|
This week the ‘Michael J. Fox Foundation for Parkinson’s Research’ and ‘The Silverstein Foundation for Parkinson’s with GBA’ announced that they are collaboratively awarding nearly US$3 million in research grants to fund studies investigating an enzyme called beta glucocerebrosidase (or GCase). Why is this enzyme important to Parkinson’s? In today’s post, we will discuss what GCase does, how it is associated with Parkinson’s, and review what some of these projects will be exploring. |
 Source: DenisonMag
Source: DenisonMag
This is Jonathan Silverstein.
He is a General Partner of Global Private Equity at OrbiMed – the world’s largest fully dedicated healthcare fund manager. During his time at OrbiMed, the company has invested in healthcare companies that have been involved with over 60 FDA approved products.
In February 2017 – at just 49 years of age – Jonathan was diagnosed with Parkinson’s.
Rather than simply accepting this diagnosis, however, Mr Silverstein decided to apply the skills that he has built over a long and successful career in funding biotech technology, and in March 2017, he and his wife, Natalie, set up the Silverstein Foundation for Parkinson’s with GBA.
 The foundation has just one mission: “to actively pursue and invest in cutting edge research with the goal of discovering new therapies for the treatment of Parkinson’s Disease in GBA mutation carriers”
The foundation has just one mission: “to actively pursue and invest in cutting edge research with the goal of discovering new therapies for the treatment of Parkinson’s Disease in GBA mutation carriers”
And it seeks to address this by achieving three goals:
- to find a way to halt the progression of Parkinson’s with GBA.
- to identify regenerative approaches to replace the damaged/lost cells
- to find preventative measures
This week, the Silverstein foundation and the Michael J. Fox Foundation for Parkinson’s Research made a big anoouncement.
The two organisations announced nearly US$3 million in grants to fund studies investigating an enzyme called glucocerebrosidase beta acid (or GCase).
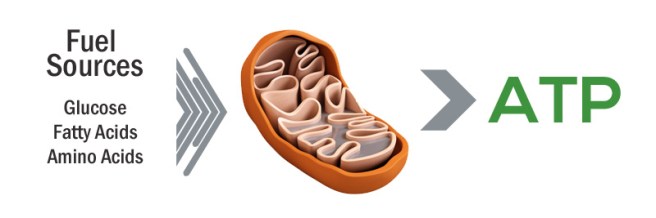
And what exactly is glucocerebrosidase?