|
# # # # Cellular activity generates a lot of waste and by-products. Cells have developed very efficient methods of dealing with this situation. As we age, however, these processes become strained, and in degenerative conditions they appear to be rather dysfunctional. New research highlights a novel mechanism – Bach1 derepression – which points towards a new class of potential therapeutics and interesting avenues of further study. In today’s post, we will discuss the results of this new research and explore the implications of it. # # # # |
Source: Maxpixel
I am marveling at the fact that I am typing these words.
And that you are reading them.
Consider for a moment the requirements of this arrangement. And I’m not talking about the tiny muscles changing the size of the pupil in your eye, or the neurons in your visual cortex firing in unison to give you a correct and colour-rich representation of the world in front of you that has nothing to do with the actual content being observed.
Rather, I’m thinking more about about what is going on one level down – actually inside of each cell:
 A liver cell. Source: Muhadharaty
A liver cell. Source: Muhadharaty
There is a universe of frenzied molecular activity in each and every cell of our bodies. And we are only just starting to build up a user guide to the densely packed, fuzzy complexity of this inner world. This video gives an extremely simplified version of some of what is going on (in reality, the interior of cells is significantly more densely packed and the activity is a vastly quicker):
And as I suggested above it should be celebrated that what occurs in these cells is so rapid, efficient and precise that I can type these words and you can read them.
All of this crazy activity, however, produces waste and by-products.
Cells have of course developed very effective means of dealing with those issues. But as we age, cells can start to struggle with the task of waste disposal. And as a result, we can start to see an accumulation of these by-products, which can lead to stress on the cell, particularly in the form of oxidative stress.
What is oxidative stress?
Oxidation is the loss of electrons from a molecule, which in turn destabilises that particular molecule.
Think of iron rusting.
Rust is the oxidation of iron – in the presence of oxygen and water, iron molecules will lose electrons over time. Given enough time, this results in the complete break down of objects made of iron.
 Rusting iron. Source: Thoughtco
Rusting iron. Source: Thoughtco
The exact same process occurs in biology. Molecules in your body go through a similar process of oxidation – losing electrons and becoming unstable. This chemical reaction leads to the production of what we call free radicals.
What are free radicals?
A free radical is an atom, molecule, or ion that has at least one unpaired electron. In effect, it is an extremely unstable entity.
 Source: Mercordianimalcare
Source: Mercordianimalcare
Due to the lack of a stable number of electrons, free radicals are in a constant state of searching for another electron to help stabilise themselves. This chaotic activity of these unstable atoms can result in damage to cells – particularly the all-important DNA – and can ultimately lead to cell death.
What can cause the production of free radicals?
Lots of things.
They can be produced naturally in the body as a by-product of metabolism, or by exposure to environmental toxins such as tobacco smoke or ultraviolet light.
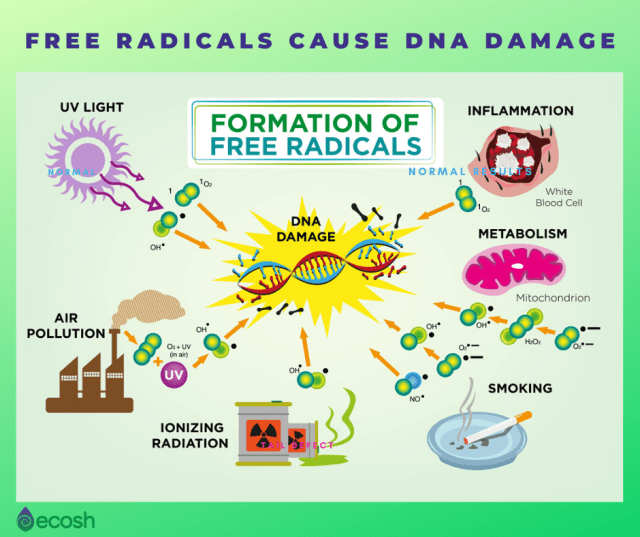 Source: Ecosh
Source: Ecosh
One very common type of free radicals are reactive oxygen species (or ROS).
What are reactive oxygen species?
ROS is an umbrella term for an large array of derivatives of molecular oxygen that occur through the normal cellular functioning of cells. When oxygen (O2) loses an electron, it results in “superoxide”.
 Source: Wikipedia
Source: Wikipedia
And just because superoxide has the word ‘super’ in it, does not mean that it is one of the good guys. Superoxide is the precursor to most other ROS.
The generation of ROS inside of cells largely occurs in the mitochondria (the power stations of cell):
 A mitochondrion (singular) and its location in the cell. Source: NCBI
A mitochondrion (singular) and its location in the cell. Source: NCBI
ROS often leak out of a structure inside of mitochondria called the electron transport chain. It is located on the inner wall of the mitochondrial membrane.
What is the electron transport chain?
The electron transport chain is a series of protein complexes that produces adenosine triphosphate (or ATP), which is the energy-carrying molecule found in the cells of all living things. ATP provides energy to drive many processes in living cells.
 Source: Youtube
Source: Youtube
An explanation of the electron transport chain requires an entire website of its own. This video, however, does an excellent good job of providing an overview:
The electron transport chain is amazing, but it is not a perfect system. And its activities result in the production of ROS, which can leak out of the mitochondria and cause trouble in cells.
Because mitochondria produce so many ROS from their continual production of energy, our cells have developed an assortment of defense systems to clean up ROS and maintain a stable environment.
One common defense is the production of antioxidants.
What is an antioxidant?
While free radicals are the bad guys in oxidation, antioxidants can be considered the good guys. They are molecules that neutralize the free radicals by donating one of their own electrons. The antioxidant do not become free radicals by donating an electron because by their very nature they are stable with or without that extra electron.
 How free radicals and antioxidants work. Source: h2miraclewater
How free radicals and antioxidants work. Source: h2miraclewater
How do our cells produce antioxidants?
There are different ways that our bodies can produce antioxidants and they can also be absorbed from our diet. One of the most well studied methods of antioxidant production that cells use involves the Nuclear Factor Erythroid 2-Related Factor 2 (or NRF2) pathway.
What is the NRF2 pathway?
NRF2 is a ‘transcription factor’ with some interesting properties. Transcription factors are proteins that are involved in the process of converting (or transcribing) DNA into RNA.
A transcription factor can be an ‘activator’ of transcription – that is initiating or helping the process of generating RNA from DNA.

An example of a transcriptional activator. Source: Khan Academy
Or it can be a repressor of transcription – blocking the machinery (required for generating RNA) from doing it’s work.

An example of a transcriptional repressor. Source: Khan Academy
NRF2 is an activator of transcription. When it binds to DNA to aids in the production of RNA, which then results in specific proteins being produced.
And this is where NRF2 gets interesting.
You see, NRF2 binds to antioxidant response elements (or ARE).
What are ARE?
Genes are sections of DNA that provide the instructions for making a particular protein or enzyme. The regulatory region of a gene is where the proteins that activate (like transcription factors) or modulate the transcription of a gene bind to.
Antioxidant response elements (or ARE) are particularly interesting because these are regions of DNA that reside within the regulatory regions of genes that encode naturally occurring antioxidant and protective proteins. And given that antioxidants and protective proteins are generally considered a good thing for sick/dying cells, you can see why the NRF2 pathway is an interesting topic of investigation.
By binding to the ARE region of antioxidant-related genes, NRF2 is directly encouraging the production of naturally occurring antioxidant and protective proteins.
 Source: Liebertpub
Source: Liebertpub
|
# RECAP #1: Antioxidants are a natural means for the body to combat the negative effects of oxidative agents, like free radicals. Free radicals can be caused by environmental stressors, but they can also result of normal cellular metabolism. The NRF2 pathway is a cellular mechanism for producing antioxidants. The NRF2 protein binds to the regulatory region of antioxidant genes and initiates their transcription. # |
This is all very interesting, but what does it have to do with Parkinson’s?
Well, an interesting preclinical study was recently published.
This is the research report here:
 Title: Bach1 derepression is neuroprotective in a mouse model of Parkinson’s disease
Title: Bach1 derepression is neuroprotective in a mouse model of Parkinson’s disease
Authors:
Journal: Proc Natl Acad Sci U S A. 2021 Nov 9;118(45):e2111643118.
PMID: 34737234
In this study, the researchers found something rather intriguing was occurring in the Parkinson’s brain: Levels of a protein called Bach1 were elevated in vulnerable regions of the brain.
 BACH1 protein structure. Source: Wikipedia
BACH1 protein structure. Source: Wikipedia
Why is that so interesting? What does Bach1 do?
Bach1 is a repressor of NRF2.
What does that mean?
Bach1 is a handbrake on NRF2 activity. It binds to the same ARE regions that NRF2 targets and by doing this, it inhibits NRF2’s ability to initiate transcription of those target genes.
Therefore, if there are higher levels of Bach1 in the vulnerable regions of the Parkinson’s brain (such as the substantia nigra), then the NRF2 protein is going to have a hard time increasing the production of antioxidants in those areas.
Got it. So the PD brains have high levels of this Bach1 handbrake. What did the researchers do next?
Next they decided to have a look at what Bach1 was doing in the brains of Parkinson’s animal models. They found that levels of the protein were also elevated in these brains as well.
In mice treated with the neurotoxin MPTP (a chemical that kills off dopamine neurons in the substantia nigra), the investigators found raised levels of Bach1. From 2 hours post delivery of the toxin out to 7 days afterwards, Bach1 levels were significantly elevated.
 Next, the researchers asked themselves “what would happen if we removed Bach1 in this model of Parkinson’s?” and they genetically engineered mice that produce no Bach1 to test this question. They treated these no-Bach1 mice with either the neurotoxin or saline (a harmless control solution), and they compared these mice to normal mice (with normal Bach1) that were treated with the same regime.
Next, the researchers asked themselves “what would happen if we removed Bach1 in this model of Parkinson’s?” and they genetically engineered mice that produce no Bach1 to test this question. They treated these no-Bach1 mice with either the neurotoxin or saline (a harmless control solution), and they compared these mice to normal mice (with normal Bach1) that were treated with the same regime.
They found that the mice with no Bach1 had significantly better survival of their dopamine neurons compared to the normal mice, when treated with the neurotoxin. They also found that the deletion of Bach1 resulted in less markers of oxidative stress and inflammation (the signaling of the immune system that something is wrong) in the brain.
These results suggested a crucial role of Bach1 in mediating the neurotoxic effects and pointed towards a novel target potential for potential pharmacological intervention.
Good stuff. Is there a drug to block Bach1?
A biotech company called vTv Therapeutics has been developing Bach1 inhibitors.
 We have previously mentioned this biotech company on the SoPD, as they have also been developing RAGE inhibitors (Click here to read that post).
We have previously mentioned this biotech company on the SoPD, as they have also been developing RAGE inhibitors (Click here to read that post).
They have a Bach1 inhibitor in clinical testing for renal indications at the moment. In December 2020, the company licensed worldwide rights to develop and commercialize that inhibitor (called HPP971) to Anteris Biopharma.
I am not sure that this particular Bach1 inhibitor (HPP971) can cross the blood brain barrier (the protective membrane surrounding the brain), so it may not be much use for neurodegenerative conditions like Parkinson’s.
But researchers from vtv Therapeutics were involved in the research report we are discussing today and they presented a new Bach1 inhibitor that can access the brain.
When the investigators tested this CNS-penetrant Bach1 inhibitor in normal mice treated with the neurotoxin MPTP, they found that it could reduce the dopamine neuron degeneration and associated oxidative stress and neuroinflammation.
Remarkably, even when the treatment of the Bach1 inhibitor was delayed until 6 days after the neurotoxin was administered, the agent could still significantly protect the dopamine neurons against the MPTP-induced neurotoxicity.
The researchers concluded their study by suggesting the Bach1 inhibitors could represent a “promising therapeutic approach for Parkinson’s”.
|
# # RECAP #2: Researchers have recently discovered that Bach1 (a protein that blocks the actions of NRF2) is elevated in the PD brain. They also found that genetic deletion of Bach1 protected a mouse model of Parkinson’s. The researchers screened a library of molecules to identify agents that could inhibit Bach1 and they tested them successfully in models of Parkinson’s. # # |
Has anyone else ever reported this?
No, this result is rather novel for Parkinson’s research, so we will have to wait and see if independent replication is possible.
But the results do potentially tackle an intriguing problem that has confronted the Parkinson’s research field around NRF2 biology.
What is that problem?
Well, everyone has been really excited about the idea of targeting the NRF2 pathway in Parkinson’s for a long time, but then in 2020 this report was published:
 Title: Systemic activation of Nrf2 pathway in Parkinson’s disease.
Title: Systemic activation of Nrf2 pathway in Parkinson’s disease.
Authors: Petrillo S, Schirinzi T, Di Lazzaro G, D’Amico J, Colona VL, Bertini E, Pierantozzi M, Mari L, Mercuri NB, Piemonte F, Pisani A.
Journal: Mov Disord. 2020 Jan;35(1):180-184.
PMID: 31682033
In this study, the researchers found that in blood cells from people with Parkinson’s, there was systemic activation of the NRF2 pathway.
What does that mean?
It means that levels of NRF2 are high in people with Parkinson’s (and the proteins of its target genes are also elevated).
This result obviously begs the question: Should we really be increasing NRF2 levels further?
And this study indicating systemic activation of the NRF2 pathway is partly supported by an older report:
 Title: Expression of Nrf2 in neurodegenerative diseases.
Title: Expression of Nrf2 in neurodegenerative diseases.
Authors: Ramsey CP, Glass CA, Montgomery MB, Lindl KA, Ritson GP, Chia LA, Hamilton RL, Chu CT, Jordan-Sciutto KL.
Journal: J Neuropathol Exp Neurol. 2007 Jan;66(1):75-85.
PMID: 17204939 (This report is OPEN ACCESS if you would like to read it)
In this older study, the researchers analysed the postmortem brains of people who had passed away with different neurodegenerative conditions and noticed something interesting about NRF2 protein in Parkinson’s.
While NRF2 protein (stained brown in the images below) was only in the cytoplasm (the area inside of a cell, but not the nucleus) of neurons in normal, Alzheimer’s (AD) and Lewy body variant of Alzheimer disease (LBVAD) brains, in the neurons of the Parkinson’s (PD) brain, the NRF2 protein was also present inside the nucleus:
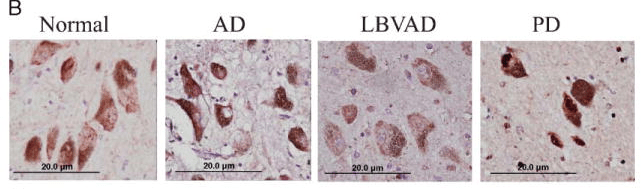 Source: PMC
Source: PMC
This finding indicated that NRF2 had translocated to the nucleus and presumably transcribing antioxidant genes. And “presumably” is the key word in that sentence, because this new Bach1 research report (reviewed in today’s post) suggests that while NRF2 may be elevated in PD, it might be limited in its function due to Bach1 blocking it.
It is a really intriguing thought.
And other researchers have already proposed that targeting Bach1 could be a useful therapeutic avenue in tackling the NRF2 pathway (Click here to read more about this).
It is also really intriguing to note that Bach1 is actually a target gene of NRF2 – increasing amounts of NRF2 protein will bind to the regulatory region of Bach1 and encourage transcription of it, therefore increasing levels of Bach1 protein (Click here to read more about this).
Has Bach1 inhibition ever worked in models of other neurodegenerative conditions?
Yes. In 2020, this report was published:
 Title: Isomeric O-methyl cannabidiolquinones with dual BACH1/NRF2 activity.
Title: Isomeric O-methyl cannabidiolquinones with dual BACH1/NRF2 activity.
Authors: Casares L, Unciti-Broceta JD, Prados ME, Caprioglio D, Mattoteia D, Higgins M, Apendino G, Dinkova-Kostova AT, Muñoz E, de la Vega L.
Journal: Redox Biol. 2020 Oct;37:101689.
PMID: 32863231 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers presented molecules that had dual functions: 1. Bach1 inhibition, and 2. NRF2 activation. They then demonstrated this effectiveness of these molecules in cellular models of the neurodegenerative condition of Huntington’s disease.
It will be interesting to see what further research stems next from these dual-mechanism agents as well as the vtv Therapeutics Bach1 inhibitors.
So what does it all mean?
We started off this post by discussing how I marvel at the fuzzy minute world of the interior of a cell, but equally I am astonished by the opposite end of the spectrum: For example, NASA’s Hubble telescope recently captured a supernova (exploding star) located in the spiral galaxy NGC 2525, which is 70 million light–years away from Earth:
Think about that for a moment.
Dinosaurs were still walking the Earth when that star actually exploded, and the light released from that event has been travelling to us across the intervening period of time and distance. It has only just arrived now.
It makes one feel rather small.
Small, but still complex.
Not complex enough, however, that problem inside us can’t be fixed or resolved. And that is where it is really stunning that we humans have evolved from hand paintings on cave walls to designing molecules that can find their way through our bodies, into the over-crowded labyrinth of intracellular space, and some how they hit their very specific biological targets.
And the targets get more and more interesting as we better understand the biology regulating specific medical conditions, like Parkinson’s. If the new results from the study reviewed in today’s post can be independently replicated, then Bach1 will be a really compelling target for pharmacological intervention. There have been many attempts made at activators of NRF2, but perhaps blocking the inhibitors of this protein could be a better approach for drug development.
Only time will tell.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, the biotech company vtv Therapeutics that is mentioned in this post is a publicly traded company. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. vtv Therapeutics has not requested that this material be produced, nor has the author had any recent contact with the company or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from me.


2 thoughts on “Turn back Bach?”