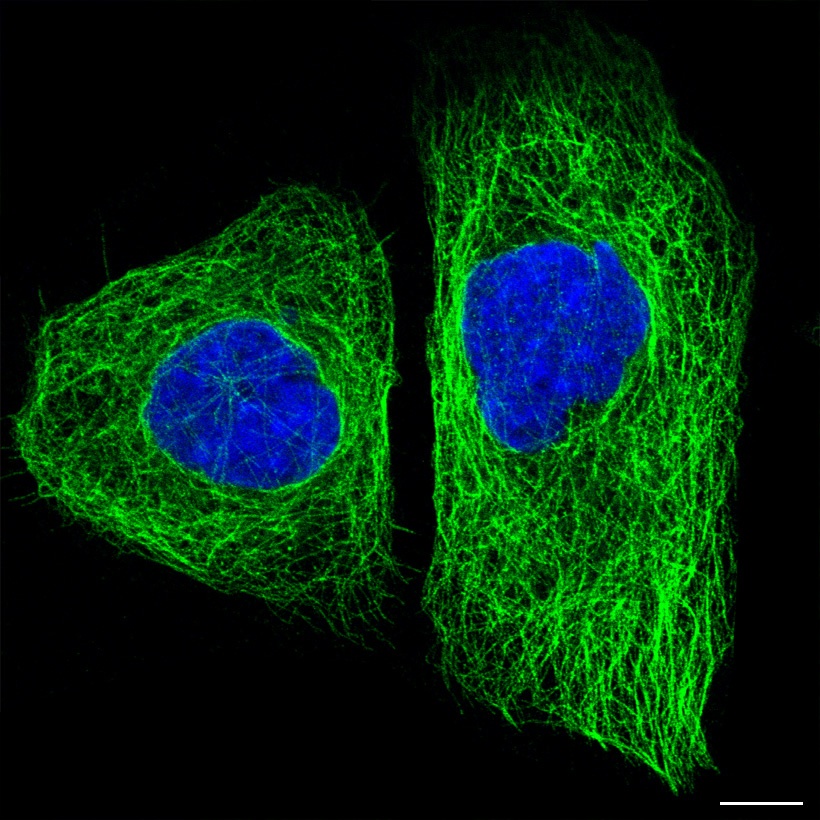
Tau protein (green) inside two cells. Source: The human protein atlas
In the image above you will see a fluorescent photo of two cells that have been labelled with two different dyes: one that highlights the nucleus (in blue) and another that stains the microtubules (in green). Everything else in the cell (membrane and all) is left invisible by this labelling process.
Pretty huh?
Pretty. But what are microtubules?
Microtubules are the highways and byways inside a cell.
They are structures inside of cells that are involved with both cellular and intracellular transportation. That is to say, these skeleton-like structures help cells move from place to place within the body (‘cellular transportation’), but they also aid in the movement of proteins from place to to within the cell (‘intracellular transportation’). On top of this, microtubules also play an important function in cell division.
The stability and maintenance of these microtubles relies on proteins like Tau.
Got it. But what is Tau?
Tau is a microtubule-associated protein – meaning it directly interacts with microtubules. It functions by stabilising these highways and byways inside a cell. Basically, this protein holds together the roads which allows vesicles (the small bags of material) to be moved around the cell (by transporter proteins like kinesin).
 TAU: a stabilising presence. Source: Hindawi
TAU: a stabilising presence. Source: Hindawi
So why is Tau interesting for neurodegeneration?
Because issues with Tau are associated with many types of neurodegeneration.
In conditions like Alzheimer’s, progressive supranuclear palsy, and corticobasal syndrome, there is a build up of Tau protein, which accumulates and clusters (or ‘aggregates’) in a similar fashion to our old friend the Parkinson’s-associated protein alpha synuclein (Click here for a good review article on this topic).
This aggregation of Tau may result in instability in microtubules, which could have disastrous consequences for the health of cells.
 Tau protein in Alzheimer’s disease. Source: Dreamstime
Tau protein in Alzheimer’s disease. Source: Dreamstime
So what did the researchers find in their “basket” study?
In their study, the researchers wanted to evaluate the safety, tolerability, and characteristics of an experimental microtubule stabilizer drug called TPI-287 (aka abeotaxane). 
TPI-287. Source: Chemsrc
The drug was designed to replace Tau and help to stabilise microtubules in people with Alzheimer’s, etc. It was also much better at accessing the brain, compared to other microtubule stabilizing molecules.
It was an interesting molecule to use to test the hypothesis of whether unstable microtubules could be influential in neurodegeneration.
The researchers decided to assess TPI-287 across multiple conditions associated with Tau aggregation (those being Alzheimer’s, progressive supranuclear palsy, and corticobasal syndrome) to determine which of these conditions could benefit from the drug..
The 68 participants recruited to the study were randomly assigned into 4 groups (3 groups treated with different doses of the drug, plus a control/placebo treated group). They were treated once every 3 weeks (intravenous TPI-287 or placebo) for 9 weeks, and then observed during an optional 6-week open-label extension study.
The researchers found that TPI-287 was less tolerated in the participants with Alzheimer’s than in those with progressive supranuclear palsy (PSP), but curiously there was an increase in the number of falls in individuals with PSP who were treated with TPI-287 compared with the placebo arm.
On exploratory endpoint measures, cognitive scores declined less in the individuals with Alzheimer’s who were treated with TPI-287 (but this effect may have been due to a faster rate of cognitive decline in the placebo group than was expected).
Overall, however, the researchers found that there was no significant change in any endpoints between the groups, indicating that the drug has no effect on the progression of any of the conditions. The researchers concluded their report by saying that “The ability to reveal different tau therapeutic effects in various tauopathy syndromes suggests that basket trials are a valuable approach to tau therapeutic early clinical development”, adding “These studies may reveal important differences in safety, tolerability, and potential efficacy between neurodegenerative syndromes that share underlying tau pathologic features”.
Interesting. Have there been any umbrella studies?
If you recall, an umbrella study is one in which different drugs are tested on a single condition, with the drug allocation being determined by the presence of particular biomarkers in each participant.
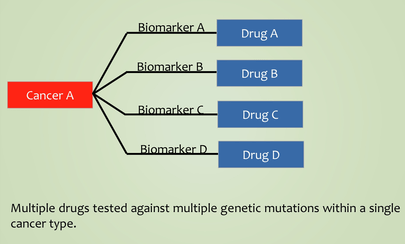 Source: Researchgate
Source: Researchgate
As far as I am aware there haven’t been any umbrella studies in neurodegenerative conditions yet (and I’d be happy to be corrected on this).
For those interested in reading an analysis on basket, umbrella, and other novel clinical trial designs in oncology, a useful meta-analysis was published in 2019 (Click here to read that analysis).
|
# # RECAP #2: Recently a drug with a specific mechanism of action was tested in a basket study of multiple neurodegenerative conditions that shared a common factor (disruption of normal Tau function). The clinical trial found no significant differences between the groups/doses tested, but did note differences in tolerability which could provide insights into important differences in the biology underlying the various conditions. # # |
So no ‘basket’ or ‘umbrella’ trials involving Parkinson’s?
Not that I’m aware of (and – again – I am happy to be corrected on this).
But there is one project that deserves mention here, which is taking a basket-like approach to neurodegeneration, but does not involve testing any experimental therapies. Rather it is taking a big step back from assuming the current biological boundaries between the various neurodegenerative conditions, and instead it is bunching all of the conditions together (in one basket) and then taking a phenotype-agnostic approach to subtyping them.
What does that mean? “Phenotype-agnostic”?
A phenotype is an individual’s observable traits (think height, eye color, and blood type). Some of these traits are determined a person’s genotype (their genetic makeup – written in their DNA), but other traits are largely determined by environmental factors.
The genotype is the genetic coding that influences what the phenotype can look like.
My phenotype is being ridiculously tall and freakishly good looking, but I am not entirely sure about the genotyping of this.
 Source: Expii
Source: Expii
Parkinson’s has certain phenotypic characteristics, like rigidity and tremor. But as we discussed near the top of this post, researchers sometimes see these traits in individuals with other conditions, such as Alzheimer’s.
This observation has led researchers in Cincinnati to initiate the Cincinnati Cohort Biomarker Program (CCBP), which is a biomarker-discovery program aimed at characterizing biological subtypes of neurodegenerative based on the “assumption we do not know which biomarkers have clinical relevance at the individual level“.
 The team behind the project published a report last year outlining what they plan to do.
The team behind the project published a report last year outlining what they plan to do.
Here is the report:
 Title: Phenotype-Agnostic Molecular Subtyping of Neurodegenerative Disorders: The Cincinnati Cohort Biomarker Program (CCBP).
Title: Phenotype-Agnostic Molecular Subtyping of Neurodegenerative Disorders: The Cincinnati Cohort Biomarker Program (CCBP).
Authors: Sturchio A, Marsili L, Vizcarra JA, Dwivedi AK, Kauffman MA, Duker AP, Lu P, Pauciulo MW, Wissel BD, Hill EJ, Stecher B, Keeling EG, Vagal AS, Wang L, Haslam DB, Robson MJ, Tanner CM, Hagey DW, El Andaloussi S, Ezzat K, Fleming RMT, Lu LJ, Little MA, Espay AJ.
Journal: Front Aging Neurosci. 2020 Oct 8;12:553635.
PMID: 33132895 (This report is OPEN ACCESS if you would like to read it)
In the Cincinnati Cohort Biomarker Program, they are planning to enroll 4,000 individuals with neurodegenerative conditions as well as 1,000 unaffected age-matched controls. These folks will be followed-up on a yearly basis for at least 5 years (hopefully extended to 10 and beyond). During each visit, clinical assessments will be made and bio samples (think blood, etc) collected.
“The main aim is to identify biological outliers defining molecular disease subtypes, with a focus on those suitable for targeting with already available therapies (repurposing) in future built-in adaptive clinical trials”
So rather than looking just at people with “Parkinson’s”, the CCBP will seek to take a basket of neurodegenerative conditions and identify biomarkers that differentiate unique biological subgroups, that can then ideally be treated with re-purposable therapies.
This impressive effort is being conducted in parallel to the even more ambitious NIH All of Us program which is also taking place in the USA.
Rather than focusing specifically on neurodegenerative conditions, however, the All of Us project is attempting to collect clinical and biological data across a very large population: 1 million people.
And the goal is “to accelerate biomedical research and improve health“.
In 2019, the huge team behind this effort publish this report outlining the massive project:
 Title: The “All of Us” Research Program.
Title: The “All of Us” Research Program.
Authors: All of Us Research Program Investigators, Denny JC, Rutter JL, Goldstein DB, Philippakis A, Smoller JW, Jenkins G, Dishman E.
Journal: N Engl J Med. 2019 Aug 15;381(7):668-676.
PMID: 31412182 (This report is OPEN ACCESS if you would like to read it)
Started in May 2018, the All of Us program will enroll at least 1 million people across the USA, via 340 research sites:
 A map of the All of Us research sites. Source: NEJM
A map of the All of Us research sites. Source: NEJM
And like the CCBP project, the All of Us program will involve an evaluation for physical measurements and biospecimen collection. Participants will agree to share their electronic health record data, donate biospecimens for laboratory analysis, respond to surveys, and undergo standardized physical evaluations.
All of Us also aims to make the research results accessible to the participants, and it is currently developing novel approaches to make the data more broadly available (to approved researchers).
 Source: NEJM
Source: NEJM
As of December 2020, more than 270,000 people had contributed biospecimens to the project (Source). And on a particularly positive note, many of these participants are from groups that have been historically underrepresented in biomedical research.
It will be interesting to watch how these ambitious projects progress, and what new insights they will provide – particularly in a few years time when they all start reaching a scale where cross platform comparative analysis can be conducted.
They will be incredible resources, along side some of the already available tools (such as Parkinson’s Progression Marker Initiative (PPMI).
So what does it all mean?
On Wednesday 1st January 2020, the American Kennel Club announced two new breeds of dog, bringing the grand total to 195 (Source). Almost 200 different kinds of dogs exist. They come is all kinds of shapes and sizes, and humans typically group them based on their phenotypes (some of the really proud dog owner types could probably get serious and fancy about genotypes as well). And for the most part the taxonomy works. My daughter has very little trouble determining “dog” from “not dog” (though for a while she couldn’t tell the difference between breeds and was referring to most of them as ‘dachshunds’).
Similar efforts with differentiating neurodegenerative conditions have not been so easy or clear cut. And this has been problematic with regards to diagnosis and treatment. But researchers have been experimenting (and continue to) with novel clinical trial designs and large phenotyping studies to confront and deal with this issue head on. Tremendous resources are being applied to the problem and ultimately it will help lead us to a better understanding of these diseases of the brain and how we can better treat them.
A thought that strikes one is what happens if each neurodegenerative condition is truly unique to the individual afflicted? What happens if we can’t group these ailments, and each treatment regime has to be tailored to specific requirements. This is probably the ultimate holy grail of medicine, but does it require a new form of clinical testing? For example, N=one studies (click here to read an old SoPD post about this). It will be interesting to see, but perhaps more thought should be given now to the challenges associated with such studies.
We will explore this in future posts.
# # # #
POSTSCRIPT: About a month after she finally gave up on the dog idea, our daughter announced during dinner one night that “cats are quite nice. Mmmm, I like cats“.
My wife and I looked at each other.
And the next morning, our daughter’s first salvo in a new round of psychological warfare was waiting for us on the kitchen table:
Wish me luck.
I’m going to need it.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from mskcc.




Hello Simon,
Thank you for this great website!
I would like to ask – There were trials in the EU using deferiprone iron chelation to reduce brain iron. I cannot find any results or progress of this trial. Are high brain iron levels still associated with Parkinson’s disease? Have the trials been abandoned?
Regards, Mark
LikeLike
Hi Mark,
Thanks for your comment – glad you like the site.
My understanding is that the FAIRPARK study (https://clinicaltrials.gov/ct2/show/NCT02655315) and the SKY study (https://clinicaltrials.gov/ct2/show/NCT02728843) – both evaluating deferiprone in Europe – should be reporting this year. I think COVID might have simply slowed things down a little, but they have not been abandoned.
Kind regards,
Simon
LikeLike
SIMON
We have always found Labradors very good with children and if you get a working strian rather than a ‘show dog’ you are more likely to have a healthy dog, without the drawbacks of in-breeding.
Thanks for this article which is about atypical parkinsonisms .. very interesting, especially the novel experimental designs.
r
egards
Keith
LikeLike
No Keith! No dog. I know families from my daughter’s school who adopted dogs during the COVID lockdown, and the fathers are all now getting used to a routine of 2-3 walks per day. A couple of them appreciate the break and the solitude of the walks, but still struggle to find the energy to do them.
So no to dogs. They are simply too dependent. A cat on the other hand would be ok – and my wife likes cats. So maybe there’s a cat in our future. We shall see.
Kind regards,
Simon
LikeLike