|
# # # # A build up of “PARkin Interacting Substrate” (or PARIS) protein has been proposed as one potential mediator of the pathology observed in some cases of Parkinson’s. The accumulation of this protein leads to the inhibition of a key protein called PGC-1α, which is a neuroprotective protein that helps to keep cells alive. For sometime, researchers have been searching for molecules that can act as inhibitors of PARIS, in the hope that blocking PARIS would allow PGC-1α to act freely. Such an agent could have potential as novel treatment for Parkinson’s. This week a research report was published that describes one possible PARIS inhibitor. It is called farnesol. In today’s post, we will look at the biology behind PARIS, review the new report, and discuss what exactly is known about farnesol. # # # # |
 Pont Royal et Musée d’Orsay. Source: Wikipedia
Pont Royal et Musée d’Orsay. Source: Wikipedia
Paris has a special place in my heart for several reasons.
The main one: I proposed to my wife there on the Pont Royal.
We had planned a day out in London, but once we got down to Waterloo “for lunch at a special restaurant“, I surprised her with two Euro Star tickets and we were off on the train for Paris – just like that (I might look the hardened tough guy on the outside, but deep down I am really just a tragic romantic).
And that night, after “dinner at a special restaurant” shortly before 10pm as we were crossing the middle of the Pont Royal, and a small miracle occurred: the traffic lights stopped traffic in both directions.
 Source: Pixels
Source: Pixels
Seizing our chance moment alone, I dropped to one knee and asked (read: begged).
Now, if she had said ‘no thanks‘, I had a back up plan: Jump over the side of the bridge, float down the Seine some ways, climb out and then join the Foreign Legions the next day as a mute (“je suis muet”).
But she didn’t say no (let’s call that the second small miracle) and thankfully for my fragile ego’s sake there wasn’t a lengthy deliberation.
When the traffic lights changed and traffic started to flow again, we received some enthusiastic honks of the klaxons (horns) as I got up and we headed off to alert our parents. It was a really nice moment.
I was recalling this moment, this week when a different type of Paris was being discussed in the news.
What do you mean “a different type of Paris”?
 Science Translational Medicine (STM) is one of the premiere scientific journals. Careers are made by publishing research in this journal. And this week a paper was published in STM that is worthy of discussion here on the SoPD.
Science Translational Medicine (STM) is one of the premiere scientific journals. Careers are made by publishing research in this journal. And this week a paper was published in STM that is worthy of discussion here on the SoPD.
This is the report:
 Title: PARIS farnesylation prevents neurodegeneration in models of Parkinson’s disease.
Title: PARIS farnesylation prevents neurodegeneration in models of Parkinson’s disease.
Authors: Jo A, Lee Y, Kam TI, Kang SU, Neifert S, Karuppagounder SS, Khang R, Kang H, Park H, Chou SC, Oh S, Jiang H, Swing DA, Ham S, Pirooznia S, Umanah GKE, Mao X, Kumar M, Ko HS, Kang HC, Lee BD, Lee YI, Andrabi SA, Park CH, Lee JY, Kim H, Kim H, Kim H, Cho JW, Paek SH, Na CH, Tessarollo L, Dawson VL, Dawson TM, Shin JH.
Journal: Sci Transl Med. 2021 Jul 28;13(604):eaax8891.
PMID: 34321320
In this study, the researchers wanted to identify inhibitors for a protein called PARIS.
What is PARIS?
PARKIN Interacting Substrate (or PARIS) is a protein that – as the label on the can suggests – interacts with PARKIN.
And what pray tell is PARKIN?
Sometimes referred to as PARK2, PARKIN is a protein that is closely associated with Parkinson’s. Genetic errors in the DNA that provides the plans for making PARKIN protein were the second genetic risk factor to be connected with PD.
 The structure of PARKIN. Source: Wikipedia
The structure of PARKIN. Source: Wikipedia
Genetic variations in PARKIN are particularly associated with early onset Parkinson’s (which are cases diagnosed under the age of 40 years of age).
Biologically speaking, what does PARKIN do?
PARKIN is an enzyme – that is a protein that interacts with other proteins and acts as a catalyst. Specifically, PARKIN functions as an E3 ubiquitin ligase.
What is an E3 ubiquitin ligase?
A ligase is an enzyme that initiates the joining of two molecules. It forms a new chemical bond between them. Inside of cells, PARKIN interacts with lots of proteins and has a role in many different functions.
And this is where we come back to PARIS.
Ok, so what is PARIS?
Parkin Interacting Substrate (or PARIS; also known as Zinc Finger Protein 746 – ZNF746) is a protein that suppresses the transcription of another protein called peroxisome proliferator-activated receptor γ coactivator-1α (PGC-1α).
Sorry, but that does that mean?
“Transcription” is the production of RNA from DNA. This video explains transciption:
Proteins, like PARIS, can bind to specific regions of DNA and by doing block the transcription of particular RNAs – thus reducing the production of that protein.
PARIS is able to block the production of PGC-1α.
Think of PARIS as the handbrake on the production of PGC-1α.
PGC-1α is a protein that activates many different neuroprotective pathways in cells. Reductions in levels of PGC-1α are actually associated with all kinds of issues in the body (not just the brain).
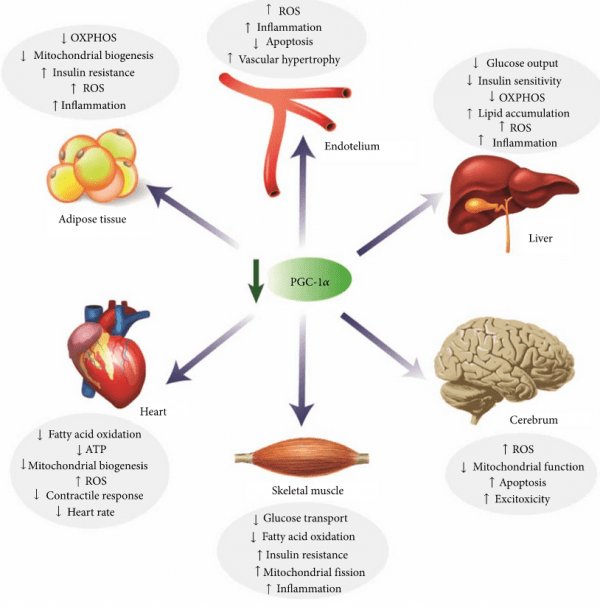 Thus, limiting the ability of PARIS to block the production of PGC-1α could be a good thing for a variety of medical conditions.
Thus, limiting the ability of PARIS to block the production of PGC-1α could be a good thing for a variety of medical conditions.
Taking the hand brake off a neuroprotective pathway.
Ok. But what happens to PARIS in Parkinson’s?
A decade ago, a group of researchers led by Prof Ted Dawson at John Hopkins School of Medicine published this study:

Title: PARIS (ZNF746) repression of PGC-1α contributes to neurodegeneration in Parkinson’s disease.
Authors: Shin JH, Ko HS, Kang H, Lee Y, Lee YI, Pletinkova O, Troconso JC, Dawson VL, Dawson TM.
Journal: Cell. 2011 Mar 4;144(5):689-702.
PMID: 21376232 (This article is OPEN ACCESS if you would like to read it)
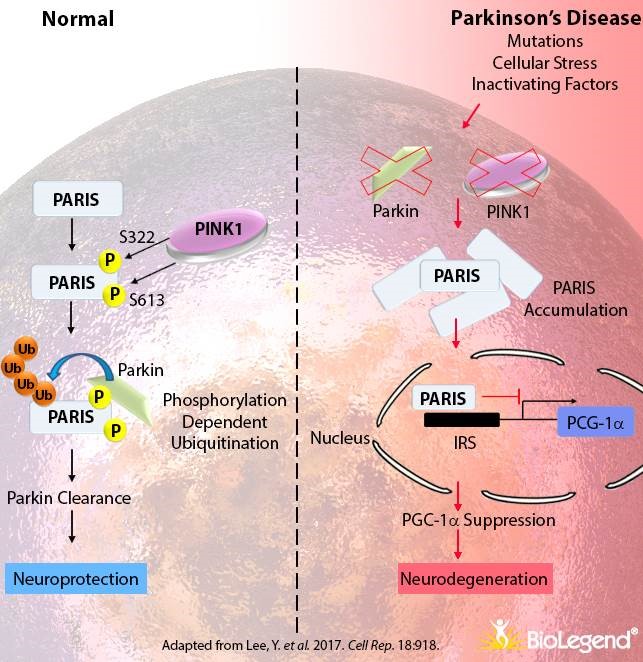
In this study, the researchers noticed that when they removed PARKIN from cells, PARIS would start to accumulate. They discovered that PARKIN was important for binding to and labelling old PARIS protein for disposal. Thus, in the absence of PARKIN, PARIS protein would not be disposed of and simply start to pile up.
They also noticed that this build up of PARIS resulted in the loss of dopamine neurons (a set of cells in the brain that are particularly vulnerable and severely affected in Parkinson’s) in mice that did not produce PARKIN. When the researchers re-introduced normal PARKIN protein, the researchers were able to rescue the cell loss. Interestingly, the researchers also found that over-production of PARIS in normal mice resulted in cell loss which could be rescued by a similar over-production of PARKIN.
The study got really interesting though when the investigators looked at postmortem human brains of people who passed away with Parkinson’s. They found that levels of PARIS protein were more than two times higher in regions affected by Parkinson’s (specifically, the striatum and the substantia nigra) when compared to healthy controls. And this was in people with sporadic idiopathic Parkinson’s (which had no potential genetic cause). And interestingly, this increase was only seen with PARIS protein, and not PARIS RNA (where the scientists saw no difference in RNA levels with control samples), suggesting a potential problem in the disposal of PARIS protein in the Parkinsonian brain.
|
# EDITOR’S SIDE NOTE HERE: Some readers might remember a recent SoPD post in which we looked at age associated changes in PARKIN protein (Click here to read that post). In that post, we reviewed a manuscript (which has now been published) that reported a fascinating discovery: As we age, PARKIN protein becomes less available. In fact, by the time we turn 50 years of age, “Parkin is largely insoluble”, meaning that the majority of the protein is no longer able to do its job. The shift appears to involve oxidation. But this “shift” could explain the increase of PARIS levels in the brains of people with idiopathic Parkinson’s reported in the 2011 study we are currently discussing: As we age, levels of active PARKIN protein drop, leaving PARIS to accumulate, and wreak havoc. # |
The investigators concluded that this meant PARIS was could be playing a role in the cell loss associated with Parkinson’s, and it suggested to them that an inhibitor of PARIS could potentially be a useful therapeutic agent for PD.
 Source: Nordicbiosite
Source: Nordicbiosite
|
# RECAP #1: PARIS is a protein that blocks the production of another protein (called PGC-1α) that is involved in activating neuroprotective pathways. In the brains of people with Parkinson’s, PARIS appears to accumulate and this could lead to less neuroprotective activity from PGC-1α. This observation has led scientists to search for molecules that will inhibit PARIS (blocking it from blocking PGC-1α production). # |
So they went looking for a molecule that would inhibit PARIS?
Yep.
They did this by screening for anything that would elevate levels of PGC-1α in the presence of PARIS.
Why did they do that? “in the presence of PARIS”?
There are lots of molecules that are known to elevate levels of PGC-1α.
But the investigators were assuming that PARIS would be competitively blocking the transcription of PGC-1α. So if a molecule was going to be really useful, it would have to block PARIS. Thus, their screening study was conducted in the presence of PARIS.
They engineered cells that make a green fluorescent protein as PGC-1α levels increase – making it easy to identify compounds that increase PGC-1α (basically the experiment was easy: all they had to do was look for the green cells).
Next they screened a library of 230,000 compounds (yes, you read that correctly: 230,000 compounds!). From all of those screening experiments, they identified 17 agents that caused a 2.5-fold increase in green fluorescence in cells (without causing any cellular toxicity). Of these compounds, three agents (AVS-3648, ABT-0529, and farnesol) were predicted to have a high likelihood of accessing the brain.
Accessing the brain?
The brain is surrounded by a protective membrane called the blood brain barrier. It keeps harmful chemicals, pathogens and toxins from entering the carefully controlled environment of the brain. But it appears to be too good at it, as it also blocks many medications from entering the brain from the blood stream.
This video provides a basic explanation of the blood brain barrier:
Based on it’s molecular structure, the researchers predicted that farnesol treatment could increased levels of PGC-1α in the brain.
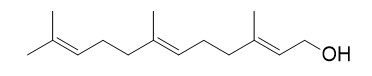
What exactly is farnesol?
Farnesol is an organic molecule produced by plants & mammals, that was originally extracted from the Farnese acacia tree, Vachellia farnesiana, by perfume makers around 1900.
 Vachellia farnesiana. Source: Wikipedia
Vachellia farnesiana. Source: Wikipedia
It is a colorless, oily liquid with a delicate sweet odor. It was originally used as an enhancer of perfume scent to emphasise the odors of sweet floral perfumes, but it was later found to be present in all eukaryotes (organisms whose cells have a nucleus), and has anti-oxidant, anti-inflammatory, & neuroprotective properties .
While it is widely used in cosmetics, it is important to understand that farnesol has never been clinically tested in humans for any disease.
Right, and they found that farnesol could cross the blood brain barrier?
Yes. The researchers fed mice on a diet supplemented with farnesol for a week and then they analysed the brains to determine levels of farnesol. They found a 37% increase in levels compared to control fed mice. And in the farnesol-fed mice, the investigators found PGC-1α levels had increased in the substantia nigra region of the brain without any change in PARIS levels (the substantia nigra being a region of the brain severely affected by Parkinson’s – it is where most of the dopamine neurons resided).
To determine if farnesol could protect dopamine neurons against an increase in PARIS levels, the researchers feed their farnesol supplemented diet to mice that produce very high levels of PARIS in brain cells (3-10x normal levels). Analyses fond that these mice exhibit significant loss of dopamine neurons (>35%) at a very young age (6 weeks
of age), but a diet high in farnesol prevented this loss of dopamine neurons.
The researchers repeated this experiment in normal mice and used carefully engineered viruses to cause cells to over produce PARIS protein. They injected this virus into the substantia nigra of the mice and over time observed dopamine cell loss. This neurodegeneration was blocked in mice that were fed on a diet high in farnesol.
So how is farnesol inhibiting PARIS?
Farnesylation
Farne…syl… what?!?!?
Farnesylation. It is a biochemical process. It is a method of modifying proteins.
Quite often in biology, for a protein to perform a specific function it must be acted on by another protein, which may change the configuration of the protein or add/subtract something to activate/inactivate it.
Farnesyltransferase is an enzyme that attaches farnesyl groups to other proteins.
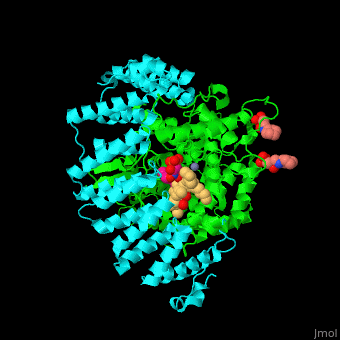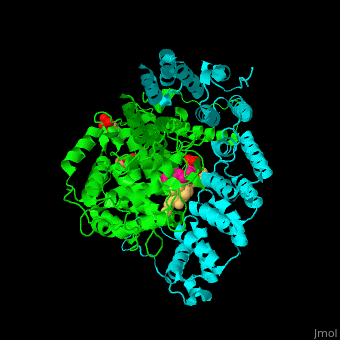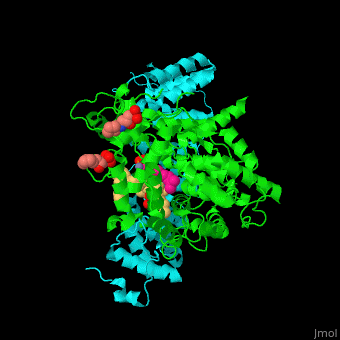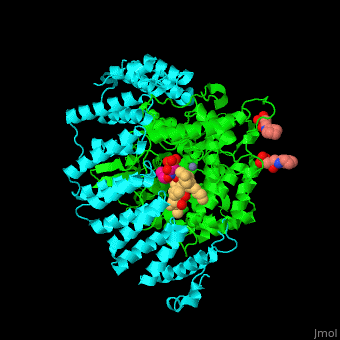 The structure of farnesyltransferase. Source: Proteopedia
The structure of farnesyltransferase. Source: Proteopedia
We have previously discussed farnesylation is a previous SoPD post (Click here to read that post).
Farnesyltransferase uses farnesol to modify the PARIS protein. By increasing farnesol levels, there is a subsequent increase in PARIS farnesylation. And this farnesylation of PARIS prevents it from binding to DNA and inhibiting PGC-1α production.
Interestingly, the investigators reported a significant reduction in PARIS farnesylation in the substantia nigra (the region badly affected in PD) of postmortem brains from people who passed away with Parkinson’s. They also saw elevated levels of PARIS protein and reduced levels of PGC-1α protein.
Interesting. What did the researchers do next?
They tested farnesol in a model of Parkinson’s which involved that introduction of aggregated alpha synuclein protein into the brain.
Alpha synuclein is a protein that accumulates and aggregates in cells in many cases of Parkinson’s. This clustering of the protein is believed to be influential in the cell death associated with Parkinson’s. Interestingly, when the researchers injected aggregated alpha synuclein into the brains of mice, they noted a reduction in levels of PGC-1α, which farnesol treatment restored. They also observed a loss of dopamine neurons, which (again) farnesol treatment rescued.
The researchers concluded their report suggesting that “farnesol may be beneficial in the
treatment of Parkinson’s by enhancing the farnesylation of PARIS and restoring PGC-1α activity”. But they also noted in their discussion that “It will be important to determine the amount of farnesol that can be safely tolerated in humans and its pharmacokinetic properties and whether farnesol protects against the degenerative effects of PD in humans“.
Lots of important questions to be addressed.
|
# # RECAP #2: A large screen of molecules identified farnesol as a direct and potent inhibitor of PARIS. Further experiments in models of Parkinson’s demonstrated neuroprotection as a result of farnesol administration. In addition, elevated levels of PARIS have been observed in the brains of people with Parkinson’s. Farnesol is an organic molecule produced by plants & mammals, with recognised anti-inflammatory properties. But it has never been clinical tested, and we have no idea of the safe or effective doses of farnesol in humans. # # |
Has farnesol ever been reported to have beneficial properties in other models of medical conditions?
Actually it has.
Very recently there was a research report published on the effects of farnesol in a model of multiple sclerosis:
 Title: Farnesol induces protection against murine CNS inflammatory demyelination and modifies gut microbiome.
Title: Farnesol induces protection against murine CNS inflammatory demyelination and modifies gut microbiome.
Authors: Sell LB, Ramelow CC, Kohl HM, Hoffman K, Bains JK, Doyle WJ, Strawn KD, Hevrin T, Kirby TO, Gibson KM, Roullet JB, Ochoa-Repáraz J.
Journal: Clin Immunol. 2021 Jun 10:108766. Online ahead of print.
PMID: 34091018
In this study, the researchers sought to determine if farnesol treatment would have a protection effect in a mouse model of multiple sclerosis ( called experimental autoimmune encephalomyelitis, or EAE). Their results indicate that daily oral treatment of farnesol (100 mg/kg) significantly reduced the severity of the symptoms of EAE (~80%) and delayed the onset of symptoms (by ~2 days).
And this study followed two previous reports by an independent research group that indicated interesting properties associated with farnesol:
 Title: Farnesol attenuates lipopolysaccharide-induced neurodegeneration in Swiss albino mice by regulating intrinsic apoptotic cascade.
Title: Farnesol attenuates lipopolysaccharide-induced neurodegeneration in Swiss albino mice by regulating intrinsic apoptotic cascade.
Authors: Santhanasabapathy R, Sudhandiran G.
Journal: Brain Res. 2015 Sep 16;1620:42-56.
PMID: 25935694
In this first study, the investigators investigated the effect of farnesol (100 mg/kg) on an inflammatory model of neurodegenerative (induced by lipopolysaccharide or LPS). They found that farnesol treatment had a neuroprotective effect in the mice by enhanced the antioxidant activity (as determined by increased levels of superoxide dismutase (SOD) and glutathione (GSH)).
And this study was published around the same time as the second report from this research group presenting additional farnesol data:
 Title: Farnesol quells oxidative stress, reactive gliosis and inflammation during acrylamide-induced neurotoxicity: Behavioral and biochemical evidence.
Title: Farnesol quells oxidative stress, reactive gliosis and inflammation during acrylamide-induced neurotoxicity: Behavioral and biochemical evidence.
Authors: Santhanasabapathy R, Vasudevan S, Anupriya K, Pabitha R, Sudhandiran G.
Journal: Neuroscience. 2015 Nov 12;308:212-27.
PMID: 26341906
In this study, the researchers explored the effects of farnesol treatment (again, 100 mg/kg) in a model of neurotoxicity involving acrylamide – an industrial pollutant. They found that farnesol supplementation improved gait performance and fine motor coordination. Pro-inflammatory marker (such as tumor necrosis factor alpha (TNF-α) and interleukin-1β (IL-1β)) were also reduced by farnesol treatment.
 Source: Sciencedirect
Source: Sciencedirect
These results suggest that farnesol is a molecule worthy of further investigation.
So when does the clinical trial start?
This is where I do my rain-on-the-parade routine.
While the additional preclinical data discussed above is encouraging, it will be necessary to see independent validation and replication of the PARIS results before we go leaping into large clinical trials of farnesol.
Plus, given that farnesol has never been clinically tested in humans (that I am aware of – happy to be corrected on this), it will be necessary to conduct some safety/tolerability and dosing Phase I studies in healthy volunteers to explore issues like target engagement (what do PARIS levels do in response to different doses of farnesol? And does it have any impact on PGC-1α in humans?). These studies could run in parallel with the independent validation work in order to speed things along.
So what does it all mean?
Many readers may be cynically thinking, “great, another wonder molecule to get excited about“, “this week’s break through discovery“, or perhaps “I wish I was a mouse with their symptoms rescued“. But it should be noted that this new report is the result of over a decade of research focusing in on a specific aspect of biology.
If the findings can be independently validated and farnesol is found to be safe and active in humans, this could represent a very significant result. Inhibition of PARIS is a novel approach to potentially treating Parkinson’s – another weapon in the arsenal that is being thrown at the condition (Click here to read more about the current drug pipeline for Parkinson’s).
And if farnesol doesn’t work out, then there are still lots of other options in that experimental arsenal So no need for plan B (“joining the Foreign Legions as a mute”) just yet.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The author of this post is an employee of Cure Parkinson’s, which was a funder of the farnesol project described above. Cure Parkinson’s has not asked that this post be written. The author is sharing the research here as he thought it would be of interest to the Parkinson’s community.
The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from selleckchem

this looks very promising to those living with Parkinsons
LikeLike
According to Wikipedia-
In a 1994 report released by five top cigarette companies, farnesol was listed as one of 599 additives to cigarettes.[1] It is a flavoring ingredient.
Hmm . . . .
LikeLike
But we must remember that cigarettes chose farnesol and not the other way around. 🙂
LikeLike
http://thegoodscentscompany.com/data/rw1290201.html
Used on lips therefore must meet IFRA
IFRA FLAVOR REQUIREMENTS:
Due to the possible ingestion of small amounts of fragrance ingredients from their use in products in Categories 1 and 6, materials must not only comply with IFRA Standards but must also be recognized as safe as a flavoring ingredient as defined by the IOFI Code of Practice (www.iofi.org). For more details see chapter 1 of the Guidance for the use of IFRA Standards.
LikeLiked by 1 person
Still, these experiments involved a lot more farnesol than would be absorbed from a lip gloss. 🙂
LikeLike
Hi Simon,
I am trying to get clear in my mind the relationship between farnesol and farnesyl, and have come up with a question. When creating farnesyl groups, can farnesyltransferase use any organic molecule other than farnesol?
Also, after reading your earlier blog post titled “Say it with me: Farn-e-syl-trans-fer-ase”, it seems likely that giving someone farnesol will cause an increase in the farnesylation of various other enzymes, creating unwanted side effects. As you say: ” … it will be necessary to conduct some safety/tolerability and dosing Phase I studies …”.
Warm Regards,
Jeff
LikeLiked by 1 person
Correction: I wrote “other enzymes” when I should have written “other proteins”.
LikeLike
Interesting that PARIS builds up specifically in areas of the brain affected by PD, including the nigrostriatal region. And also interesting that there is no problem of over-production (as evidenced by normal levels of the RNA used to produce PARIS).
That, as you say, suggests a disposal problem that, at least for non-hereditary PD, might *not* be due to lack of sufficient PARKIN to tag (with ubiquitin) the PARIS for disposal.
Rather, the buildup may be due to the well-known effect that aggregated alpha-synuclein has of gumming up the mechanisms of protein disposal (e.g., proteasomes)–a problem which could allow excess PARIS to accumulate even if there *were* sufficient PARKIN available to tag it for disposal.
If that turns out to be the case, then what we have with farnesol is a way to disable just *one* of the harmful side-effects (PARIS’ disabling of PGC-1α transcription) of the accumulation of excess amounts of *one* such protein (PARIS). We would still have the underlying disposal problem, and thus we would still have the negative inflammatory and mitochondrial effects of the aggregated alpha-synuclein itself (albeit, perhaps offset somewhat by the anti-inflammatory effects of the restored PGC-1α, but are those effects any more powerful than, say, just taking some powerful anti-inflammatory supplements like Longvida curcumin, baicalin/baicalein, EGCg or quercetin?).
And then we have to look, as you say, at the possible side-effects of farnesol in the fairly large amounts that would be needed to replicate these PARIS-overproducing-mouse experiments in humans.
I’m not saying that this is not an exciting development. I mean, PGC-1α seems like a very important molecule, and restoring proper levels of it could have multiple beneficial effects. But I come from a position (which I confess is ideological), that it is better to interfere with a disease process as close to its root causes as possible, rather than to offset some downstream effects of that process in ways that may also inadvertently tweak other things that one does not intend to tweak.
So if the underlying problem for idiopathic PD is aggregated alpha-synuclein, then disaggregating that protein (and then calming its knock-on auto-inflammatory effects that may have developed a life of their own) would appear to be the holy grail for slowing or stopping PD progression. And I don’t mean developing monoclonal antibodies that attempt to rally the immune system to destroy aggregated alpha-synuclein–I mean substances that have been shown (OK, maybe not in humans yet) to prevent and even reverse aggregation, such as curcumin, baicalin/baicalein, EGCg and quercetin.
Success in disaggregating AS could lead to improvements not just in the motor symptoms of PD (including better disposal of proteins like PARIS), but also in the autonomic and other effects that aggregated alpha-synuclein has outside the nigrostriatal area.
[As an aside, I’m going to relate that my partner who has PD has, after years of using the four supplements listed above, attained a significant and lasting improvement in her previously-serious autonomic problems (neurogenic orthostatic hypotension and temperature control issues). I am in the process of reconstructing her history in that regard as we did not keep very good contemporaneous records, but it is undeniable that she once needed the use of compression stockings every day to avoid faintness and fainting from nOH, and for the past year at least she has not needed to use them at all. And she also no longer has the episodes of profuse sweating that she once had, even during the recent hot weather.
Motor-wise, she still has serious problems when not properly medicated (those dopamine neurons that have been destroyed, as expected, have not grown back), but we’ve gotten pretty sloppy lately with getting her levodopa doses on time, and sometimes she goes for many hours between doses (she was diagnosed over 15 years ago, and as I write this, it’s been nearly ten hours since her last dose, because she’s been engrossed in some art that she’s creating on the PC). It is definitely better when we keep the dosing on schedule, but the reason that we have gotten so sloppy is because she now can get away with it for longer intervals with significantly less severe repercussions.
I hope you won’t mind if I drop you an email when we get the sequence of events reconstructed in case you might find that this could have significance beyond our immediate household; for us it has been a very pleasant surprise. Of course, I can’t say for sure that the anti-aggregation effects of these supplements caused the improvement, but it is our working hypothesis. Perhaps, though, it is just their anti-inflammatory effects that modulated some of the worst effects of the AS in the autonomic system. The thing is, though, this has been a long-term and gradual effect, and I would have expected any improvements due to anti-inflammatory effects to be more immediate.]
LikeLike
” … the well-known effect that aggregated alpha-synuclein has of gumming up the mechanisms of protein disposal …”
What are your thoughts on Annovis Bio’s approach to dealing with this problem? Based on something learned from familial PD (i.e. that too much a-synuclein, in itself, can cause PD), they are attempting to lessen a-synuclein-induced pathogenesis by reducing the synthesis of the a-synuclein protein.
They have a big milestone coming up within the next few months, when they are scheduled to release Phase 2 results.
https://www.newsfilecorp.com/release/92796/Annovis-Bio-Completes-Dosing-of-Parkinsons-Disease-Patients-with-ANVS401-in-Phase-2a-Clinical-Trial
Posiphen (ANVS401) mode of action:
Posiphen is an orally available small molecule drug that inhibits the translation of a-synuclein, by increasing the affinity of Iron Regulatory Protein-1 (IRP1) to the Iron-Response Element (IRE) of the SNCA gene’s messenger RNA (mRNA). This represses translation by preventing the association of the mRNA with the ribosome.
LikeLike
I was reading a paper from Dr. Schlossmacher in Ottawa https://scienmag.com/parkinsons-discovery-points-to-possible-future-treatment-approaches/ about the PARKIN protein and it sounds like two different things compared to this article. Am I missing something? The paper states, ” the parkin protein works in two ways. First, it acts like a powerful antioxidant that disarms potentially harmful oxidants in the brain, including dopamine radicals. Second, as the brain ages and dopamine radicals continue to build up, parkin sequesters these harmful molecules in a special storage site within vulnerable nerve cells, so they can continue to function normally throughout our lifespan.”
LikeLike