|
Chinese researchers recently published pre-clinical research demonstrating the use of their protocol for generating stem cell-derived neurons for cell transplantation in Parkinson’s. The data represents the last step/proof-of-principle stage for taking this procedure into clinical trials (which are ongoing). In today’s post, we will discuss what cell transplantation is, we will review the new data, and we will consider some of the issues associated with taking this procedure to the clinic. |
Brain surgery. Source Bionews-tx
As we have discussed before, any ‘cure’ for Parkinson’s requires 3 components:
- A disease halting mechanism – slowing or stopping the progression of the condition
- A neuroprotective agent – a treatment that will protect and support the remaining cells
- Some form of cell replacement therapy – introducing new cells to replace the ones that have been lost
Now, the bad new is that there is no ‘silver bullet’ on the horizon that provides all three (for example, there is no neuroprotective agent that also replaces lost cells).
But the good news is that we have a great deal of clinical research being conducted in all three of these areas. This video provides an overview of just some of the many different ways we are approaching all three components:
Recently a research report focused on a cell transplantation (a form of cell replacement therapy) approach for Parkinson’s was published by a group of researchers in China. They have proposed that the results presented in the report justify their efforts to take this approach forward into clinical testing.
What is cell transplantation?
Parkinson’s is a progressive neurodegenerative condition.
This means that cells in the brain are slowly being lost over time.
What makes Parkinson’s particularly interesting is that certain types of brain cells are more affected than others. The classic example of this is the dopamine neurons in an area of the brain called the substantia nigra, which resides in the midbrain.
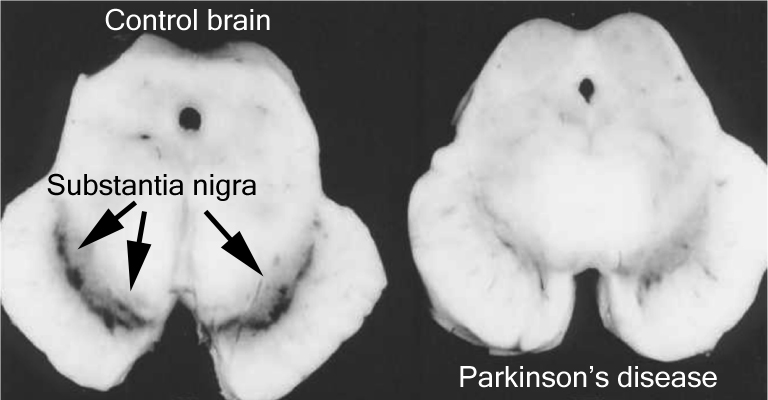
The number of dark pigmented dopamine cells in the substantia nigra are reduced in the Parkinson’s brain (right). Source: Adapted from Memorangapp
Approximately 50% of the dopamine neurons in the midbrain have been lost by the time a person is diagnosed with Parkinson’s (note the lack of dark colouration in the substantia nigra of the Parkinsonian brain in the image above), and as the condition progresses the motor features – associated with the loss of dopamine neurons – gradually get worse. This is why dopamine replacement treatments (like L-dopa) are used for controlling the motor symptoms of Parkinson’s.
A lot of research effort is being spent on finding disease slowing/halting treatments, but these will leave many people who have already been diagnosed with Parkinson’s still dealing with the condition. What those individuals will require is a therapy that will be able to replace the lost cells (particularly the dopamine neurons). And researchers are also spending a great deal of time and effort on findings ways to do this. One of the most viable approaches at present is cell transplantation therapy. This approach involves actually injecting cells back into the brain to adopt the functions of the lost cells.
How does cell transplantation work?
Many different types of cells have been tested in cell transplantation experiments for Parkinson’s (Click here for a review of this topic), but to date the cells that have given the best results have been those dissected from the developing midbrain of aborted embryos.
This ‘old fashioned’ approach to cell transplantation involved dissecting out the region of the developing dopamine neurons from a donor embryo, breaking up the tissue into small pieces that could be passed through a tiny syringe, and then injecting those cells into the brain of a person with Parkinson’s.
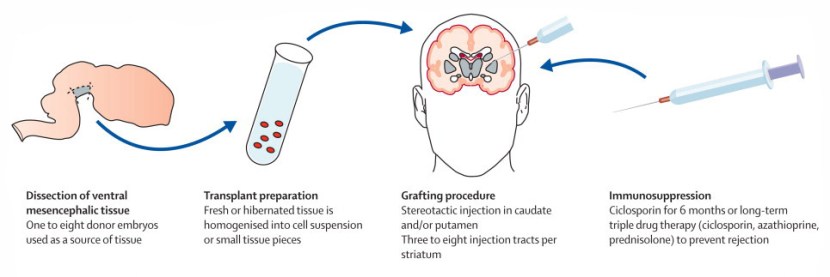
The old cell transplantation process for Parkinson’s. Source: The Lancet
Critically, the people receiving this sort of transplant would require ‘immunosuppression treatment’ for a long period of time after the surgery. This additional treatment involves taking drugs that suppress the immune system’s ability to defend the body from foreign agents. This step is necessary, however, in order to stop the body’s immune system from attacking the transplanted cells (which would not be considered ‘self’ by the immune system), allowing those cells to have time to mature, integrate into the brain and produce dopamine.
The transplanted cells are injected into an area of the brain called the putamen. This is one of the main regions of the brain where the dopamine neurons of the substantia nigra release their dopamine. The image below demonstrates the loss of dopamine (the dark staining) over time as a result of Parkinson’s (PLEASE NOTE that the time scale presented here varies from person to person):

The loss of dopamine in the putamen as Parkinson’s progresses. Source: Brain
In cell transplant procedures for Parkinson’s, multiple injections are usually made in the putamen, allowing for deposits in different areas of the structure. These multiple sites allow for the transplanted cells to produce dopamine in the entire extent of the putamen. And ideally, the cells should remain localised to the putamen, so that they are not producing dopamine in areas of the brain where it is not desired (possibly leading to side effects).

Targeting transplants into the putamen. Source: Intechopen
Postmortem analysis – of the brains of individuals who have previously received transplants of dopamine neurons and then subsequently died from natural causes – has revealed that the transplanted cells can survive the surgical procedure and integrate into the host brain. In the image below, you can see rich brown areas of the putamen in panel A. These brown areas are the dopamine producing cells. A magnified image of individual dopamine producing neurons (their circular bodies and their branches are stained in brown) can be seen in panel B:

Transplanted dopamine neurons. Source: Sciencedirect
The transplanted cells take several years to develop into mature neurons after the transplantation surgery, and the benefits of the transplantation technique may not be apparent for some time (2-3 years on average). Once mature, however, it has also been demonstrated (using brain imaging techniques) that these transplanted cells can produce dopamine. As you can see in the images below, there is less dopamine being processed (indicated in red) in the putamen of the Parkinsonian brain on the left than the brain on the right (several years after bi-lateral – both sides of the brain – transplants):

Brain imaging of dopamine processing before (left) and after transplantation. Source: NIH
Sounds like a great therapy for Parkinson’s right?
Well, thus far, cell transplantation has had a checkered past.
What do you mean?
There has been an enormous amount of cell transplant work conducted in rodent models of Parkinson’s. Many different types of cells have been used in these experiments, but by far the most successful have been immature dopamine neurons (collected from embryos). The success of that work resulted in numerous clinical trials in the 1990s. Those trials began with a group in Lund (Sweden) who, in 1991, transplanted fetal midbrain tissue into six patients: four with advanced idiopathic Parkinson’s and two from the ‘Frozen addicts’ cohort (see the book “The case of the frozen addicts” for an explanation of these two individuals).
Similar programmes had been initiated in England, Spain, Mexico, Cuba, France, and Belgium. These were all considered to be relatively successful, except for the fact that they were all open-label/not blinded studies, meaning that everyone involved in the study knew who was getting transplanted.
In the USA, these developments took place amid a major debates about the ban on federal funding for fetal tissue research that had been introduced by the Reagan administration in 1988. The Clinton administration lifted this ban in January, 1993, and this reversal opened the way for the National Institutes of Health (NIH) to provide funding for the two placebo-controlled studies. Those two trials were:
Trial no. 1: The Colorado/Columbia Trial:
A double blind trial in which 40 subjects (with advanced PD) received transplants of fetal midbrain tissue and 34 additional subjects (with advanced PD) had a sham surgery and were considered controls. Critically, neither the subjects nor the practitioners knew who was in which group. No patients in either arm of the study received immunosuppression, meaning that their immune systems were free to attack the injected cells (which would have been considered foreign by the body). The patients were followed up for 1 year after surgery and the success of the trial was judged on the basis of a self-report rating of clinical improvement or deterioration, scored by patients in their own homes and then sent to the investigator. The report of the study was published in the New England Journal of Medicine:
Title: Transplantation of embryonic dopamine neurons for severe Parkinson’s disease.
Authors: Freed CR, Greene PE, Breeze RE, Tsai WY, DuMouchel W, Kao R, Dillon S, Winfield H, Culver S, Trojanowski JQ, Eidelberg D, Fahn S.
Journal: New England Journal of Medicine 2001 Mar 8;344(10):710-9.
PMID: 11236774
Trial no. 2: The Tampa Bay Trial:
34 patients were randomly assigned either to receive a transplant of fetal midbrain tissue or to undergo a sham surgery. All patients received 6 months of immunosuppression after surgery. The primary endpoint for this study was a significant difference between the groups at 24 months after surgery. The report of this study was published in the journal, Annals of Neurology:
Title: A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson’s disease.
Authors: Olanow CW, Goetz CG, Kordower JH, Stoessl AJ, Sossi V, Brin MF, Shannon KM, Nauert GM, Perl DP, Godbold J, Freeman TB.
Journal: Ann Neurol. 2003 Sep;54(3):403-14.
PMID: 12953276
Both of these clinical studies failed to show any significant improvement at their endpoints.
In fact, 15% – 50% of transplanted subjects developed what are called ‘graft induced dyskinesias’ (or GID’s). This is where the subjects display uncontrollable/erratic movement. Interestingly, patients under 60 years of age did show signs of improvement on when assessed both clinically (using the UPDRS-III) and when assessed using brain imaging techniques (increased F-dopa uptake on PET).
And despite the fact that many of the early transplanted individuals demonstrated long-term benefits from cell replacement therapy (Click here to read more about this), the negative results from these two double-blind studies left a shadow over this field of Parkinson’s research for the better part of a decade.
Only recently have the clouds started to clear.
Are there any clinical trials for cell transplantation in Parkinson’s currently being conducted?
Yes, there are currently three:
Firstly, there is the Transeuro being conducted in Europe.
The Transeuro trial. Source: Transeuro
The Transeuro trial is an open label study, involving 13 subjects, transplanted in Sweden and the UK. They will receive immunosuppression for at least 12 months post surgery, and the end point of the study will be 3 years post surgery, with success being based on brain imaging of dopamine release from the transplanted cells (PET scans). Based on the results of the previous NIH funding double blind clinical studies discussed above, only subject under 65 years of age have been enrolled in the study.
The European consortium behind the Transeuro trial. Source: Transeuro
In addition to testing the efficacy of the cell transplantation approach for Parkinson’s, another goal of the Transeuro trial is to optimise the surgical procedures with the aim of ultimately shifting over to an embryonic stem cells oriented technique in the near future (the Transeuro is testing the old approach to cell transplantation).
The second clinical study of cell transplantation for Parkinson’s is being conducted in Melbourne (Australia), by an American company called International Stem Cell Corporation.

This study is taking the new approach to cell transplantation, but the company is using a different type of stem cell to produce dopamine neurons in the Parkinsonian brain.
Specifically, the researchers will be transplanting human parthenogenetic stem cells-derived neural stem cells (hpNSC). These hpNSCs come from an unfertilized egg – that is to say, no sperm cell is involved. The female egg cell is chemically encouraged to start dividing and then it becoming a collection of cells that is called a blastocyst, which ultimately go on to contain stem cells that can be chemically encouraged to become any type of cell, including dopamine neurons.
This process is called ‘Parthenogenesis’, and it’s not actually as crazy as it sounds as it occurs naturally in some plants and animals (Click here to read more about this). Proponents of the parthenogenic approach suggest that this is a more ethical way of generating ES cells as it does not result in the destruction of a viable organism.
Regular readers of this blog will be aware that I am extremely concerned about the Australian trial (and it’s not because I’m a kiwi and the trial is being conducted in Australia – Click here, here and here to read previous posts about this matter).
Interesting. And there is a third trial?
Yes, there is.
Which brings us to the research report we’re going to review today. There is currently a cell transplantation clinical trial being conducted in China (more on this in a minute). And recently, the researchers in China published a research report in which they outlined the approach that they are using:
Title: Human Clinical-Grade Parthenogenetic ESC-Derived Dopaminergic Neurons Recover Locomotive Defects of Nonhuman Primate Models of Parkinson’s Disease
Authors: Wang YK, Zhu WW, Wu MH, Wu YH, Liu ZX, Liang LM, Sheng C, Hao J, Wang L, Li W, Zhou Q, Hu BY
Journal: Stem Cell Reports. 2018 Jun 7. pii: S2213-6711(18)30227-3.
PMID: 29910127 (This report is OPEN ACCESS if you would like to read it)
In this study, the Chinese researchers evaluated the generation of dopamine neurons from human parthenogenetic embryonic stem cells and investigated the safety and efficacy of these dopamine neurons in a primate model of Parkinson’s (MPTP model). The reason they chose the parthenogenetic embryonic stem cells is because these have been accredited by the China Food and Drug Administration (CFDA). They had previously demonstrated that they could grow human cells in clinical-grade preparations:
Title: Accreditation of Biosafe Clinical-Grade Human Embryonic Stem Cells According to Chinese Regulations.
Authors: Gu Q, Wang J, Wang L, Liu ZX, Zhu WW, Tan YQ, Han WF, Wu J, Feng CJ, Fang JH, Liu L, Wang L, Li W, Zhao XY, Hu BY, Hao J, Zhou Q.
Journal: Stem Cell Reports. 2017 Jul 11;9(1):366-380.
PMID: 28506532 (This report is OPEN ACCESS if you would like to read it)
The investigators grew their parthenogenetic embryonic stem cells and encouraged them to become firstly neural cells, and then dopamine neurons. They did this using two different protocols for making dopamine neurons, and managed to achieve rates of 20-40% dopamine neurons (as a percentage of the total number of cells in the culture) using both protocols. These rates were the same as those achieved using normal embryonic stem cells (which they were growing along side the parthenogenetic embryonic stem cells for comparative sake). And these neurons behaved in a very similar fashion to normal brain-derived neurons (according to electrophysiological recordings made on the cells).
Before we go any further, can I just ask: What are embryonic stem cells?
Embryonic stem cells are a special kind of cell that has the potential to become any type of cell in the body – this means that they are ‘pluripotent’. Embryonic stem cells can be encouraged (using specific chemicals) to become whatever kind of cell you want them to be. This feature makes them particularly appealing to research scientists and clinicians who are interested in regenerative medicine (or replacing cells that are lost due to injury or disease).

Embryonic stem cells in a petridish. Source: Wikipedia
Embryonic stem cells are derived from a fertilised egg cell. The egg cell will divide, to become two cells, then four, eight, sixteen, etc. Gradually, it enters a stage called the ‘blastocyst’, which is a small ball of cells. Inside the blastocyst is a group of cell that are called the ‘inner stem cell mass’, and it is these cells that can be collected and used as embryonic stem cells.

The process of attaining embryonic stem cells. Source: Howstuffworks
So what did the Chinese researchers do with the cells once they could make neurons?
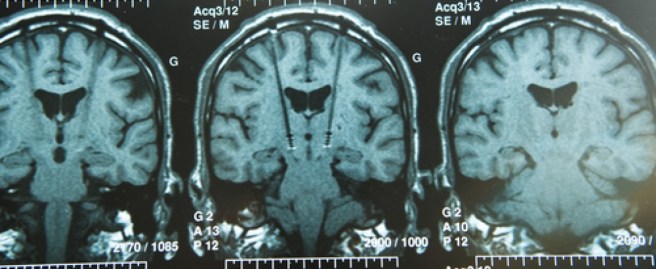
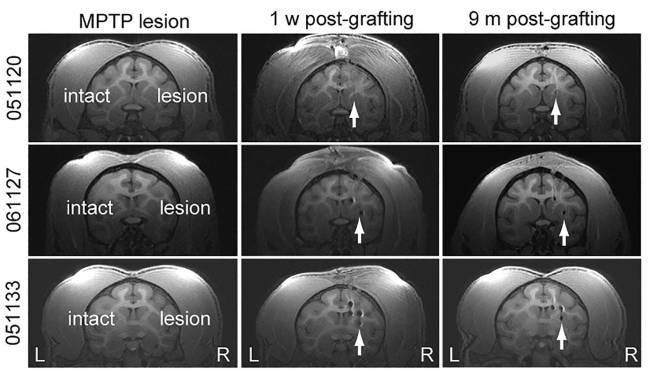
The labelled the cells with nanoparticles (to help identify the neurons) and they transplanted the cells into a primate model of Parkinson’s. 10 primates had been injected with a neurotoxin (MPTP) and behaviourally tested for 11 months. They were then divided into 3 groups (Control, Protocol #1 and Protocol #2). The control animals (3 of them) received no transplant, while the rest of the animals received cells made using either protocol #1 or protocol #2 (4 & 3 animals in each group, respectively). A total of 2 million cells were injected into both ends of the striatum (the region where the chemical dopamine is usually released – the putamen and the caudate) on the left side of the brain in each of the animals given a transplant. Brain scans immediately after surgery confirmed that the cells were placed correctly (you can see in the image below white arrows indicating the location of transplanted cells in three primates (#051120 was a control).
Examples of MRI brain images. Source: Cell
One unanswered question regarding this study is when exactly in the development of the stem cell-derived dopamine neurons were the cells transplanted. It takes 40+ days to grow dopamine neurons in cell culture using the two protocols, and the report’s only reference to which stage the cells were collected at was the phase “committed dopamine neurons” in the results section (I’m happy to be corrected on this if I am wrong).
The timing of the collection of cells is an important detail.
If the cells are collected too early, they may not become dopamine neurons – they may become a different type of neuron (which would reduce the amount of dopamine being released by the transplanted cells). And if the cells are collected too late – at a point where the neurons are mature and have grown branches that make contacts with surrounding cells – the process of collecting and preparing the cells for transplantation could actually kill the cells (for example, tearing their branches off). Previously the Chinese research group had suggested that they would transplant ‘stem-cell-derived neuronal-precursor cells‘ (Source), which are very young neurons that could become any kind of mature neuron. In this new report, though they appear to have taken a later time point, but exactly when that time point is, is not clear.
The animals were transplanted on the left side of their brains, while the neurotoxin affected dopamine neurons on both sides. Using this unilateral approach, the investigators could compare the motor function on the transplanted side of the body with the untransplanted side. And this assessment conducted on a monthly basis in a blinded manner by experienced examiners (blinded meaning that the assessors were not sure which primates had been transplanted, nor which side of the brain was transplanted). For comparative sake it could have been useful to have some primates that received no neurotoxin and no transplant, but this was missing in the current study.
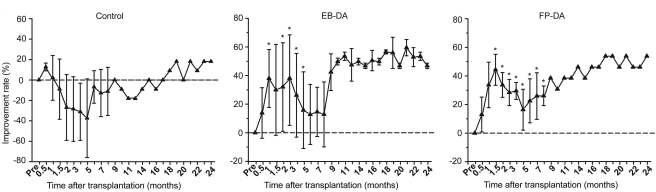
While the control treated animals exhibited little recovery at 8 months post transplantation, the behavioural assessment results demonstrated a significant locomotive performance rating scores improvement in the transplanted primates. And in the small number of animals that were kept till the end of the study, this behavioural improvement was sustained for those 24 months. As you can see in the graph below, the line on the control graph never climbs above 20% improvement, while both the transplanted groups demonstrate positive trends.
Behavioural improvement for EB-DA & FP-DA transplanted animals. Source: Cell
The brains of 6 animals were analysed 9 months after transplantation and there were no signs of tumour growth or any other kind of abnormality. And while there was some cell migration away from the transplantation site, there was no evidence of tumour growth in any other organs in the body. There was also evidence of microglia cells around the transplant site. Microglia are the resident immune cells of the brain, and their presence suggests a moderate but tolerable immune response in the host brain.
Immunosuppression treatment is used in cell transplantation trials to ensure that the grafted cells have the best chance of surviving. Without immunosuppression, the immune system might attack the cells as they are foreign objects and not considered ‘self’. The monkeys in this study only received immunosuppression every 2 days for 2 weeks following the transplantation procedure, which could partly explain the observed microglial response.
When the researchers looked at the number of surviving cells in the transplants, they found a large range of variability between animals (from 350,000 to 950,000 cells). And of the proportion of dopamine neurons among those survival cells varied between 5% and 8% (of the total number of cells).
Roll on to clinical testing. These results look good right?
The researchers should be congratulated for the completion of a mammoth project, but (you knew there was a ‘but’ coming) I do have one concern with regards to this project justifying the moving of this cell transplantation protocol to the clinic: the data between animals was extremely variable.
And to their credit, the researchers shared this aspect of the data.
For example, they note that the maximum individual behavioural improvement observed was a 69.2% improvement in one animal. This score occurred just 2 months after the transplantation procedure and the effect diminished during the next 6 months, resulting in an improvement of just 23.1% at the end of 8 months. And if you look at the behavioural data there is a great deal of variability in the data during the first 9 months.
Now to be fair, this is a common feature of studies involving small numbers of subjects, but it does raise concerns with regards to moving this procedure to the clinical setting too quickly. Ideally, we want a protocol that will provide a very consistent result every time, and not so much variability.
But ‘ideal’ can be a hard-to-reach ideal.
So what do we know about the Chinese clinical study?
The clinical trial (Titled: A Phase I/II, Open-Label Study to Assess the Safety and Efficacy of Striatum Transplantation of Human Embryonic Stem Cells-derived Neural Precursor Cells in Patients With Parkinson’s Disease) is taking place at the First Affiliated Hospital of Zhengzhou University in Henan province (Click here to read more about the trial).
The study is a Phase I/II, open-label, non randomised clinical trial that will enroll 50 people with Parkinson’s (having had the condition for more than 5 years post diagnosis).
There will also be a series of secondary outcome measures which will assess efficacy. These include:
- Change in Unified Parkinson’s Disease Rating Scale (UPDRS) score at 12 months post surgery, when compared to baseline scores. Each subject was independently rated by two observers at each study visit and a mean score was calculated for analysis.
- Change in DATscan brain imaging at 12 months when compared to a baseline brain scan taken before surgery. DATscan imaging provides an indication of dopamine processing.
- Change in Hoehn and Yahr Stage at 12 months, compared to baseline scores. The Hoehn and Yahr scale is a commonly used system for Parkinson’s.
The estimated date of completion is December 2020.
So what does it all mean?
Chinese researchers have present data from primate studies – which are considered the last step before testing in humans – that they believe justifies their ongoing efforts to test their cell transplantation procedure in people with Parkinson’s.
This post is hopefully just the first of a series of posts addressing efforts across the global to take embryonic stem cell-based cell transplantation to the clinic for Parkinson’s. In the near future (before the end of this year hopefully) we should also be hearing from a group in Japan and another group in the USA (led by Bluerock Therapeutics) as they begin their clinical trials for cell transplantation in Parkinson’s.
In addition, there is an effort being driven by the biotech firm Novo Nordisk, in partnerships with the Swedish biotech company BioLamina and researchers at Lund University researchers (Click here to read more about this).
We will most likely be reading more about pre-clinical research from some of these research groups very soon, which will – like the research reviewed in today’s post – be used to justify moving forward in the clinic. And all of this bodes very well for our attempts to replace some of the cells and function that have been lost in Parkinson’s (the third component of any ‘cure’ for Parkinson’s.
EDITORIAL NOTE #1 – It is important for all readers of this post to appreciate that cell transplantation for Parkinson’s is still experimental. Anyone declaring otherwise (or selling a procedure based on this approach) should not be trusted. While we appreciate the desperate desire of the Parkinson’s community to treat this condition ‘by any means possible’, bad or poor outcomes at the clinical trial stage for this technology could have serious consequences for the individuals receiving the procedure and negative ramifications for all future research in the cell transplantation field.
EDITORIAL NOTE #2 – the author of this blog is associated with research groups conducting the current Transeuro trial. He has endeavoured to present an unbiased coverage of the news surrounding the current clinical trials. Any other opinions offered here are solely his own and do not represent those of any associated parties. He had not discussed the recent research report with any colleagues before publishing this post.
The banner for today’s post was sourced from Wikipedia








Great as always Simon!
What are your thoughts on Mesenchymal stem cells for PD? I know they aren’t replacements, But it there any credibility data on potential benefits from the various factors they supposedly release?
LikeLike
Hi Double,
Glad you liked it. Regarding mesenchymal stem cells for PD: I am yet to see anything convincing for cell transplantation outside of fetal tissue or the new embryonic stem cell approaches. There has been a long history of different types of cells being injected into the brain for Parkinson’s, and thus far only fetal tissue and embryonic stem cell approaches have provided anything near consistent results. And I say this with a deep reservations about the direct-to-consumer market that is selling mesenchymal stem cells for PD – there is absolutely no clinically approved evidence of efficacy for those cells.
Kind regards,
Simon
LikeLike
Thanks Simon!
Yeah the snake oil stem cells seem scary.
LikeLike
Great article, thank you.
As you said it takes 2 to 3 years for stem cells to get mature in brain in produce dopamine, but the studies don in US, monitored the results in much shorter duration after the transplantation. Couldn’t this lead to wrong results in the those studies?
Regards
Hessam
LikeLike
Hi Simon,
Excellent explanations! Thank you! I found my way to this article because I had the same question about the age of the mDA neurons used for the PD study led by Qi Zhou’s group. The way the publication makes it sound, they’re transplanting mature mDA neurons. But then everything you read about the clinical trial says they’re transplanting neural precursor cells. As it’s been a few years now since the start of the trial I’m wondering if you’ve learned anything new? Do you think it’s possible that the CFDA approved the trial using NPCs even though they used more mature mDA neurons in the monkey studies? Or that either the publication or the info on the trial is misleading about the age of the cells? Any thoughts? Thanks!
Shannon
LikeLike