|
# # # # Today’s post involves a product from a company. The SoPD has not had any contact with the company or associated parties. This post should not be considered as an endorsement or an advertisement of the product. Recently published results from a clinical trial were interesting enough to stimulate this discussion. Omega-3 and omega-6 represent two families of fatty acids that have important biological functions in our bodies. A careful balance of them is required in our diets in order for us to function normally. A recent report from a small clinical trial indicates that daily supplementation with a formulation that includes these molecules could have beneficial effects in Parkinson’s motor symptoms. In today’s post, we will discuss what omega-3 and omega-6 fatty acids are, we will review the new report outlining the study results, and discuss why these results could be interesting. # # # # |
 Source: Foodandwine
Source: Foodandwine
Back when I was young and less beautiful, I had a formidable appetite.
Seriously. My consumption rate was the stuff of legend. There were local all-you-can-eat restaurants that would refuse to serve me, for fear that I would liquidate them.
But I am man enough to admit that I was nothing compared to my friend Jason’s younger brother “Peter”.
One day the three of us went down to the local MacDonald’s during one of their promotions (something like 50 cents per Big Mac) and we challenged ourselves to see who could eat the most. Jason sensibly stopped after finishing 3 burgers, while I had to finally throw in the towel on my 6th burger (to be honest I was struggling from burger #4).
 Source: Thrillist
Source: Thrillist
Jason and I had to ask Peter to stop on burger #9.
I kid you not.
Think about that for a second: NINE Big Macs!
We were watching in bloated horror as this skinny teenage kid was just sitting there – with a milk shake in one hand – throwing back these burgers like they were nothing. Even now it is grotesque to reminisce about, and I really wonder if we didn’t do serious damage to our livers that day.
That is disgusting. What does it have to do with Parkinson’s?
Well, we all do silly things when we are young and invincible. At that age it seems like you can eat whatever you want and there are no consequences. But of course as we get older, we need to start carefully considering what you are putting into your body.
And recently the results of a clinical study were published that indicate that what we consume could also influence the course of Parkinson’s.
What do you mean?
Last month, this research report was published:
 Title: Neuroaspis PLP10™, a nutritional formula rich in omega-3 and omega-6 fatty acids with antioxidant vitamins including gamma-tocopherol in early Parkinson’s disease: A randomized, double-blind, placebo-controlled trial.
Title: Neuroaspis PLP10™, a nutritional formula rich in omega-3 and omega-6 fatty acids with antioxidant vitamins including gamma-tocopherol in early Parkinson’s disease: A randomized, double-blind, placebo-controlled trial.
Authors: Pantzaris M, Loukaides G, Paraskevis D, Kostaki EG, Patrikios I.
Journal: Clin Neurol Neurosurg. 2021 Sep 17;210:106954.
PMID: 34607196
In this study, the researchers conducted a small randomized, double-blind, placebo-controlled study of a product called Neuroaspis PLP10™ (which is a solution that contains omega-3 & omega-6 fatty acids and a bunch of vitamins) in individuals with recently diagnosed Parkinson’s.
What are omega-3 and omega-6 fatty acids?
About 90% of your dietary fats come in the form of triglycerides. These are a type of lipid (an organic compound that is insoluble in water) that are used by the body as a means of long term energy storage.
Triglycerides are made up of a glycerol backbone and three fatty acid chains:
 Source: Lumenlearning
Source: Lumenlearning
When you zoom in on the fatty acid chains, you find that they are made up of a chain of carbon atoms. And each carbon atom has a number of hydrogen atoms attached to it – the exact number of hydrogen atoms on each carbon determines whether the fat is saturated or unsaturated:
 Source: Expii
Source: Expii
If the fatty acid chain contains the maximum level of hydrogen atoms possible, it is considered a saturated fatty acid. By comparison, in unsaturated fatty acids some of the hydrogen atoms are missing. These are replaced with double bonds between the carbon atoms.
Both omega-3 and omega-6 are families of fatty acids.
More specifically, they are families of polyunsaturated fatty acids.
What does polyunsaturated mean?
A fat can be defined as “monounsaturated fatty acids” (or MUFA) if there is one double bond in the fatty acid chain. An example of this is oleic acid (as shown in the image below). “Polyunsaturated fatty acids” (or PUFA) refers to a fat that has two or more double bonds (such as linoleic acid):
 Source: Zoeharcombe
Source: Zoeharcombe
And as I said, both omega-3 and omega-6 are families of polyunsaturated fatty acids.
You said omega-3 and omega-6 are “families” of fatty acids. What do you mean by families?
Omega-3 and omega-6 are not singular fatty acids, but rather they each represent groups of them (all group being characterised by the number of carbon atoms before the double bond – see image below where I have place red dots of the left hand end of each fatty acid to illustrate the carbon atoms before the double bond):
 Source: Researchgate
Source: Researchgate
As you can hopefully see, there are different types of omega-3 and omega-6 fatty acids.
Where does the “3” in omega-3 come from?
The number in omega refers to the number of the carbon atom at which the first double bond occurs, and this relates to the “omega,” or tail end of the molecular chain. For omega 3 fatty acids, the third carbon atom is where the first double bond occurs, and for omega 6… you get the idea:
 Source: Eufic
Source: Eufic
I’ve also heard of trans fatty acids. Out of curiosity, what is meant by “trans” fatty acids?
As we have seen above, fatty acids differ significantly in terms of shape and structure. There are two additional categories are referred to as ‘cis ’ and ‘trans ’.
Trans fatty acids are unsaturated fatty acids that contain at least one double bond in the trans configuration:
 Source: PMC
Source: PMC
Trans fatty acids are industrially-produced fats, which are created by a process called hydrogenation. This helps to stabilise polyunsaturated oils to keep them solid at room temperature and giving them a longer shelf life.
 Source: Megalac
Source: Megalac
Trans fatty acids are produced naturally in the body (in the rumen) and those are generally considered beneficial, but the industrially-produced variety are believed to be detrimental to health.
Interesting. What exactly do the omega-3 and omega-6 fatty acids do in the body?
They are important components of cell membranes and are precursors to many other substances in the body such as those involved in regulating blood pressure and inflammatory responses.
Three omega−3 fatty acids are important in human biology:
- α-linolenic acid (ALA) – mainly in plant oils such as flaxseed, soybean, and canola oils. It is necessary for normal human growth and development
- Eicosapentaenoic acid (EPA) – found in fish oil (like DHA), it is known to have anti-inflammatory properties
- Docosahexaenoic acid (DHA) – the most abundant omega-3 fatty acid in the brain and retina
ALA is called an “essential” fatty acid. This is because your body cannot make it by itself, so it is essential that you get it from the food you consume. Your body can convert ALA into EPA and then to DHA – but this only generates very small amounts so it is important that you get EPA and DHA from foods as well.
- Linoleic acid (LA) – mostly found in plant oils, this fatty acid is used in the biosynthesis of cell membranes.
- Arachidonic Acid (AA) – the starting material for many inflammatory mediators
- dihomo-γ-linolenic acid (DGLA) – an important constituent of neuronal membrane
An important thing to understand is that omega-3 and omega-6 fatty acids are competitively metabolized by the same set of enzymes in our bodies. Human beings have evolved to survive and thrive on a diet with a basic ratio of omega-6 to omega-3 fatty acids of approximately 1:1.
Unfortunately, the modern Western diet has distorted that ratio somewhat, with many of us consuming a ratio of 15:1.
Basically, we consume way too much food with omega-6 fatty acids.
Omega 6 fatty acids are necessary for our bodies, but they are generally associated with their pro-inflammatory properties by health-oriented websites.
A careful balance of omega-6 and omega-3 fatty acids is needed for a healthy body.
|
# RECAP #1: Omega-6 and omega-3 are families of polyunsaturated fatty acids that are necessary for a healthy body and mind. Omega 3 fatty acids are generally considered anti-inflammatory, while omega 6 are viewed as inflammatory. Both have a wide variety of additional functions, and a balanced diet of both is required. The modern Western diet, however, is heavily skewed towards omega 6 fatty acids. # |
Ok, so what were the results of the clinical study?
In their study, the researchers recruited 40 individuals with recently diagnosed Parkinson’s, and blindly randomised them (at a ration of 1:1) to receive either 20 ml dose of a control solution (pure virgin olive oil) or their product Neuroaspis PLP10™. They blindly took their treatment once per day for 30 months.
What is Neuroaspis PLP10™?
Neuroaspis PLP10™ is a solution formula “containing a mixture of omega-3 (810 mg Eicosapentaenoic acid and 4140 mg Docosahexaenoic acid) and omega-6 fatty acids (1800 mg gamma-Linolenic acid and 3150 mg Linoleic acid) (1:1 w/w), with 0.6 mg vitamin A, vitamin E (22 mg) plus pure gamma (γ)-tocopherol (760 mg)” (Source). It is manufactured and sold by PALUPA Medical Ltd, in Cyprus.
NOTE: Again, please remember that this is not an advertisement or endorsement of this product. In addition, it is also important to note that some of the researchers who conducted this study are co-founders of PALUPA Medical Ltd.
The two randomly assigned groups in the study were evenly matched (average age: 65.5 vs 66.5 years) and there were no significant differences between them in terms of stage of Parkinson’s or level of daily PD treatments. The participants were clinically assessed every 6 months for the duration of the entire study (30 months).
What were the results of the study?
The results of the study demonstrate that over the 30 months of treatment, the Neuroaspis PLP10™ treated group exhibited no progression in their motor symptoms (as measured by the Unified Parkinson’s Disease Rating Scale (UPDRS) Part III). As you can see in the numbers below, the scores of the Neuroaspis PLP10™ treated group remain relatively constant (vs the increasing scores of the placebo-treated group):
- At baseline : 10.6 (3.9) vs 12.0 (4.2) p=0.209
- At 6 months : 10.4 (3.7) vs 12.2 (3.8) p=0.097
- At 12 months: 10.5 (3.7) vs 13.8 (3.6) p=0.004
- At 18 months: 10.9 (3.4) vs 15.1 (3.3) p=<0.001
- At 24 months: 11.3 (3.3) vs 16.5 (3.1) p=<0.001
- At 30 months: 11.7 (3.2) vs 17.3 (2.8) p=<0.001
The scores of the Neuroaspis PLP10™ treated group remained around 10 – 11, while the placebo treated group progressed from 12 – 17, indicating a worsening of motor symptoms over the 30 month trial.
The researchers also found a statistically significant increase of the daily intake of Levodopa treatment within the placebo-treated group compared to the Neuroaspis PLP10™-treated group. This effect was evident at the 18 month time point and remained to the end of the 30 months study:
Daily Levodopa Dosage (mean, SD) for the Neuroaspis PLP10™-treated group vs the placebo-treated group:
- At baseline : 422.5 (158.5) vs 442.5 (144.4) p=0.6790
- At 6 months : 422.5 (158.5) vs 442.5 (144.4) p=0.6790
- At 12 months : 435.0 (152.3) vs 505.0 (142.3) p=0.1413
- At 18 months: 447.5 (150.9) vs 570.0 (135.1) p=0.0102
- At 24 months: 477.5 (144.6) vs 647.5 (144.6) p=0.0006
- At 30 months: 515.0 (139.6) vs 700.0 (159.8) p=0.0004
There wasn’t much difference between dopamine agonists or MAO-B inhibitors treatments across the study, but this was only a small study of 40 participants.
Unfortunately, the results section is rather brief and there is no mention of any results in terms of non-motor features, no measures of adverse events (side effects, etc), and nothing is written about the safety or tolerability of the treatment in people with Parkinson’s – which is disappointing.
But the effect on motor symptoms is encouraging.
Given that the co-founders of the company that makes Neuroaspis PLP10™ conducted this study, it will be even more encouraging to see if the effect can be independently replicated.
|
# # RECAP #2: A clinical study involving 40 participants found that treatment with an omega-3 and omega-6 fatty acid rich solution resulted in a stabilisation of motor symptoms during a 30 month treatment period. In addition, no increase in levodopa treatment was observed in the treatment group (compared to the placebo-treated group). Independent replication of this finding is required. # # |
You sound skeptical. Are you?
The job of a scientist is to question everything (for example, “Could some other factor explain a particular set of results?”, “What happens if I add chemical A to condition B?”, and “Why do birds suddenly appear, every time I am near?”).
My first boss in science said it best:
“9 times out of 10 in science, the result is going to be negative, but it is that one time where the experiment works… that you should actually really worry, because statistically speaking, it should have failed”
I would be remiss if I did not question a set of results, and I am always seeking independent replication of new findings. It is a critical step in the validation of new discoveries.
And it is only natural to question positive clinical trial results for Parkinson’s when we have gone for such a long period of time with only negative outcomes. Remember, everything that we have previously thrown at this condition has not made an impact (and yes, exenatide might be an except to this statement), so the first question to ask when confronted with a positive finding is “what is different this time?”
Importantly, this particular result – as encouraging as it is – might not have attracted my attention, if it wasn’t for the fact that this treatment has already demonstrated interesting properties in previous clinical studies.
Wait. What?
In 2013, this report was published:
 Title: A novel oral nutraceutical formula of omega-3 and omega-6 fatty acids with vitamins (PLP10) in relapsing remitting multiple sclerosis: a randomised, double-blind, placebo-controlled proof-of-concept clinical trial.
Title: A novel oral nutraceutical formula of omega-3 and omega-6 fatty acids with vitamins (PLP10) in relapsing remitting multiple sclerosis: a randomised, double-blind, placebo-controlled proof-of-concept clinical trial.
Authors: Pantzaris MC, Loukaides GN, Ntzani EE, Patrikios IS.
Journal: BMJ Open. 2013 Apr 17;3(4):e002170.
PMID: 23599375 (This report is OPEN ACCESS if you would like to read it)
In this study, the investigators (and yes, the co-founders of the company were among the authors) conducted a 30-month randomised, double-blind, placebo-controlled, parallel design, Phase II proof-of-concept clinical study. They recruited 80 participants with relapsing–remitting multiple sclerosis.
Remind me, what is multiple sclerosis?
Multiple sclerosis (or MS) is a chronic neuroimmunologic condition in which the immune system starts to attack the insulating protective covering (or myelin) on the branches (or axons) of neurons. As that myelin is lost, a range of symptoms start to show themselves. Symptoms can include double vision, blindness in one eye, muscle weakness and trouble with sensation or coordination.
Like Parkinson’s, there is no cure for MS.
This video explains MS:
The 80 participants in the study were randomised them into four groups (20 people in each group). The groups were administered one of four treatments that were taken daily 30 min before dinner for 30 months:
- A solution with omega-3 fatty acids and omega-6 fatty acids (at 1:1 ratio). Specifically, the omega-3 fatty acids were DHA and EPA at a 3:1 ratio, and the omega-6 fatty acids were LA and DGLA at a 2:1 ratio. Vitamin A and vitamin E (α-tocopherol) were also included.
- The same as #1 with the addition of γ-tocopherol (form of vitamin E) – NOTE: This is what would go on to become “Neuroaspis PLP10™”
- A solution of γ-tocopherol alone.
- The placebo treatment.
Of the 80 participants to start this study, only 41 (51%) completed the full 30-months of the study – the majority discontinued due to the taste of the treatments. But of these, the participants (n=10) treated with Neuroaspis PLP10™ experienced only eight relapses versus 25 relapses in the placebo-treated group (n=12). This represented a 64% reduction for the Neuroaspis PLP10™-treated group.
In addition, the Neuroaspis PLP10™ slowed progression of the condition, as indicated in this graph (Neuroaspis PLP10™ is the green line, placebo is the red line):
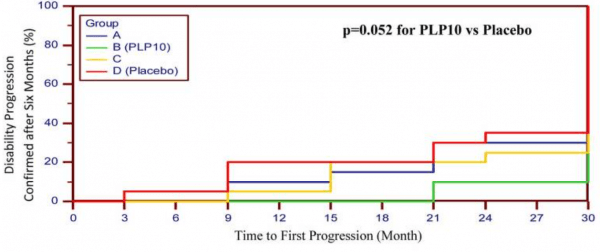 Source: PMC
Source: PMC
Encouraged by these results, the researchers set up the MINERAL study, which is a Phase III clinical study that will recruit 200 people with relapsing-remitting multiple sclerosis and treat them with either Neuroaspis PLP10™ or placebo once a day for 30 months. We are now waiting for the results of that larger study.
And this was not the only clinical study evaluating Neuroaspis PLP10™.
In 2020, this report was published:
 Title: The Effects of a 6-Month High Dose Omega-3 and Omega-6 Polyunsaturated Fatty Acids and Antioxidant Vitamins Supplementation on Cognitive Function and Functional Capacity in Older Adults with Mild Cognitive Impairment.
Title: The Effects of a 6-Month High Dose Omega-3 and Omega-6 Polyunsaturated Fatty Acids and Antioxidant Vitamins Supplementation on Cognitive Function and Functional Capacity in Older Adults with Mild Cognitive Impairment.
Authors: Stavrinou PS, Andreou E, Aphamis G, Pantzaris M, Ioannou M, Patrikios IS, Giannaki CD.
Journal: Nutrients. 2020 Jan 26;12(2):325.
PMID: 31991898 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers (and yes, again – full disclosure – the co-founders of the company were among the authors) conducted a 6 month randomized, double-blind, placebo-controlled study testing Neuroaspis PLP10™ in 56 elderly adults with mild cognitive impairment (average age: 78.8 years). Participants were randomly (1:1 ratio) assigned to either once daily Neuroaspis PLP10™ or placebo treatment.
The results indicated a significant improvement in the Neuroaspis PLP10™-treated group (compared to the placebo-treated group) at 6 months, based on the Addenbrooke’s Cognitive Examination-Revised (ACE-R), Mini-Mental State Examination (MMSE) and Stroop Color and Word Tests. In the graph below, you can see that the majority of the Neuroaspis PLP10™-treated group improved by almost 10 points in the ACE-R test:
 Source: PMC
Source: PMC
The researchers concluded that this “nutritional modality could be promising for reducing cognitive and functional decline in the elderly with mild cognitive impairment”
Both the mild cognitive impairment and multiple sclerosis studies present a lot more data than is presented in the Parkinson’s study which is disappointing as one feels that we are not getting a full picture of the study results. But the fact that this omega 3/omega 6 solution is having an impact across these conditions represents a compelling justification for further investigation.
Interesting. So what does it all mean?
Here are the SoPD, I am reluctant to write about products that are sold online as therapies for Parkinson’s, particularly when there is little (or no) research backing them. I especially hate the ones that have “testimonials” – that simply says to me that the manufacturer of the product can’t even be bothered to do a bit of research, and would rather rely on reviews from customers to sell their un-validated product (I don’t know about you, but online reviews carry zero weight with me).
Every now and then, however, we come across some research that indicates that there may just be something of interest with a particular product and one feels compelled to share it with the Parkinson’s community. The determining factor is that the research is that it is peer-reviewed and published in solid scientific journals. I have done this before with other SoPD posts – for example the “Wim Hof method” post (Click here to read that post).
When I read the results of a 30 month clinical trial indicating that a solution containing omega-3 and omega-6 fatty acids had a significant positive impact on the progression of motor symptoms in a group of individuals with Parkinson’s, I was made curious. The study was randomised and double-blinded, meaning that no one involved in the study had any idea as to who was being treated with the investigational agent or the placebo treatment. And while the results are limited in description, I dug a little further and found additional the studies in mild cognitive impairment and multiple sclerosis.
If all of these findings can be independently replicated – the current studies have been conducted by the co-founders of the company that produces the product – then further studies on the biology of omega-3 and omega-6 fatty acids in the context of Parkinson’s would obviously be warranted.
I guess this means we need to see an independent replication then.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: Neuroaspis PLP 10™ is a product that is produced and promoted by a commercial company called PALUPA Medical Ltd. At the time of writing, the author of this blog has had no contact with any individuals associated with that company or connected parties, nor has he benefited in any way from writing this post. This post does not represent an advertisement or endorsement of the product. The author was simply curious about the recently published results of the clinical study in Parkinson’s and is sharing it here in order to generate discussion.
The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from eufic

Studies in PD and MSA model mice that found that dietary DHA increased pathology. Does this indicate a problem with the models?
review article: https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC6099649/
LikeLike
Hi Rhyothemis,
Thanks for your message. Yes, there are studies that indicate negative consequences for PUFAs like DHA (for example: https://pubmed.ncbi.nlm.nih.gov/31686426), but DHA has also been reported to have benefits in clinical trials of Spinocerebellar Ataxia (https://pubmed.ncbi.nlm.nih.gov/30862453/). Maybe it is an issue with the models, I am not sure. I am also not promoting DHA either. This post was simply to initiate a discussion about an interesting clinical result. As I said several times in the post, I think independent validation is required now.
Kind regards,
Simon
LikeLiked by 1 person
I understand and thank you for blogging on this important topic. I really hope there is more investigation on this as it is quite puzzling and seems like it would be crucial to know if the models are flawed with respect to lipids.
LikeLike
There was another trial of omega-3 and vitamin E in PD published in 2018 that found benefit; article is paywalled:https://www.sciencedirect.com/science/article/abs/pii/S0303846718304633
It was reported on in this news article:https://parkinsonsnewstoday.com/2018/12/19/dietary-supplements-control-brain-inflammation-metabolism-parkinsons-study/
According to the news article the omega-3 was from flax oil which is mostly ALA; I have read that men and post-menopausal women don’t convert ALA to DHA very efficiently. The article doesn’t mention if flax lignans were present in the supplement or what form of vitamin E was used.
LikeLiked by 1 person
This is a great find – after three months with simple omega-3 supplementation from flaxseed oil, test subjects did almost as well after three months as the test subjects in the Neuroaspis study did after 30 months – 5 points net improvement in UPDRS part III versus 5.5 points for Neuroaspis. Average age of the participants was 66, so any deficiencies in the conversion of ALA to DHA by the elderly is accounted for in the results. From the omega-3 study:
https://pubmed.ncbi.nlm.nih.gov/28342967/
“Subjects were randomly allocated into two groups to take either 1000 mg of omega-3 fatty acids from flaxseed oil plus 400 IU of vitamin E supplements or placebo (n = 20 each group) for 12 weeks. Subjects were requested to consume supplements or placebo after lunch.”
From the omega-3 study fulltext:
UPDRS part III
Change
test arm −1.9 ± 5.5
placebo arm 3.1 ± 9.8
net difference 5.0
p = .05
UPDRS total
Change
test arm −3.4 ± 8.1
placebo arm 7.1 ± 14.6
net difference 10.4
p = 0.008
I am a bit concerned that the placebo arm showed so much progression after only three months. That seems excessive. Be that as it may, this is a very encouraging result.
According to the following study omega-3 fatty acids constitute half of flaxseed oil. It is a simple matter to consume a couple grams of flaxseed oil daily. Does including some Omega-6 and some Omega-9 in whole flaxseed oil make a difference? Does not seem like it should since this is already present in foodstuffs.
Complete fatty acid analysis data of flaxseed oil using GC-FID method
https://www.sciencedirect.com/science/article/pii/S2352340919301969
Sum of Omega-3 (n-3) ω3FA 51.376 %
Sum of Omega-6 (n-6) ω6FA 15.801 %
Sum of Omega-9 (n-9) ω9FA 20.591 %
Will make a related post at HU in due course.
LikeLike
Sounds like a combination of fish oil, borage oil and wheat germ oil. Borage / starflower is supposed to have anti-inflammatory effects as well as traditionally being used at menopause. Wheat germ has all sorts of good stuff and high vit E.
Eat more fish and put a bit of each of the last 2 ingredients as salad oil or added to granola. They taste good. Much better than popping more pills or slurping oil. Can’t do any harm!
LikeLike
How might omega fas work? In combination with low dose aspirin they generate resolvins that ‘resolve’ inflammation. https://www.frontiersin.org/articles/10.3389/fphar.2020.00612/full
I take EPA+DHA daily, 75 mg aspirin ca weekly. Fingers crossed.
LikeLike
Static stretching also promotes resolvin production. I got rid of sciatica and plantar fasciitis with static stretching. Helene Langevin has some talks on YouTube about it.
LikeLike
Thank you for this good article.
fyi – just explored it and found a company’s homepage in Germany. However, when clicking on it my Kasperksy security programm stopped a download – because it has detected malicious adware which could harm my computer or data.
Not really inspiring confidence….
LikeLike
Thanks Simon,
In case you’ve missed it, the results of the Phase 3 MINERAL study looking at the Omega-3 combination in MS patients were published late 2023 and are online here – again, some promising findings that give hope:
https://pubmed.ncbi.nlm.nih.gov/36353267/
LikeLike