
|
Two months ago a research report was published in the scientific journal ‘Nature’ and it caused a bit of a fuss in the embryonic stem cell world. Embryonic stem (ES) cells are currently being pushed towards the clinic as a possible source of cells for regenerative medicine. But this new report suggested that quite a few of the embryonic stem cells being tested may be carrying genetic variations that could be bad. Bad as in cancer bad. In this post, I will review the study and discuss what it means for cell transplantation therapy for Parkinson’s disease. |

Source: Medicalexpress
For folks in the stem cell field, the absolute go-to source for all things stem cell related is Prof Paul Knoepfler‘s blog “The Niche“. From the latest scientific research to exciting new stem cell biotech ventures (and even all of the regulatory changes being proposed in congress), Paul’s blog is a daily must read for anyone serious about stem cell research. He has his finger on the pulse and takes the whole field very, very seriously.

Prof Paul Knoepfler during his TED talk. Source: ipscell
For a long time now, Paul has been on a personal crusade. Like many others in the field (including yours truly), he has been expressing concern about the unsavoury practices of the growing direct-to-consumer, stem cell clinic industry. You may have seen him mentioned in the media regarding this topic (such as this article).
The real concern is that while much of the field is still experimental, many stem cell clinics are making grossly unsubstantiated claims to draw in customers. From exaggerated levels of successful outcomes (100% satisfaction rate?) all the way through to talking about clinical trials that simply do not exist. The industry is badly (read: barely) regulated which is ultimately putting patients at risk (one example: three patients were left blind after undergoing an unproven stem cell treatment – click here to read more on this).
While the stem cell research field fully understands and appreciates the desperate desire of the communities affected by various degenerative conditions, there has to be regulations and strict control standards that all practitioners must abide by. And first amongst any proposed standards should be that the therapy has been proven to be effective for a particular condition in independently audited double blind, placebo controlled trials. Until such proof is provided, the sellers of such products are simply preying on the desperation of the people seeking these types of procedures.
So stem cells are not safe then?
I am NOT saying that.
Cars are all safe in the right hands, but this is because the government has very strict guidelines with regards to which cars can be made available to the general public and then the government enforces laws with regards to who can drive them. Severe punishments can be handed down at all stages of this process.
So stem cells are safe then?
We’ll move on before this turns into a circular argument.
Just understand that great caution must be taken with this technology – by both patients and clinical practitioners. This is an important issue that is getting a lot of attention from the media – just this week alone the Guardian newspaper had an article on this topic.
And this issue of caution has recently been further emphasised by a research report that has left even the scientists in the field a bit staggered.
This is the report here:

Title: Human pluripotent stem cells recurrently acquire and expand dominant negative P53 mutations.
Authors: Merkle FT, Ghosh S, Kamitaki N, Mitchell J, Avior Y, Mello C, Kashin S, Mekhoubad S, Ilic D, Charlton M, Saphier G, Handsaker RE, Genovese G, Bar S, Benvenisty N, McCarroll SA, Eggan K.
Journal: Nature. 2017 May 11;545(7653):229-233.
PMID: 28445466
In the study, researchers at Harvard University asked scientists around the world to send them a batch of the human embryonic stem cells they are using for research. There are only a few dozen commonly used human embroynic stem cell lines and they have been shared with researchers all over the world. The Harvard researchers were seeking to determine whether the different lines of cells had acquired any genetic anomalies as they had been expanded by all these different research groups.
What are embryonic stem cells?
Embryonic stem cells have the potential to become any type of cell in the body – this means that they are ‘pluripotent’. Embryonic stem cells can be encouraged (using specific chemicals) to become whatever kind of cell you want them to be. This feature makes them particularly appealing to research scientists and clinicians who are interested in regenerative medicine (or replacing cells that are lost due to injury or disease).

Embryonic stem cells in a petridish. Source: Wikipedia
Embryonic stem cells are derived from a fertilised egg cell. The egg cell will divide, to become two cells, then four, eight, sixteen, etc. Gradually, it enters a stage called the ‘blastocyst’, which is a small ball of cells. Inside the blastocyst is a group of cell that are called the ‘inner stem cell mass’, and it is these cells that can be collected and used as embryonic stem cells.
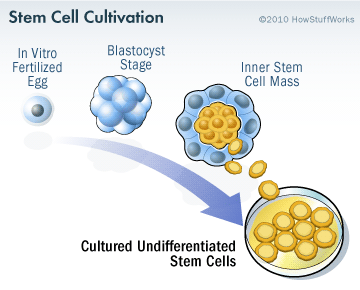
The process of attaining ES cells. Source: Howstuffworks
Ok, so what did the researchers find when they collected all of the ES cells from around the world?
The researcher collected 140 independent batches of human embryonic stem cells, (including 26 batches which have been prepared for potential clinical use) from 19 research institutes from around the world. They next sequenced the DNA of each of these batches and looked for genetic variations within and between the batches.
What they found kind of shocked them.
They identified 263 variations, of which 28 were predicted to have a damaging or disruptive effect on the function of the mutated gene.
Critically, they found six genetic mutations in the TP53 gene in five unrelated embryonic stem cell batches (including a clinically-prepared cell line – MShef10 – which actually carried two distinct TP53 variants – G245S and R248W).
What is TP53?
TP53 is a gene on chromosome 17 that encodes a protein that has three major functions: controlling cell division, DNA repair, and apoptosis (or cell death). p53 (the protein) performs these functions as a transcriptional activator (that is a protein that binds to DNA and helps produce RNA (the process of transcription) – see our previous post explaining this).

p53 protein structure, bound to DNA (in gold). Source: Wikipedia
In regulating the cell division, p53 prevents cells from dividing too much and in this role it is known as a tumour suppression – it suppresses the emergence of cancerous tumours. Genetic mutations in the p53 gene can result in run away cell division, and (surprise!) as many as 50% of all human tumours contain mutations in the p53 gene.
Read that again: As many as 50% of all human tumours contain mutations in the p53 gene. This is definitely not a gene that you want mutated in a cell that you are potentially injecting into someone’s body!

Cancer vs no cancer. Source: Khan Academy
With regards to DNA repair, p53 is sometimes called “the guardian of the genome” as it prevents mutations and helps to conserve stability in the genome. This function also serves to prevent the development of cancer, by helping to repair potentially cancer causing mutations….and in this role it is known as a tumour suppression. Obviously, if there is a mutation in the p53 gene, less DNA repair will occur – increasing the risk of cancer occurring.
AGAIN: This is definitely not a gene that you want mutated in a cell that you are going to inject it into someone’s body!
And finally, in cell death, p53 plays a critical role in telling a cell when to die. And (continuing with the cancer theme), if there is a mutation in the p53 gene, fewer cells will be told to die – increasing the risk of cancer occurring. And in this role p53 is known as a tumour suppression (are you picking up on the cancer theme yet?)
ONE LAST TIME: This is definitely not a gene that you want mutated in an ES cell that you are potentially injecting into someone’s body!
So TP53 mutations cause cancer?
Not necessarily. It depends on where the mutation is on the gene, but genetic variations in the gene certain increases the likelihood of cancer occurring. For example, individuals with Li–Fraumeni syndrome – an autosomal dominant disease caused by a TP53 mutation (at location R337H) – have a lifetime cancer risk of nearly 100%. That is to say, they are almost certainly going to develop cancer within their lifetime.
So the researchers should get rid of those batches of ES cells then, right?
Unfortunately, it’s not that easy.
You see, the Harvard group’s findings suggest that each of the TP53 mutations identified in the embryonic stem cells was an acquired mutation. That is to say, the cells gained the mutation as they were dividing in cell culture over time. Some batches of the same embryonic stem cells that are used by different research groups do not have the mutations.
And this is the real take home message of the report: The genetic variants have been picked up over time by the cells as they quietly divided in petri dishes. This suggests that these mutations could potentially occur in any embryonic stem cell being prepared for use in transplantation therapy.
And the cells with the mutation will gradually come to represent a significant fraction of cells in those affected batches, because they have a natural advantage in their ability to divide and survive over the other non-mutated cells.
So stem cell are not safe then?
I’m not saying that.
The point of this whole post is not to scare people away from the use of embryonic stem cells or any other kind of stem cells for use in regenerative medicine. Stem cells represent an amazing potential future for the field of medicine, in which cells could be grown in petri dishes and then used to replace/repair organs within the body.
I am merely trying to raise awareness about the potential consequences of using protocols and procedures that are not thoroughly tested and approved.
What is going to happen next then?
There are now efforts by various research groups around the world to devise procedures and protocols to limit the possibility of transplanting cells with undesirable genetic variants. For example, cells could have their DNA sequenced at the start of cell culture process and again just before those same cells are going to be transplanted and if any anomalies are found, the procedure could be postposed.
Has this been done before?
Yes it has.
Awareness of these sorts of issues has already resulted in one experimental surgery being stopped.

Nunobiki Herb Garden in Kobe. Source: Wikimedia
In March 2015, researchers at the RIKEN research institute in Kobe (Japan) suspended a clinical study of induced pluripotent stem (IPS) cells for a degenerative eye condition (age-related macular degeneration) because they found six different variations in the cells to be transplanted into the second patient involve in the study. Those variations were not present in the original cells that the researchers started with (IPS cells are stem cells that are derived from a patient’s own skin cells. I have previously written a post about IPS cells – click here to read that post).
But how often have tumours developed following stem cell procedures?
To be completely fair, not very often. But it has occurred. And I hope you will agree that more than once is once too often.
In the medical literature, there are at least two cases that I am aware of in which tumours developed following stem cell surgery. In 2009, this case report was published:

Title: Donor-derived brain tumor following neural stem cell transplantation in an ataxia telangiectasia patient.
Authors: Amariglio N, Hirshberg A, Scheithauer BW, Cohen Y, Loewenthal R, Trakhtenbrot L, Paz N, Koren-Michowitz M, Waldman D, Leider-Trejo L, Toren A, Constantini S, Rechavi G.
Journal: PLoS Med. 2009 Feb 17;6(2):e1000029. doi: 10.1371/journal.pmed.1000029.
PMID: 19226183 (This article is OPEN ACCESS if you would like to read it)
In this case report, a 9 year-old boy affected by an inherited neurodegenerative condition called ataxia telangiectasia, had been treated in Moscow with repeated transplantations of fetal neural stem cells. Four years later, the patient complained about headaches, and a brain scan investigation revealed a small tumour compressing the brain stem (BS and arrow in panel A of the image below). There were also signs of tumour growth further down the spinal cord (arrow in panel B of the image below).
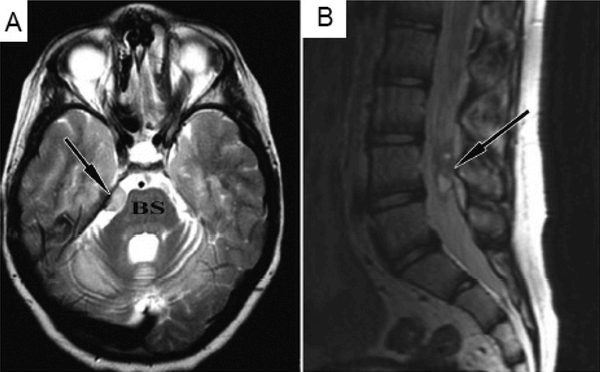
Source: Ncbi
Molecular analysis of biopsies from the tumours was performed, and the results led the investigators to conclude that the tumour is derived from the neural stem cells. Luckily the slow growth suggests a relatively benign tumour which the investigators hope suggests a favourable prognosis.
The researchers concluded that their “findings therefore do not imply that the research in stem cell therapeutics should be abandoned. They do, however, suggest that extensive research into the biology of stem cells and in-depth preclinical studies, especially of safety, should be pursued in order to maximize the potential benefits of regenerative medicine while minimizing the risks“.
That first report was followed up last year by another case study:

Title: Glioproliferative Lesion of the Spinal Cord as a Complication of “Stem-Cell Tourism”.
Authors: Berkowitz AL, Miller MB, Mir SA, Cagney D, Chavakula V, Guleria I, Aizer A, Ligon KL, Chi JH.
Journal: N Engl J Med. 2016 Jul 14;375(2):196-8. doi: 10.1056/NEJMc1600188. Epub 2016 Jun 22. No abstract available.
PMID: 27331440 (This article is OPEN ACCESS if you would like to read it)
In this case study, the researchers report about the case a 66-year-old man who had suffered a stroke. He had ventured to commercial stem-cell clinics in China, Argentina, and Mexico, and had had three different kinds of stem cells (mesenchymal, embryonic, and fetal neural stem cells) injected into his spinal canal.
Progressive lower back pain, impairment in motor or sensory function of the lower extremities, and urinary incontinence slowly developed following these procedures. Magnetic resonance imaging (MRI) revealed a series of small tumours in the spinal cord. Luckily for this individual, analysis of the tumours indicated no cancer associated genetic variants in the DNA of the tumour cells, and radiation therapy led to decreased back pain, improved mobility, and a decrease in the size of the tumours on MRI.
The investigators concluded by noting that “The unregulated commercial stem-cell industry is not only potentially harmful to individual patients but also undermines attempts to study stem-cell therapies in clinical trials“.
In both of these cases, the affected individuals have been lucky that the cancerous growths were not more aggressive. Indeed the field of stem cell transplantation research is lucky that the situations were not worse. But they reinforce the necessity for caution in ongoing clinical trials using embryonic stem cells (such as the new cell transplantation for Parkinson’s disease trial starting in China – click here to read more about this) or the controversial clinical trial being conducted in Australia (Click here for more on this). One has to wonder if the cells being used in those trials have been screened for genetic variants.
What does it all mean?
This post resulted from a discussion in the lab where I work. We were talking about the research report from the Harvard group and discussing various protocols/procedures that could be set up to reduce the risk of something bad being transplanted. With such implementations, the chances of a cancerous tumour would be further reduced.
Again, the point of this post is not to terrify everyone or argue against the use of stem cell therapy for neurodegenerative conditions. It was only supposed to make readers aware of the potential dangers if standards are slack (and in some of the direct-to-consumer stem cell clinic market, the standards are slack).
I did not want to appear melodramatic or too ‘over the top’ in writing this post, but it can be hard not to at times. I am very concerned about the current state of commercially available stem cell services, and I have grave fears about bad practices delaying the future developments in this exciting area.
FULL DISCLOSURE – The author of this blog is associated with research groups conducting the current Transeuro transplantation trials and the proposed G-Force embryonic stem cell trials planned for 2018. It is important for all readers of this post to appreciate that cell transplantation for Parkinson’s disease is still experimental. Anyone declaring otherwise (or selling a procedure based on this approach) should probably not be trusted. While I appreciate the desperate desire of the Parkinson’s community to treat the disease ‘by any means possible’, bad or poor outcomes for this technology could have serious consequences for the individuals receiving the procedure and negative ramifications for all future research in the stem cell transplantation area.
The banner for todays post was sourced from Psypost
Reading the news out of Japan today reminding me of this post. I view this as the endgame… Let’s hope we see some good news in the next couple years without reckless science forcing a regulatory clampdown.
LikeLike
Fingers crossed Double!
LikeLike