|
In 2019, researcher from around the world gathered for a special meeting in Toronto (Canada) to discuss/debate some of the most important issues slowing Parkinson’s research. Specifically, they asked if Parkinson’s is not a single condition, but rather a collection of diseases that look very similar, can current research efforts be revised to cater for this shift in definitions, or does Parkinson’s research need to be reconstructed? Recently a summary of the discussions at the meeting has been published. In today’s post, we will review that summary document, explore some of the topics discussed, and consider some of their proposed solutions. |
 Source: Medium
Source: Medium
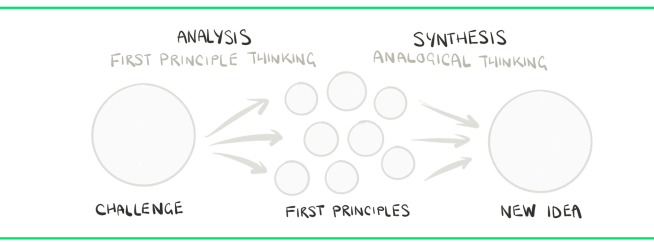
Let’s start this post with a simple question: What are the first principles of Parkinson’s?
What do you mean “first principles”?
First principles are what philosophy and engineering types considered the most basic propositions (or assumptions) that cannot be deduced from any other propositions or assumptions.
Simply, they are the fundamental facts from which everything else stems.
Elon Musk likes to talk about first principles in all of his projects, and on all matters he suggests we should : “boil things down to their fundamental truths and say, ‘What are we sure is true?’… and then reason up from there, as opposed to reasoning by analogy”.
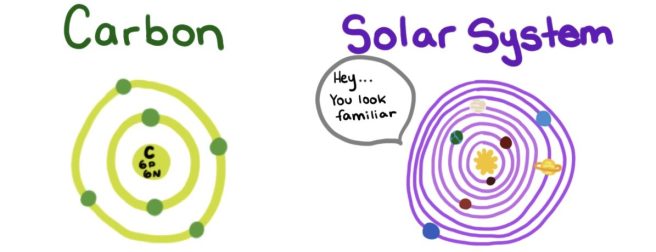
What is meant by ‘analogy’ here?
Analogy refers building knowledge and solving problems based on prior assumptions; using beliefs widely held and approved by a majority of people. It allows you to take your understanding of one domain and compare (or apply) it with another.
The example of analogy is that it is easier to teach students that electrons whizz around an atom’s nucleus the same way planets orbit the sun because they will have been exposed to this idea, than actually explaining the intricacies of nuclear physics…even though there are more than a few differences.
 Source: Blog44
Source: Blog44
Let’s return to the initial question though: What are the first principles of Parkinson’s?
I don’t know. Are you going to tell me?
Nope. I don’t know either.
Ok. Um. So is this going to be a really short SoPD post then?
No.
Recently Parkinson’s researchers around the world have been calling for efforts to establish the “first principles” of Parkinson’s (and Parkinson’s is not alone here – neurodegenerative research in general is going through a similar period of self reflection – click here to read more about this in Alzheimer’s).
A good example of this process is what happened in Toronto last April.
What happened in Toronto?
 Toronto. Source: Joinef
Toronto. Source: Joinef
Several remarkable things happened in Toronto in early 2019.
First, the city’s NBA bastketball team, the Toronto Raptors, won the championship on the back of a MASSIVE “We the North” supporters campaign:
It was fantastic fairy tale stuff!
And the team is doing fairly well this season… (“cough”, go Celtics!).
The other interesting thing was a meeting that was held at the Krembil research institute:
 Source: Uhn
Source: Uhn
The “Krembil Knowledge Gaps in PD Symposium” was held between April 24 to 26th, and the organisers wanted to tackle a fundamental conceptual challenge that the field of Parkinson’s research is facing at present.
Specifically, they wanted to discuss:
If Parkinson’s is not a single condition (but rather a collection of similar looking diseases), can research into biomarkers and potentially disease modifying treatments be revised to incorporate this concept, or do we need to reconstruct it so that it can be applied to smaller subgroups of individuals?
The meeting was a gathering of revisionists and reconstructionists (and everyone in between), and they were brought together to address this and some of the other big questions surrounding Parkinson’s research.
Interesting. What was the outcome of the meeting?
Last week a summary document of the meeting was published.
This is the report here:
 Title: Disease modification and biomarker development in Parkinson disease: Revision or reconstruction?
Title: Disease modification and biomarker development in Parkinson disease: Revision or reconstruction?
Authors: Espay AJ, Kalia LV, Gan-Or Z, Williams-Gray CH, Bedard PL, Rowe SM, Morgante F, Fasano A, Stecher B, Kauffman MA, Farrer MJ, Coffey CS, Schwarzschild MA, Sherer T, Postuma RB, Strafella AP, Singleton AB, Barker RA, Kieburtz K, Olanow CW, Lozano A, Kordower JH, Cedarbaum JM, Brundin P, Standaert DG, Lang AE.
Journal: Neurology. 2020 Feb 26. pii: 10.1212/WNL.0000000000009107. [Epub ahead of print].
PMID: 32102975
In their report, some of the attendees of the meeting outlined various topics/questions that were addressed during the meeting.
To be very clear, I was not at the meeting, so this post is based solely on my reading of the report rather than first hand information. But I have been told that the meeting was excellent by several individuals who attended, and I thought the summary was very interesting – certainly enough to warrant a SoPD post being written about it.
The summary of the meeting proposed various ‘knowledge gaps’ and then briefly discussed ‘key road blocks’ slowing progress, as well as potential ‘revisionist’ and/or ‘reconstructionist’ solutions.
The first of these Knowledge gaps was:
Does the variation between individual with Parkinson’s (in terms of symptoms, speed of progression, etc) represent “a spectrum with different clinical subtypes” or does it suggest distinct conditions that could each be considered a definable disease?
For a long time, the first idea (a spectrum) has been the dominating theme in the research field – after all, it is referred to as ‘Parkinson’s disease’ singular. And by conceptualising the condition as such, it has allowed the research community to collect enormous amounts of data from large cohorts of people, like the Michael J Fox foundation supported Parkinson’s Progression Marker Initiative.
But thus far, attempts to find biomarkers of progression in Parkinson’s has struggled and reproducing published results has been tricky (Click here to read more about this).
It should be stated very clearly here that this does not mean that these large cohort initiatives should not be conducted. I actually would suggest that in the absence of definitive biomarkers, larger cohorts are required so that bigger pools of data can be analysed. If Parkinson’s is made up of a dozen ‘distinct conditions‘, a cohort of 1000 individuals with the condition is rather small for identifying biomarkers that may fluctuate with progression within each subgroup (assuming each individual is at different stages since their diagnosis).
The second Knowledge gap listed in the summary is:
Would delivering experimental therapies to larger and earlier cohorts, or to “smaller, biologically defined populations” increase chances of successful intervention?
When clinical trials for intervention in Parkinson’s have previously not had a positive outcome, the report suggests that the results have often been attributed to one (or more) of three things:
- Shortcomings of the clinical trial itself (for example, wrong dose, drug didn’t access the brain enough, issues with outcome measures, etc).
- Wrong cohort (perhaps participants were too advanced for the intervention to be effective).
- Poor translatability from animal models to human trials (there are no good animal models of Parkinson’s).
The attendees of the Krembil Knowledge Gaps meeting proposed ‘revisions’ involving the study of larger, earlier cohorts (people who are presymptomatic or recently diagnosed), and creating better experimental models of Parkinson’s. Their proposals for ‘reconstruction’ focused on recruiting more defined cohorts that are identified by specific biomarkers that are related to particular disease mechanisms (but they don’t give many details regarding this in the summary).
Next the summary looked at lessons learnt from other fields of medicine.
And the first example they give is cystic fibrosis.
What is cystic fibrosis?
Cystic fibrosis is a genetic/inherited condition in which the lungs and digestive system become clogged with a lining of thick mucus. Long-term issues include difficulty breathing and frequent lung infections.
Cystic fibrosis is caused by genetic mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene.
Like Parkinson’s, there is no cure for cystic fibrosis, but several treatment methods have been developed and the management of the condition has improved significantly over the past 70 years – in the 1950s infants born with the condition were unlikely to survive beyond their first year, while today they are likely to live well into adulthood.
Watch this video for an explanation of cystic fibrosis:
One of the first FDA approved treatment for cystic fibrosis that provided disease-modification benefits was lumacaftor/ivacaftor, which was approved by the US FDA in 2015. It improves lung function by about 10% in a small, genetically defined portion of the cystic fibrosis-affected community (in people with the F508del mutation), but it represents a proof of concept that genetically subtyped conditions can be modified therapeutically. And subsequent research has built on this success with combinations of drugs further improving the beneficial effects.
The summary also stressed the importance of the Therapeutic Development Program of the Cystic Fibrosis Foundation in achieving the goal of disease modification in cystic fibrosis.
One of the presenters at the Krembil Knowledge Gaps meeting was Prof Steven Rowe, Director of the Gregory Fleming Cystic Fibrosis Research Center at the University of Alabama at Birmingham.
He provided the attendees with an overview of the steps and developments that proved to be game changers in providing better treatments for the CF community.
Here is a video of Prof Rowe talking about cystic fibrosis research:
While it provides a nice example of success in disease modification, the comparison between cystic fibrosis and Parkinson’s is not ideal. The former represents a condition characterised by a single gene, while Parkinson’s is very loosely associated with any one of 80+ genetic risk factors in about 20% of cases (Click here to read a previous SoPD post on the topic of PD genetics).
But the presentation of this case study at the Krembil Knowledge Gaps meeting served to stimulate the attendees into discussing the need for better defined subtypes of “Parkinson’s” that can be more clearly targeted therapeutically.
To be fair, we are already seeing this in the research field – for example the Prevail Therapeutics clinical trial which is introducing the correct version of a gene (GBA) using gene therapy in a genetically defined group of people with Parkinson’s (GBA-associated PD – click here to read a previous SoPD post on this topic).
The milestones that allowed for this progress in cystic fibrosis were the use of highly specific cell-based screening and biomarker platforms, identification of a treatment that would work in a defined, genetic mutation cohort, and then multi drug combination of potentiator and corrector therapies with potential for synergism that was evaluated from the outset.
A second case study of lessons learnt from other fields of medicine in the summary was the research conducted on breast cancer.
What is breast cancer?
Very simply, breast cancer is cancerous growth of the breast tissue.
It occurs in females 100 times more frequently than males, and there are different, but well characterised types. Knowing the type of breast cancer an individual has, helps doctors to plan the most appropriate treatment.
 Source: EverydayHealth
Source: EverydayHealth
And advances in treatments has certainly improved outcomes for breast cancer. Prognosis vary depending on the cancer type, geography, the stage of disease, and personal characteristics (such as age), but the five-year survival rates in England and the US have climbed to between 80 and 90% today.
Dr Philippe Bedard, an oncologist at the Princess Margaret Cancer Centre and Co-Principal Investigator of the OICR-supported Ontario-wide Cancer TArgeted Nucleic Acid Evaluation (OCTANE) study, was one of the speakers at the Krembil Knowledge Gaps meeting.
Here is a video of him discussing more personalised cancer therapies:
The summary of the Krembil Knowledge Gaps meeting pointed out the importance of post-hoc analysis of negative clinical trial data in the determining of particular sub-groups in breast cancer – which ultimately led to successful treatment of individuals with particular characteristics.
The attendees at the meeting discussed 3 developmental milestones surounding the progression of breast cancer research, including
- the discovery and recognition of a molecular basis for disease heterogeneity
- subtyping individuals based on tests based on 1., and
- clinical trials with targeted drug treatments based on 1.
After exploring these two case studies, the summary report moved on to discussing a number of “Conceptual roadblocks in the future application of these lessons to Parkinson’s”. And they began with the question: Should pathology remain the gold standard for Parkinson’s diagnosis and biomarker development?
This is basically a disguised way of saying “Are we too obsessed with alpha synuclein?”
Remind me again, what is alpha synuclein?
Alpha synuclein sounds like a distant galaxy, but it is one of the most common proteins in our brains. It makes up about 1% of all the protein in a neuron.
It is also associated with Parkinson’s.
Individuals with specific genetic variations in the alpha synuclein gene have a higher risk of developing Parkinson’s. In addition, when a neuropathologist makes an examination of the brain of a person who passed away with the clinical features of Parkinson’s, there are two characteristic hallmarks that they will be looking for in order to provide a final definitive diagnosis of the condition.
 Neuropathologists conducting a gross examination of a brain. Source: NBC
Neuropathologists conducting a gross examination of a brain. Source: NBC
1. They are looking for the loss of specific populations of cells in the brain, such as the dopamine producing neurons in a region called the substantia nigra, which lies in an area called the midbrain (at the base of the brain/top of the brain stem).
 The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinson’s brain (right). Source:Memorangapp
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinson’s brain (right). Source:Memorangapp
And,
2. They are looking for dense, circular clusters (or aggregates) of alpha synuclein protein within cells, which are called Lewy bodies.
 A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
What is a Lewy body?
A Lewy body is referred to as a cellular inclusion (that is, ‘a thing that is included within a whole’), as they are almost always found inside the cell body. They generally measure between 5–25 microns in diameter (5 microns is 0.005 mm) thus they are tiny, but when compared to the neuron within which they reside they are rather large (neurons usually measures 40-100 microns in diameter).
 A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
Aggregated alpha synuclein protein makes up a significant portion of each Lewy body.
Ok, these sound like “first principles” of Parkinson’s: Loss of dopamine neurons, and Lewy bodies. What’s the problem?
Well, not all cases of Parkinson’s have loss of dopamine neurons.
And not all cases of Parkinson’s have lewy bodies.
Que?
As discussed in the summary, a significant proportion of individuals with genetic risk factors for Parkinson’s (such as Leucine-rich repeat kinase 2 (or LRRK2) and Parkin) will have loss of substantia
nigra dopamine neurons and the associated motor problems that are consistent with Parkinson’s, but they wil have no Lewy bodies (and we recently explored this in a SoPD post – click here to read that).
In addition, there are individuals with Parkinson’s who are known as SWEDDs. Nothing to do with Sweden.
SWEDDS stands for “Scans without Evidence of a Dopaminergic Deficit”. These folks have Parkinson’s-like features, but no evidence of dopamine cell loss according to brain imaging.
These simple observations beg the question: Should pathology remain the gold standard for Parkinson’s diagnosis and biomarker development?
Interesting. What else do they discuss?
They also looked at the “Challenges to clinical trials of disease modifying drugs“, or the issues of matching the “what” to the “who” and also the “how” and the “when”.
And here the discussion covered a lot of different areas. For example, it explored the practicalities of stratifying participants in a clinical trial based on specific characteristics (eg. genetic variations) versus generalization to all people with Parkinson’s.
And here there was consideration given to the idea that GBA- and LRRK2-targetting therapies may be useful not only to individuals with genetic variations in these genes, but also to the wider affected community (Click here to read a previous SoPD post exploring this idea in the context of LRRK2).
The summary also covered the “what” with regards to testing individual therapies vs combinations. But the authors were quick (and right) to point out that success with a single intervention will be required before a targeted cocktail of therapies can be tested (as was the case for cystic fibrosis). And for these therapies (single or combinations), defining a therapeutic window of opportunity for each agent may add further complexity.
One interesting aspect of the summary that was given consideration attention was the idea of adaptive clinical trial designs.
What are adaptive clinical trial designs?
Adaptive clinical trials are studies that allow the for the reviewing of trial data as it accumulates during ongoing clinical trials. And by assessing progress midway through a trial, there is the potential to modify trial characteristics, such as stopping treatments that are not having an effect and shifting participants to ones that are. You can adapt the trial as it is occurring.
A common form of adaptive clinical trial is referred to as a multi-arm, multi-stage (MAMS) platform trial. These compare multiple drugs at the same time – stopping any drugs that fail to demonstrate benefits, while continuing with those that do.
 Multi-arm, multi-stage (MAMS) platform trial. Source: Eupati
Multi-arm, multi-stage (MAMS) platform trial. Source: Eupati
Parkinson’s has not had an adaptive trials yet – they are large and extremely costly – but there have been examples of multiple arm studies, such as the ongoing Austrialian Parkinson’s Mission effort (Click here to read more about this).
 Source: APM
Source: APM
One of the best examples of an adaptive clinical trial is called I-SPY – which are focused on cancer.
 Source: ispytrials
Source: ispytrials
I-SPY (“Investigation of Serial studies to Predict Your therapeutic response with imaging and molecular analysis”) is an adaptive trial design that has enabled two experimental breast cancer drugs to deliver promising results after just six months of clinical testing (those drugs being Veliparib and Neratinib – Click here to read more about this).
The value of the I-SPY trials, however, go well beyond the clinical results, as they have given proof of principle validation to the concept, resulting in a host of additional adaptive clinical trial projects for various medical conditions, such as Prostate cancer (the Stampede trials), Multiple Sclerosis (the MS-Smart trial) and Alzheimer’s (the EP-AD consortium).
Very interesting. Have other Parkinson’s researchers been exploring these ‘first principles’ sort of ideas?
Yes.
One of the main ‘first principles’-kind of effort for Parkinson’s is the “Aligning Science Across Parkinson’s” (or ASAP) initiative.
What is ASAP?
Funded by the Sergey Brin (think: Google co-founder) family foundation, ASAP “will allocate financial resources that significantly enlarge the basic science effort currently supported by the government and private foundations with the goal of understanding the underlying biology of Parkinson’s“.
The program is seeking to speed up Parkinson’s research by going back to basic science and taking a different look at Parkinson’s. By doing so, we will hopefully be able to establish some “first principles” of “Parkinson’s”.
We have previously dicsussed ASAP (Click here to read a previous SoPD post on this topic).
So what does it all mean?
The Krembil Knowledge Gaps meeting looks like it was a fascinating, multifaceted discussion of various aspects of Parkinson’s research (Click here for a copy of the agenda). It is a shame that the presentations & discussion were not recorded/streamed and made openly available, but it will hopefully stimulate similar types of events to be conducted. From discussions with attendees, I was particularly pleased to hear that members of the Parkinson’s community were present and vocal in the discussions (including Toronto local Ben Stecher).
 The man, the myth, the legend. Source: McGill
The man, the myth, the legend. Source: McGill
It would be easy to assume from this post (and the projects described within) that we are at some kind of cross roads in our understanding of the condition we know as “Parkinson’s”. But this statement belies an underlying truth: we are always at cross roads in our understanding of things. With each new discovery, we need to revise/reconstruct our interpretation of things.
Being 7 cans short of a 6 pack (intellectually, speaking), I am certainly not well placed to argue with Aristotle – one of the first acknowledged proponents of first priniciples – but the concept of first principles outside of the realms of maths, engineering and philosophy sounds a little romantic to me. I struggle to imagine what these fundamental truths for medical conditions would look like given the personal nature of ailments. They are certainly worthy goals, but I suspect significant progress can be made before we truly understand any basic truths.
For example, inoculation for smallpox has been conducted for centuries in China (see Zhang Yan’s Zhongdou Xinshu (種痘新書), or New book on smallpox inoculation, written in 1741), but it is only recently (the last 50 years) that we have begun to understand the biology underlying the condition. This suggests that progress can be made, even for complex conditions like Parkinson’s. And to re-evaluate/revise our foundational beliefs on a particular condition should not be considered as a sign of weakness or defeat, but rather a healthy exercise.
One to be performed regularly.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from BCU






Simon
I perhaps expected too much from this meeting, but the conclusion seems somewhat lacking; that we should ” re-evaluate/revise our foundational beliefs” at frequent intervals”. The meting seems to have addressed the right quwstions without proposing any answers.
I was surprised at y9ur self description as “7 cans short of a 6 pack (intellectually…). you must have a mega intellect to be able to retain so much and produce the quantity if output that you do.
Regards Keith
LikeLike
Very kind of you Keith, but sadly none of it sticks in my old noggin. I will often find an interesting drug online and do a google search for it in the context of PD, only to discover that I wrote a post about it 6 months ago 🙂
Regarding the meeting, I agree that there were few answers in the summary, but I would add that we need to lower our expectations. This was the first of these types of meetings and it was more about clarifying the questions than proposing answers. It is an effort in the right direction though.
Kind regards,
Simon
LikeLike