|
# # # # Inflammation is the means by which tissue in our bodies communicate with the immune system to indicate when something is wrong. Tiny messenger proteins are released from stressed or damaged cells to alert neighbouring cells of their situation. Ailing cells can also release additional components – such as DNA – that can activate immune cells and cause inflamation. Recently, researchers have identified both messenger proteins and specific types of DNA that are present in the blood of individuals with a genetically-associated sub-type of Parkinson’s. The discovery could provide both novel biomarkers, but also point towards specific biological pathways that could be therapeutically targetted. In today’s post, we will review this new research. # # # # |
When cells in your body are stressed, damaged, or sick, they begin to release large amounts of tiny messenger proteins which inform the rest of your body that something is wrong.
When enough of these messenger proteins are released, cells of the immune system will become activated, and come looking for the source of the trouble.
This is inflammation.
 Source: Youtube
Source: Youtube
Inflammation is a critical part of the immune system’s response to problems. It is the body’s way of communicating with the immune system and explaining that something is wrong. This also aid in activating the immune system so that it can help deal with the situation.
By releasing the messenger proteins (called cytokines), injured/sick cells kick off a process that results in multiple types of immune cells entering the troubled area of the body and undertaking very specific tasks.
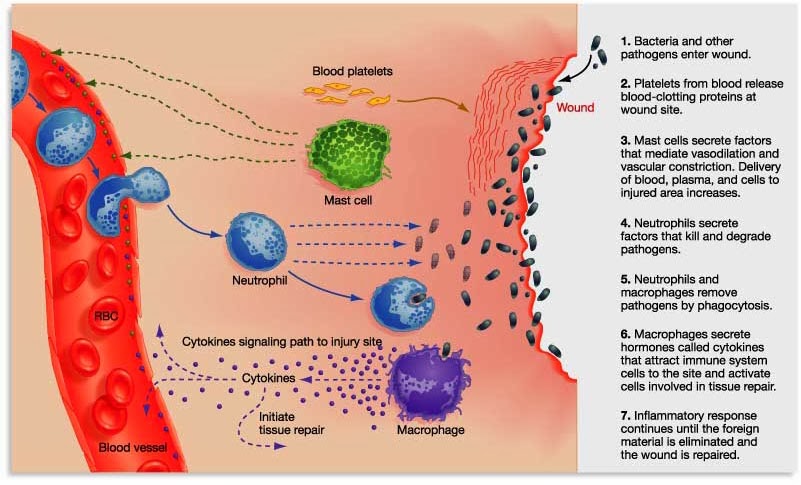 The inflammatory process. Source: Trainingcor
The inflammatory process. Source: Trainingcor
The strength of the immune response depends on the volume of the signal arising from those released messenger proteins.
For a long time, it has been hoped that some of these messenger proteins might be useful as biomarkers for conditions like Parkinson’s. And recently, researchers have published data suggesting that they might have found one cytokine that could be very useful for a specific sub-set of people with Parkinson’s.
What did they find?
This report was published this week:
 Title: Mitochondrial damage-associated inflammation highlights biomarkers in PRKN/PINK1 parkinsonism
Title: Mitochondrial damage-associated inflammation highlights biomarkers in PRKN/PINK1 parkinsonism
Authors: .
Journal: Brain, Oct 8; awaa246. Online ahead of print.
PMID: 33029617 (This report is OPEN ACCESS if you would like to read it)
In this report, the researchers were interested in the Parkinon’s associated proteins PARKIN and PINK1.
What are PARKIN and PINK1?
PARKIN and PINK1 are two proteins that play critical roles in a process called mitophagy.
And what is mitophagy?
Mitophagy involves the disposal of old or dysfunctional mitochondria.
And before you ask, mitochondria are the power stations of each cell. They help to keep the lights on. Without them, the party is over and the cell dies.

Mitochondria and their location in the cell. Source: NCBI
You may remember from high school biology class that mitochondria are tiny bean-shaped objects within the cell. They convert nutrients from food into Adenosine Triphosphate (or ATP). ATP is the fuel which cells run on. Given their critical role in energy supply, mitochondria are plentiful (some cells have thousands) and highly organised within the cell, being moved around to wherever they are needed.
Like you, me and all other things in life, mitochondria have a use-by date. And as mitochondria get old and worn out (or damaged) with time, the cell will dispose of them via the process of mitophagy.
How are PARKIN and PINK1 involved in mitophagy?
In the process of mitophagy, PINK1 acts like a kind of handle on the surface of mitochondria. In normal, healthy cells, the PINK1 protein attaches to the surface of mitochondria and it is slowly absorbed until it completely disappears from the surface and is degraded. In unhealthy cells, however, this process is inhibited and PINK1 starts to accumulate on the outer surface of the mitochondria. Lots of handles poking out of the surface of the mitochondria.
Now, if PINK1 is a handle, then PARKIN is a flag that likes to hold onto the PINK1 handle. While exposed on the surface of mitochondria PINK1 will grab on to the PARKIN protein. This pairing is a signal to the cell that this particular mitochondrion (singular) is not healthy and needs to be removed. The pairing starts the process that leads to the development of the phagophore (a sack-like structure that surrounds the damaged/old mitochondria and degrades it) and eventually mitophagy.

Pink1 and Parkin in normal (right) and unhealthy (left) situations. Source: Hindawi
In the absence of normal PINK1 or PARKIN proteins, there is no handle-flag system and old/damaged mitochondria start to pile up. They are not disposed of appropriately and as a result the cell gets sick and ultimately dies.
Now as I said above, these two proteins are associated with Parkinson’s. Some people carry particular tiny genetic variations in the regions of DNA that provide the instructions for making PINK1 or PARKIN (these regions are called genes). As a result of these genetic mutations, these individuals are more vulnerable to developing an early onset form of Parkinson’s. It has been proposed that the dysfunctional disposal of (and accumulation of) old mitochondria is part of the reason why these individuals develop the condition at such an early age.
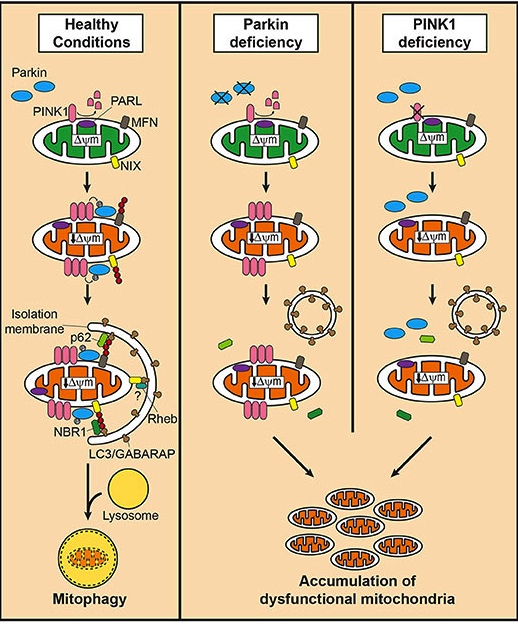 Mitophagy. Source: Frontiersin
Mitophagy. Source: Frontiersin
|
# RECAP #1: PARKIN and PINK1 are two Parkinson’s-associated proteins that are involved with the removal of old/damaged mitochondria (the powerstations of cells). People with genetic variations in the regions of DNA that make PINK1 and PARKIN are more vulnerable to developing early onset forms of PD. In the absence of normal functioning PARKIN or PINK1 proteins, mitochondria are believed to start accumulating inside cells and causing cellular stress, which may ultimately lead to cell death. # |
Ok, so that explains PINK1 and PARKIN, but what was the messenger protein (or cytokine) that the researchers were interested in?
The researchers were interested in a cytokine called IL-6, and they wanted to investigate it within the context of the Parkinson’s-associated proteins PINK1 and PARKIN.
What is IL-6?
Interleukin 6 (or IL-6) is a protein that acts as a pro-inflammatory cytokine, meaning that it is a messenger protein that spreads the ‘something is wrong’ signal to the immune system.
This video provides a good introduction to IL-6 and it functions:
Interesting, but why were they interested in this particular cytokine?
In a previous research report (which we discussed here on the SoPD – click here to see that post) the investigators noticed something interesting about IL-6.
This was the report:
 Title: Parkin and PINK1 mitigate STING-induced inflammation
Title: Parkin and PINK1 mitigate STING-induced inflammation
Authors: Sliter DA, Martinez J, Hao L, Chen X, Sun N, Fischer TD, Burman JL, Li Y, Zhang Z, Narendra DP, Cai H, Borsche M, Klein C & Youle RJ
Journal: Nature, 2018, Sep;561(7722):258-262.
PMID: 30135585 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that when they exercised different types of mice to exhaustion, the mice that were genetically engineered to have variations in their PINK1 or PARKIN genes exhibited massively elevated levels of IL-6, not only immediately after exercise, but also 24 hours later:
 Source: PMC
Source: PMC
Interestingly, when the researchers looked at blood samples from humans with PINK1 and PARKIN genetic variations, they also noticed elevations in IL-6:
 Source: PMC
Source: PMC
In the graph above, IL-6 levels were analysed in healthy controls (HC), people with Parkinson’s who have a genetic variation in one copy of their PINK1 gene (PINK1 heterozygotes or P1H), people without Parkinson’s who have a variation in one copy of their PARKIN gene (unaffected PARKIN heterozygotes or UPH), people with Parkinson’s who have variations in both copies of their PARKIN gene (affected PARKIN biallelic or APB), and idiopathic PD patients (IPD).
NOTE: Remember that in our DNA we have two copies of each gene – one from your mother and one from your father.
It was of interest to the researchers that some people in the different sub-types of Parkinson’s had evelated levels of IL-6, and they wanted to investigate this further.
|
# # RECAP #2: IL-6 is a messenger protein (or a cytokine) that communicates with the immune system. It is a pro-inflammatory cytokine, meaning that it is telling the immune system that something is wrong and this activates the immune system. Data suggests that some people with Parkinson’s have elevated levels of IL-6. # # |
So what did the researchers do next?
The investigators analysed levels of IL-6, C-reactive protein, and circulating cell-free mtDNA in the blood of 245 individuals who take part in assessments at two independent movement disorder research centres.
Hang on a second! What is C-reactive protein?
C-reactive protein (or CRP) is a protein made by your liver and sent out into your bloodstream in response to inflammation. It is used as a biomarker of inflammation.
 CRP. Source: Wikipedia
CRP. Source: Wikipedia
CRP floats around the blood system, attaching itself to the surface of dying cells and some bacteria. This attracts the attention of immune cells, like macrophages which come in and remove these items (via phagocytosis – basically ‘engulfing and digesting’ stuff).
Ok, and what about circulating cell-free mtDNA? What is that?
A curious feature of mitochondria is that while they live inside of cells, they actually have their own DNA. This DNA is refered to as mitochondrial DNA (or mtDNA) and is completely separate to the DNA in the nucleus of the cell (or nucleic DNA).
This video explains more about mitochondrial DNA:
When mitochondria are damaged, they will sometimes release mtDNA.
This “free” mtDNA can then also be released from the particular cell that the mitochondria lives in (perhaps if the cell is damaged), and this mtDNA is referred to as circulating cell-free mtDNA. It is relatively robust stuff and can be found floating around in the blood system.
Got it. So the investigators looked at the blood of 245 individuals for IL-6, C-reactive protein, and circulating cell-free mtDNA. What did they find?
They reported elevated IL-6 levels in people with genetic variations in both copies of their PINK1 or PARKIN genes (referred to as biallelic mutations). There were 23 people with biallelic mutations in their PARKIN gene and 11 people with biallelic mutations in their PINK1 gene. And this result was confirmed in both a German cohort and an Italian cohort (data from two independent research groups):
![]() Source: Brain
Source: Brain
There was also a correlation between IL-6 levels and disease duration (or the time since diagnosis) in individuals with PARKIN or PINK1 mutations. Curiously, there was no such association in the idiopathic (or spontaneous) Parkinson’s group. The researchers proposed that might suggest the potential of IL-6 as a biomarker of disease progression in PARKIN or PINK1-associated Parkinson’s.
There was a “trend towards increased CRP levels” in individuals with PARKIN- or PINK1-associated Parkinson’s (compared to the control samples), but no statistical difference was seen between the various groupings.
The researchers did, however, report a significant increase in circulating cell-free mtDNA serum levels in individuals with PARKIN or PINK1-associated Parkinson’s (regardless of whether they have genetic mutations in one or both copies of their genes):
![]() Source: Brain
Source: Brain
Importantly, the circulating cell-free mtDNA levels in individuals with idiopathic Parkinson’s and those without Parkinson’s (but who carry a PARKIN or PINK1 mutation) were comparable to control levels. It would be interesting to follow a cohort of people with a PARKIN or PINK1 mutation over time to see if (and how) circulating cell-free mtDNA levels change over time.
Based on these results, the researchers proposed that circulating cell-free mtDNA levels may be useful as a biomarker in discriminating between idiopathic Parkinson’s and PARKIN- or PINK1-associated Parkinson’s. And the data also “implicates inflammation due to impaired mitophagy and subsequent mtDNA release in the pathogenesis” of PARKIN- or PINK1-associated Parkinson’s.
 Source: MedicalPress
Source: MedicalPress
|
# # # RECAP #3: Researchers have found that both IL-6 and circulating cell-free mtDNA levels were elevated in the blood of individuals with PARKIN- or PINK1-associated Parkinson’s. There was also a correlation between IL-6 levels and disease duration. These results not only point towards potential biomarkers, but also implicate inflammation as playing an important role in PARKIN- or PINK1-associated Parkinson’s. # # # |
How does high levels of mtDNA affect the immune system?
An interesting feature of circulating cell-free mtDNA is that the immune system can register it, and this can cause an inflammatory response (as the immune system will assume something is wrong if mtDNA is not where it is supposed to be – inside the mitochondria).
And this response from the immune system to circulating mtDNA has been reported by numerous research groups (Click here and here to read review articles about this).
The circulating mtDNA appears to be able to activate different proinflammatory pathways, including the inflammasome, toll-like receptors (TLR), and the cGAS-STING pathway:
 Source: EMBO
Source: EMBO
We have previously discussed the inflammasome on the SoPD (Click here to read a post about that), but of particular interest to the researchers in the report we are reviewing today was the cGas-STING pathway.
What is the cGas-STING pathway?
Stimulator of interferon genes (or STING) is a signalling molecule that sits on the outer surface of the endoplasmic reticulum – the part of the cell where proteins are produced. The activation of STING leads to the production of messenger protein interferon (Type I IFNs in the image below), which then exit the cell, and bind to the cell’s own interferon (IFN) receptor. And activation of the IFN receptor leads to the activation of pro-inflammatory genes.
 The STING pathway. Source: Nimbus
The STING pathway. Source: Nimbus
It is a round-about kind of process, but it helps to cause only affected cells to alert the immune system that something is wrong (Click here for a good open access review of STING).
And what about cGAS?
So, Cyclic guanosine monophosphate (GMP)-AMP synthase (or cGAS) is a protein that acts as a sensor (or detector) of DNA floating around inside the cell. Upon DNA binding to cGAS, the activated cGAS initiates the production of cyclic GMP-AMP (or cGAMP). cGAMP then acts as a messenger protein and activates STING.
Is anyone looking at inhibiting cGAS or STING in humans?
Yes, there are biotech firms currently trying to develop STING inhibitors (or antagonists).
For example, a company called IFM Therapeutics:
 IFM Therapeutics was bought by the pharmaceutical company Bristol-Myers Squibb in 2017, but they are continuing to develop antagonists of the STING pathway.
IFM Therapeutics was bought by the pharmaceutical company Bristol-Myers Squibb in 2017, but they are continuing to develop antagonists of the STING pathway.
When these companies have STING antagonists ready for clinical testing, it may be interesting to have a look at whether these compounds could be useful in Parkinson’s.
And what about cGAS?
There is evidence suggesting that acetylsalicylic acid is effective at inhibiting cGAS.
Acety-what?
Aspirin.
 Source: ox.ac.uk
Source: ox.ac.uk
In early 2019, this report was published:
 Title: Acetylation Blocks cGAS Activity and Inhibits Self-DNA-Induced Autoimmunity.
Title: Acetylation Blocks cGAS Activity and Inhibits Self-DNA-Induced Autoimmunity.
Authors: Dai J, Huang YJ, He X, Zhao M, Wang X, Liu ZS, Xue W, Cai H, Zhan XY, Huang SY, He K, Wang H, Wang N, Sang Z, Li T, Han QY, Mao J, Diao X, Song N, Chen Y, Li WH, Man JH, Li AL, Zhou T, Liu ZG, Zhang XM, Li T.
Journal: Cell. 2019 Mar 7;176(6):1447-1460.e14.
PMID: 30799039 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that a process called acetylation (substitution of an acetyl group for an active hydrogen atom on a compound) suppresses cGAS activity. So they went looking for drugs able to acetylate proteins.
And that search led them to aspirin – a non-steroidal anti-inflammatory drug that is known to acetylate proteins (such as cyclooxygenase or COX).
Using both cell culture and mouse models of autoimmune conditions, the investigators reported that aspirin treatment inhibited cGAS-mediated production of interferon.
I have a PARKIN variant. Should I start taking aspirin?
No, you shouldn’t.
First, all of the research discussed in today’s post needs to be independently replicated before we can make any conclusions or judgements. In addition, we have no idea if aspirin has any effect on inflammation in the context of a PARKIN or PINK1 variants.
Second, the researchers used aspirin at a high concentration (4 mM) in their cell culture experiments. Doses of aspirin resulting in serum concentrations of ≥3 mM can cause complications (salicylate poisoning, gastrointestinal symptoms, tinnitus and other adverse effects). In their mouse models, the researchers used a lower dose (1.5 mM), but chronic use of aspirin at this concentration carries the risk of causing problems like gastrointestinal bleeding.
Long story short, there is a lot we don’t yet know and more research is required.
For those interested, we have previously discussed aspirin on the SoPD (Click here to read that post), but please discuss with your clinician before considering any changes to your current treatment regime.
So what does it all mean?
Researchers have identified Interleukin 6 (or IL-6) and circulating cell-free mitochondrial DNA as being elevated in particular sub-types of Parkinson’s – those associated with genetic variations in the PARKIN and PINK1 genes. These findings not only point towards potential biomarkers, but also indicate that inflammation could be an important factor in these specific types of Parkinson’s.
Anti-inflammatory therapeutics are now being developed and tested in Parkinson’s, and it could be interesting if sub-type analyses (focused on genetics and blood-based biomarkers) could be conducted in the clinical trials evaluating these novel agents.
The results discussed in today’s post need to be independently replicated and further expanded on, but they add to the growing body of research that is leading us towards a future of more “precision medicine”-based therapies for Parkinson’s.
Some of which may take some of the STING out of PD.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, many of the companies mentioned on this website are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with any of the companies or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from ScienceMag


It be interesting to know of your of your opinion on ibuprofen in this pathway along with other agents that globally reduce inflammation
LikeLike
Hi M,
We discussed ibuprofen in a SoPD post over the summer (https://scienceofparkinsons.com/2020/08/16/ibuprofen-2/). Hope this helps.
Kind regards,
Simon
LikeLike
Thank you for these reports and summaries. They are masterfully written and a valuable resource.
LikeLike
Thanks for your comment and kind words Cha – much appreciated.
Kind regards,
Simon
LikeLike