|
Numerous readers have asked about a curious new clinical trial being conducted by a biotech firm called ‘Alkahest’. The company has recently initiated a large (90 participants) Phase II study of their Parkinson’s-focused treatment called GRF6021. This is an experimental, intravenously-administered treatment, which is derived from a components of blood. In today’s post, we will discuss some of the research behind GRF6021, what this new clinical trial involves, and have a look at some other interesting Parkinson’s-related activities that Alkahest has ongoing.
|
 Source: SFN
Source: SFN
The Society of Neuroscience meeting is the largest annual research conference on brain relelated research, bringing approximately 40,000 neuroscientists together in October. At the Society of Neuroscience meeting in San Diego this year, however, there was considerable interest focused on several presentations dealing with blood.
The first presentation was from a group of researchers at the University of California, San Francisco.
 The research team – led by group leader Dr Saul Villeda – were presenting new data suggesting that circulating immune cells were most likely responsible for the age-related reduction in neurogenesis (formation of new neurons) that occurs in certain areas of the brain (Click here to read the abstract for this presentation). They reported that the aged hematopoietic (blood) system led to impaired neurogenesis. Their take-home-message: the older the blood system, the less new cells being produced by the brain.
The research team – led by group leader Dr Saul Villeda – were presenting new data suggesting that circulating immune cells were most likely responsible for the age-related reduction in neurogenesis (formation of new neurons) that occurs in certain areas of the brain (Click here to read the abstract for this presentation). They reported that the aged hematopoietic (blood) system led to impaired neurogenesis. Their take-home-message: the older the blood system, the less new cells being produced by the brain.
Sounds interesting right?
Well, at the same time in another part of the conference a second group of researchers were presenting equally impressive data: They have zeroed in of a small fraction of normal, young blood that they believe has interesting properties, particularly in reversing the cognitive deficits associated with aging mice (Click here to read the abstract of this presentation).
Their research has even narrowed down to a specific protein, called C-C chemokine receptor type 3 (or CCR3), which when inhibited was found to improve cognitive function and decreased neuroinflammation in aged mice (Click here to read the abstract of the presentation).
The humble lab mouse. Source: Pinterest
But specifically for our interests here at the SoPD, these same researchers displayed data which demonstrated that treatment with a novel fraction of human plasma resulted in significant improvements in motor function, cell survival and neuroinflammation three weeks after treatment in multiple mouse models of Parkinson’s (Click here to read the abstract of the poster).
(PLEASE NOTE: The author of this blog was not present at the SFN meeting and is working solely with the abstracts provided)
This second group of scientists were from a company called Alkahest, and they have recently started a clinical trial for people with Parkinson’s based on these results. That trial has garnered quite a bit of interest in the Parkinson’s community. What do Alkahest do?
What do Alkahest do?
Alkahest – the name coming from the hypothetical universal solvent sought by alchemists – is a California-based biotech firm, that is developing therapies derived from components of blood. They have a focus on improving vitality and function in old age, with a particular interest on neurodegenerative conditions.
What do you mean by “derived from components of blood”?
There are three chief components of blood:
- Red blood cells
- White blood cells
- Plasma (92% water and 8% other stuff – think blood clotting proteins, waste, nutrients, etc)
By volume, the red blood cells constitute about 45% of whole blood, the plasma about 54.3%, and white cells about 0.7%.
 Source: KhanAcademy
Source: KhanAcademy
Now the important part of the description above is the “8% other stuff” mentioned in the plasma faction. There are all sorts of interesting proteins floating around that that ‘other stuff’ that can have very beneficial effects.
 Bags of plasma are used in modern medicine. Source: Inverse
Bags of plasma are used in modern medicine. Source: Inverse
And Alkahest has identified specific proteins (which they call “chronokines”) that increase as we age and are very high in age-related medical conditions, like neurodegeneration. They also believe that by reducing these chronokines that this treatment may have positive benefits on conditions associated with aging, such as dementia and Parkinson’s.
How do they plan to reduce these chronokines?
Two methods.
Either by using inhibitors which block specific chronokines, or by supplementing/replacing the plasma of an individual with components from younger plasma (less chronokines).
What do you mean ‘components from younger plasma’?
I mean that the company has spent a great deal of time collecting blood from young individuals and isolating components of the plasma from those younger individuals, and they believe that they have identified components from that younger plasma which could have beneficial effects for older individuals.
Is there any evidence supporting this idea?
Pre-clinically, yes there is.
Back in 2005, some Californian researchers noticed something interesting:
 Title: Rejuvenation of aged progenitor cells by exposure to a young systemic environment
Title: Rejuvenation of aged progenitor cells by exposure to a young systemic environment
Authors: Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA.
Journal: Nature. 2005 Feb 17;433(7027):760-4.
PMID: 15716955
In this study, the scientists wanted to examine the influence of “systemic factors” (basically, proteins floating around in the blood) on aged progenitor cells in certain tissues. They tested this by establishing parabiotic pairings of mice.
What are parabiotic pairings?
Ok, before we go any further – if you are remotely squimmish, please skip this section.
Parabiotic pairing of mice is a technique used in scientific research to evaluate the effect of a shared blood system. That is to say, this approach requires the surgical joining of two mice (each with different conditions – eg. fat mouse and lean mouse) and then they co-habitate with a combined blood system for a given period of time to determine if one condition can influence another – see image below:
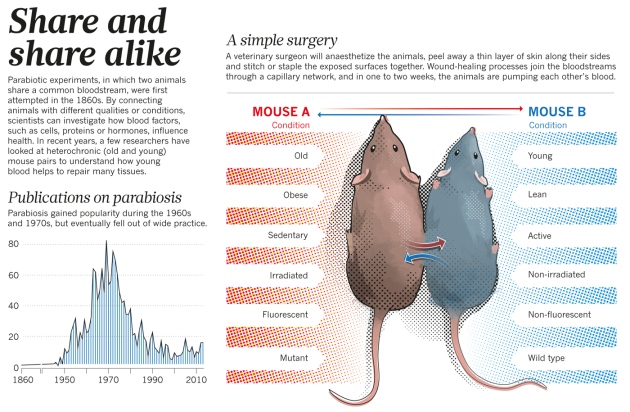 Source: Nature
Source: Nature
For a good OPEN ACCESS historical review of parabiotic pairing research – Click here.
In the current study, the researchers shared the circulatory system of young and old mice (this is called a heterochronic parabiosis) for 5 weeks. This combination exposed the old mice to factors that are present in blood (including all the ‘other stuff’ in the plasma) of young mice, and allowed the researchers to determine if this exposure benefitted the older mice.
Eeekkk! Sounds awful… But what did they find?
The researchers found evidence of improved rejuvenation in certain cell types that were analysed. For example, they noted the restoration of specific biological pathways (such as Notch signalling) as well as the proliferative capacity in the satellite cells of the older mice. Satellite cells are the precursors to skeletal muscle cells.
The researchers also analysed different cell types in the liver and noted similar rejuvenation results.
Given these results, the researchers concluded that “the age-related decline of progenitor cell activity can be modulated by systemic factors that change with age“.
And subsequent studies have found similar results – they also demonstrated the reverse effect: Young animals exposed to older systemic factors exhibited reduced rejuvenative properties (Click here and here to read more about this).
What about the brain?
This question has been addressed in similar studies to the one described above – and the results have been published in reports, such as this one:
 Title: The ageing systemic milieu negatively regulates neurogenesis and cognitive function.
Title: The ageing systemic milieu negatively regulates neurogenesis and cognitive function.
Authors: Villeda SA, Luo J, Mosher KI, Zou B, Britschgi M, Bieri G, Stan TM, Fainberg N, Ding Z, Eggel A, Lucin KM, Czirr E, Park JS, Couillard-Després S, Aigner L, Li G, Peskind ER, Kaye JA, Quinn JF, Galasko DR, Xie XS, Rando TA, Wyss-Coray T.
Journal: Nature. 2011 Aug 31;477(7362):90-4. doi: 10.1038/nature10357.
PMID: 21886162 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers used the heterochronic parabiosis model of young and old mice to determine if young blood could counteract (or reverse) the decline in neurogenesis (the turn-over of new cells) which is associated with normal aging.
Neurogenesis in the adult human and rodent brain primarily isolated to two regions: the subventricular zone of the striatum and the subgranular zone of the hippocampus.
 Neurogenesis. Source: Development
Neurogenesis. Source: Development
As we age, these regions exhibit less and less neurogenesis.
The investigators who conducted this study found that blood-based factors can inhibit or promote adult neurogenesis – in an age-dependent fashion (in mice at least). Older mice who were exposed to the young mouse blood system exhibited increased signs of neurogenesis.
Interestingly, by exposing young mice to the old systemic environment (or to plasma from old mice), the researchers reported decreased neuronal activity (using electrophysiology) and reduced performance on contextual fear conditioning and spatial memory tests.
Next the investigators attempted to identify specific factors in the blood that may be causing these effects. They isolated a protein messenger (or chemokine) called C-C motif chemokine 11 (or CCL11). CCL11 (also known as eosinophil chemotactic protein and eotaxin-1) is a small immunomodulatory protein (or cytokine) that increases as healthy humans age and correlates with reduced neurogenesis.
The researchers found that by increasing CCL11 levels in young mice, there was a subsequent decrease in neurogenesis as well as reduced performance in learning and memory tests. The investigators concluded that “the decline in neurogenesis, and cognitive impairments, observed during aging can be in part attributed to changes in blood-borne factors”.
And these initial brain-related results have been replicated in additional studies, such as this one:
 Title: Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors.
Title: Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors.
Authors: Katsimpardi L, Litterman NK, Schein PA, Miller CM, Loffredo FS, Wojtkiewicz GR, Chen JW, Lee RT, Wagers AJ, Rubin LL.
Journal: Science. 2014 May 9;344(6184):630-4.
PMID: 24797482 (This report is OPEN ACCESS if you would like to read it)
In this study, the investigators found that 5 weeks exposure to factors in young blood was associated with increased vasculature (blood vessels) in the hippocampus and in the subventricular zone, and this was associated with increased neurogenesis in these regions as well as improved performance in olfactory discrimination tests performed on aging mice (who were exposed to young blood).
The researchers behind this study had recently identified a plasma-borne factor that was found to improve cardiac muscle in aged animals. That factor is called Growth differentiation factor 11 (GDF11; also known as bone morphogenetic protein 11 or BMP11 – Click here to read more about that research).
GDF11 is a circulating member of the BMP/TGF-β family and when the investigators exposed old mice to GDF11 treatment, they observed signs of increased vasculature as well as neurogenesis. GDF11 treatment has also been found to have beneficial properties in other organs, such as muscle (Click here to read more about this).
Based on these results, the scientists proposed that factors like GDF11 could represent “new therapeutic strategies for treating age-related neurodegenerative conditions”.
(NOTE: There have been several efforts made by independent research groups to replicate the GDF11 findings, but they have been unsuccessful – Click here and here for an example)
And that report was quickly followed by a similar report:
 Title: Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice.
Title: Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice.
Authors: Villeda SA, Plambeck KE, Middeldorp J, Castellano JM, Mosher KI, Luo J, Smith LK, Bieri G, Lin K, Berdnik D, Wabl R, Udeochu J, Wheatley EG, Zou B, Simmons DA, Xie XS, Longo FM, Wyss-Coray T.
Journal: Nat Med. 2014 Jun;20(6):659-63.
PMID: 24793238 (This report is OPEN ACCESS if you would like to read it)
In this study, after five weeks of sharing a circulatory system with young mice, the older mice exhibited evidence of increased connections between mature neurons in a region of the brain called the hippocampus, as well as increased synaptic plasticity (neuronal activity as measured by electrophysiology) in that region.
On the cognitive level, the researchers also demonstrated that systemic administration of blood plasma (collected from young mice) into aged mice not only improved the age-related impairments in a fear conditioning test, but also in a spatial learning/memory assessment.
The researchers concluded that their “data demonstrate that exposure to young blood counteracts aging at the molecular, structural, functional and cognitive levels in the aged hippocampus”.
Given all of these intriguing results, the researchers involved with this study went on to test young blood plasma in models of Alzheimer’s.
And this report was published in 2016:
 Title: Preclinical Assessment of Young Blood Plasma for Alzheimer Disease.
Title: Preclinical Assessment of Young Blood Plasma for Alzheimer Disease.
Authors: Middeldorp J, Lehallier B, Villeda SA, Miedema SS, Evans E, Czirr E, Zhang H, Luo J, Stan T, Mosher KI, Masliah E, Wyss-Coray T.
Journal: JAMA Neurol. 2016 Nov 1;73(11):1325-1333.
PMID: 27598869 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers used a genetically engineered mouse that produces a mutated versions of the Alzheimer’s-associated protein amyloid precursor protein (two mutations: hAPP751 cDNA containing the London (V717I) and Swedish (K670M/N671L) variants – Click here to read more about these mice). In these mice, by 5-6 months, the characteristic hallmarks of Alzheimer’s – beta amyloid-containing amyloid plaques – were present in various regions of the brain.
Parabiotic pairings with young mice were set up at middle age (10 months of age) and later age (16 months of age). After 5 weeks exposure to a young blood system, the researchers found that amyloid plaques were not affected by the treatment (they looked the same between groups), nor was the microglial activation (the resident immune cells in the brain) affected.
But what the researchers did note was “a near complete restoration in levels of synaptic and neuronal proteins after exposure to young blood“. And this effect was associated with improved performance in a fear conditioning test and a spatial learning/memory assessment.
The conclusions of this study was that “Factors in young blood have the potential to ameliorate disease in a model of Alzheimer’s”. And this has led to the establishing of clinical trials for people with Alzheimer’s – more on this below.
(NOTE: The senior researcher on several of these reports mentioned above is Prof Tony Wyss-Coray. Prof Wyss-Coray is the founder, board member and Chairman of the Scientific Advisory board at Alkahest).
This is very interesting, but what does all of this have to do with Parkinson’s?
On the 4th December, Alkahest announced that they have dosed the first participant in a Phase II clinical trial of their product GRF6021 in people with Parkinson’s and cognitive impairments (Click here for the press release). This study is being supported by the Michael J Fox Foundation.
In addition, earlier in the year Alkahest also announced that they had received another funding grant from the Michael J Fox Foundation to conduct a clinical trial of GRF6019 in people with Parkinson’s (Click here to read the press release).
What are GRF6021 and GRF6019?
Both of these experimental treatments are ‘plasma fractions’ (evidently made up of approximately 400 proteins – source). It has been developed by Alkahest and manufactured in collaboration with Grifols.
Grifols is a Spanish pharmaceutical manufacturer, which is one of the world’s largest producers of blood plasma-based products (a lot of experinence/expertise in this field!).
Both GRF6021 and GRF6019 represent second-generation experimental treatments based on earlier clinical research infusing young adult plasma to treat age-related neurodegenerative conditions (see below for more on this). In addition to the Parkinson’s-GRF6021 clinical trial, Alkahest are also testing GRF6019 in two clinical trials for mild and late-stage Alzheimer’s (Click here and here to read more about those trials).
What does the new Parkinson’s clinical trial involve?
The study is a randomised, double-blind, placebo-controlled Phase II study in 90 people with Parkinson’s and cognitive impairment. It will be assessing the safety and tolerability of GRF6021 over a period of 7 months. The treatment (or placebo) will be administered by intravenous infusion for 5 consecutive days at Week 1 and Week 13 of the study.
Safety and tolerability is the primary end point, but change from baseline in the MDS-UPDRS score is a secondary end point so we will hopefully get a measure of whether the treatment is having any effect (Click here to read more about the details of this clinical study).
As we mentioned above, Alkahest has dosed the first participant in the study in early December (Click here for the press release), and the trial is set to run across six U.S. research centers. It will hopefully be finished in November 2019, so we can expect to see the results in early 2020.
Has GRF6021 ever been tested in humans before?
No, it has not (that I am aware of, happy to be corrected).
But as we mentioned above, GRF6021 is a second generation product, and previously researchers at Alkahest and Stanford University conducted a double-blinded Phase I clinical trial on 18 people with mild to moderate Alzheimer’s. This trial was referred to as the “PLasma for Alzheimer SymptoM Amelioration” (PLASMA) Study (Click here to read more about it).
The results of that study were recently published:
 Title: Safety, Tolerability, and Feasibility of Young Plasma Infusion in the Plasma for Alzheimer Symptom Amelioration Study: A Randomized Clinical Trial.
Title: Safety, Tolerability, and Feasibility of Young Plasma Infusion in the Plasma for Alzheimer Symptom Amelioration Study: A Randomized Clinical Trial.
Authors: Sha SJ, Deutsch GK, Tian L, Richardson K, Coburn M, Gaudioso JL, Marcal T, Solomon E, Boumis A, Bet A, Mennes M, van Oort E, Beckmann CF, Braithwaite SP, Jackson S, Nikolich K, Stephens D, Kerchner GA, Wyss-Coray T.
Journal: JAMA Neurol. 2018 Oct 29.
PMID: 30383097
This was a 12-week study that involved a four-week treatment regimen of once-per-week injections of one unit of plasma (from males aged <30 years – Click here to read more about this). This treatment was compared to a placebo treatment, but there was a cross-over design incorporated in the study:
 Safety was the primary outcome, and the results of the study suggested that the treatment was safe and well tolerated. The small size (both in terms of participants and time) and the open label nature of the study, however, mean that we can not really take much else away from the results beyond the safety/tolerability finding. That said, the researchers suggested that the treatment also appeared to yield “a signal on functional/daily activities tests” and this is being used as justification for a larger Phase II clinical trial.
Safety was the primary outcome, and the results of the study suggested that the treatment was safe and well tolerated. The small size (both in terms of participants and time) and the open label nature of the study, however, mean that we can not really take much else away from the results beyond the safety/tolerability finding. That said, the researchers suggested that the treatment also appeared to yield “a signal on functional/daily activities tests” and this is being used as justification for a larger Phase II clinical trial.
GRF6021 was a reformulation based on this first study. And importantly, unlike plasma, GRF6021 does not require blood typing/matching. This limits the risk of adverse side effects (like an immune respose), and means that new product could potentially be administered to anyone.
Has blood plasma ever been clinically tested in Parkinson’s?
Yes.
There is an ongoing clinical study called “The Stanford Parkinson’s Plasma Study”, which is being conducted at the Stanford Movement Disorders Center.
 Source: Med.stanford
Source: Med.stanford
This Phase I clinical study is testing whether young plasma infusions can be safely used on people with Parkinson’s. It is a single group (no placebo arm), open label (meaning everyone knows what the treatment is) study, in which participants have recieved 4 twice-per-week infusions of 1 unit of young plasma (from males ages between 18-25) and been monitored over 8 weeks (Click here to read more about this study).
The study is nearing completion and the results should be available in the new year (2019) – expect an SoPD post on this.
Very interesting. So what does it all mean?
Yeah, not just yet.
We’re not summing until we have a quick look at another piece of this story.
You see, Alkahest is working on something else that is really interesting.
Que?
In February 2018, Alkahest received another grant from the Michael J Fox Foundation for the preclinical testing of ALK4290 (Click here to read the press release).
What is ALK4290?
ALK4290 is an orally-administered small molecule that acts as a modulator of inflammation. ALK4290 is a C-C motif chemokine 11 (or CCL11) inhibitor. As we discussed above (reviewing the 2011 Nature report), CCL11 (also known as eosinophil chemotactic protein and eotaxin-1) is a small immunomodulatory protein (or cytokine) that increases as humans age.
CCL11 binds to the receptor C-C chemokine receptor type 3 (or CCR3) which we mentioned in the intro to this post (one of the presentations from the Society of Neuroscience meeting). Alkahest has some (unpublished) data demonstrating the beneficial effects of inhibiting CCR3 in models of Parkinson’s.
Alkahest acquired ALK4290 from Boehringer-Ingelheim and has exclusive rights for development/commercialisation worldwide.
 ALK4290 represents the first non-plasma-derived product in Alkahest’s pipeline, but also a more targetted approach to their overall thesis.
ALK4290 represents the first non-plasma-derived product in Alkahest’s pipeline, but also a more targetted approach to their overall thesis.
The preclinical research has evaluated the effect of ALK4290 in two pre-clinical models of Parkinson’s: A neurotoxin-induced model and a transgenic model of the condition. These experiments were conducted to assess whether ALK4290 could reverse or rescue the motor function and loss of dopamine neurons that are observed in these models. It will be interesting to see these recults published in the new year, and possibly the initiation of the clinical testing of ALK4290 in Parkinson’s.
Has ALK4290 ever been tested in humans before?
Yes, it has.
There have been two European studies (registered on the as ALK4290-201 and ALK4290-202). There are both open-label Phase 2 clinical trials investigating the safety and efficacy of ALK4290 over the course of a six-week oral treatment regime in patients with either newly diagnosed wAMD or refractory wAMD. These studies were started in January of this year (2018) and are being conducted in Hungary and Poland (Click here to read the press release).
What is wAMD?
Wet Age-related Macular Degeneration (or wAMD) is one of two forms of age-related macular degeneration (AMD) – dry being the second form. AMD is an eye condition that generally affects people over the age of 50. It causes problems with central vision, but does not lead to total loss of sight.
If these initial studies demonstrate that ALK4290 is safe, it should be relatively straight forward asking the regulators to test the drug in Parkinson’s – particularly if supported by some data from preclinical PD models.
So what does it all mean?
This post resonates with me.
I have a 6 year old little force of nature that has a robust immune system (based on all the stuff that goes in her mouth) and a seemingless endless supply of energy. What I wouldn’t give for a little bit of the latter in particular. The idea that there are compounds in the young blood system that help humans survive to adulthood is not a hard sell, and the possibility that these could also make life easier in the process of aging (and age-related neurodegenerative conditions) is certainly worth exploring.
Recently a new clinical trial has commenced that is investigating a novel form of treatment for Parkinson’s. A biotech company called Alkahest has initiated a clinical trial exploring the use of components of young blood to help alleviate Parkinson’s. While I am not one to slow down the progression of research on Parkinson’s, it would be agreeable for the preclinical data supporting these clinical trial efforts to be published in the new year – particularly given that the research is being partly funded by grants from the MJFF.
We will be looking forward to any news regarding GRF6021 in 2019, and we will be reporting it here.
The banner for today’s post was sourced from Wikipedia



