|
# # # # Good sleep patterns have important implications for all of us in terms of health and well being, but sleep is often disrupted for people with Parkinson’s. Research suggests that people with Parkinson’s have reduced amounts of slow wave and REM sleep, and increased periods of wakefulness. A new report has found that increasing levels of slow wave sleep could have beneficial effects in reducing the accumulation of alpha synuclein protein in the brain. In today’s post, we will discuss what sleep is, how it is affected in Parkinson’s, and what the new research indicates about slow wave sleep. # # # # |
Source: Dlanham
I am a night owl.
One that is extremely reluctant to give up each day to [the waste of precious time that is] sleep. There is always something else that can be done before going to bed. And I can often be found pottering around at 1 or 2am on a week night.
Heck, most of the SoPD posts are written in the wee small hours (hence all of the typos).

Source: Iristech
As a result of this foolish attitude, I am probably one of the many who live in a state of sleep deprivation – I am a little bit nervous about doing the spoon test:
And the true stupidity of my reluctance to adopt a healthy sleep pattern is that I fully understand that sleep is extremely important for our general level of health and well being.
In addition, I am also well aware of an accumulating pool of research that suggests sleep could be influential in the initiation and progression of neurodegenerative conditions, like Parkinson’s.
Wait, how is sleep associated with Parkinson’s?
Problems with sleep are some of the most common non-motor features of Parkinson’s, and many studies have demonstrated this.
And there has been a lot of research on this topic. A good example of this is the PRIAMO (PaRkinson And non Motor symptOms) study. It was a longitudinal 24 month observational study conducted by 59 neurology centers across Italy to establish the prevalence and incidence of the non-motor features in Parkinson’s. The study involved 1307 people with Parkinson’s.

Title: The PRIAMO study: A multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease.
Authors: Barone P, Antonini A, Colosimo C, Marconi R, Morgante L, Avarello TP, Bottacchi E, Cannas A, Ceravolo G, Ceravolo R, Cicarelli G, Gaglio RM, Giglia RM, Iemolo F, Manfredi M, Meco G, Nicoletti A, Pederzoli M, Petrone A, Pisani A, Pontieri FE, Quatrale R, Ramat S, Scala R, Volpe G, Zappulla S, Bentivoglio AR, Stocchi F, Trianni G, Dotto PD; PRIAMO study group.
Journal: Mov Disord. 2009 Aug 15;24(11):1641-9.
PMID: 19514014
The PRIAMO study found that 98.6% of the participants reported the presence of non-motor features, and one of the most common was sleep problems – occurring in 64.1% of cases.
And these sleep related issues could be further broken down into:
- REM sleep behaviour disorder (29.6% of cases)
- Insomnia (36.8%)
- Excessive day time sleepiness (21.2%)
- Restless legs (15.2%)
These results have been replicated numerous times. In fact, the incidence of sleep problems in Parkinson’s is generally stated as occurring in 40% to 90% of cases (Click here for a good review on the topic).
What happens in the brain in Parkinson’s that causes sleep problems?
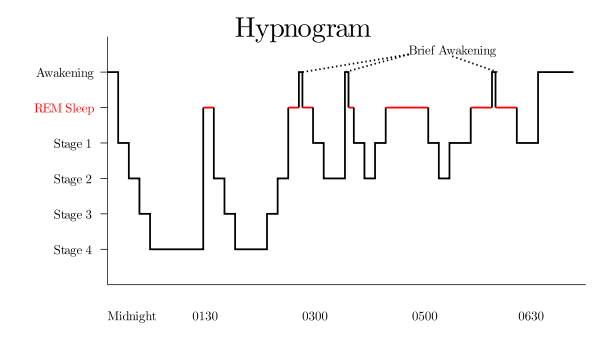
As you will be aware, sleep is an unconscious state that the body goes into when we lie down during the night. It is characterised by immobility and reduced responsiveness. There are two broad phases of sleep:
- Rapid eye movement (REM) sleep
- Non-rapid eye movement (NREM) sleep.
We pass through these phases in a wave-like cycles as we sleep:
Stages of sleep. Source: Wikipedia
In addition, there are different stages that make up the NREM sleep parts of these cycles (stage 1-4, on the left hand side of the image above), which we pass through on our way down to stage 4 and back again.
These stages exhibit different patterns of brain activity, which – when recorded – look like this:
Brain activity during stages of sleep. Source: Homesteadschools
The brain is very active during REM sleep (this is the period during which we dream), whereas during NREM sleep the brain goes through periods of synchronised neuronal activity. Stages 3 and 4 are referred to as ‘slow wave sleep‘ – this is important to our discussions below.
Stage 3 sleep contains approximately 20% of slow wave activity, while stage 4 sleep typically has more than 50% of slow wave activity (Source).
Ok, but what does sleep in Parkinson’s look like?
In many cases of Parkinson’s, the pattern of sleep can be slightly different.
Parkinson’s is associated with increased sleep latency (that is, the length of time that it takes to fall sleep – more specifically, to move from full wakefulness to stage 1 in the image above). It is also associated with increased amounts of stage I sleep and reduced levels of stage 2, 3 and REM sleep:
The % of time spent in different stages of sleep. Source: Scialert
This abnormal pattern of sleep is multifactorial in origin. It can of course relate to the motor issues of Parkinson’s, such as stiffness. Nocturia (needing to urinate) and medication effects (for example, dopamine agonists) can also disrupt sleep.
The sleep issues can also stem from the cell loss associated with Parkinson’s.
It was once believed that the brain had a specific “sleep region” (in a central region called the hypothalamus). And working in opposition to this was a separate “wakefulness region” (in the brainstem). But recent research has demonstrated that this proposal is too simplistic. Now it appears that wakefulness is not centralised but regulated by a whole network of structures in the brainstem, hypothalamus and basal forebrain (Click here to read a good review on this topic).
The circuitry of sleep. Source: Howsleepworks
Unfortunately, some of the areas of the brain stem associated with sleep are badly affected in Parkinson’s. This is particularly evident in the REM stage of sleep.
Normally during REM sleep, reduced levels of serotonin activity in the brain leads to immobility of the body, through a network of brainstem neurons. But in Parkinson’s, some of these regions suffer cell loss which results in the body losing its immobility during REM. And this is why during the very early stages of Parkinson’s some people will act out their dreams (Click here for a review on the structures of the brain involved in sleep and how they are affected by Parkinson’s).
This acting out of dreams is a feature of a condition known as REM sleep behaviour disorder.
|
# RECAP #1: Sleep is a period of immobility and non-responsiveness for the body that organisms partake in on a regular basis in order to remain healthy and functional. In humans, sleep involves different stages and patterns of brain activity. Sleep can be badly affected by Parkinson’s. The condition can disrupt different stages of sleep and patterns of brain activity. In particular, REM sleep and slow wave sleep appear to be severely affected. # |
So slow wave sleep is badly affected by Parkinson’s. Has anyone ever looked at what impact this could have on the brain in PD?
Yes. Researchers have looked at this in models of PD, which brings us to the topic of today’s post.
Very recently, this report was published:
Title: Slow-wave sleep affects synucleinopathy and regulates proteostatic processes in mouse models of Parkinson’s disease.
Authors: Morawska MM, Moreira CG, Ginde VR, Valko PO, Weiss T, Büchele F, Imbach LL, Masneuf S, Kollarik S, Prymaczok N, Gerez JA, Riek R, Baumann CR, Noain D.
Journal: Sci Transl Med. 2021 Dec 8;13(623):eabe7099.
PMID: 34878820
In this study, the researchers wanted to explore the sleep activity in mice that had been genetically engineered to produce very low levels of VMAT2.
What is VMAT2?
Vesicular monoamine transporter 2 (or VMAT2) is a protein that is critically involved with collecting chemicals like dopamine and serotonin and packaging them into small sacks (called vesicles) within neurons.
These vesicles are then transported to the tip of a neuron – where it meets another neuron, a region called the synapse – and the vesicle then releases its contents into that ‘synaptic’ space between the cells. Some of the chemicals will make contact with receptors on the surface of the next neuron, and by binding to those receptors, they will help to pass on a signal. The chemicals that do not achieve this task, however, will be taken back into the releasing neuron and be recycled or broken down and disposed of.

VMAT, vesicles and the synapse. Source: Nature
Mice with low levels of VMAT2 were viable, but they produce only half the normal levels of dopamine and serotonin (Source). And when the researchers behind this new report compared the sleep patterns of the VMAT2 mice with normal control mice they found some intriguing differences.
The VMAT2 mice exhibited increased wakefulness and reduced REM and non-REM sleep patterns.
To try and correct this, the researchers treated the mice with sodium oxybate.
What is sodium oxybate?
Sodium oxybate (also known as gamma-hydroxybutyrate) is primarily used to treat excessive daytime sleepiness and cataplexy (loss of voluntary muscle control) in narcolepsy.
 Source: Wikipedia
Source: Wikipedia
What is narcolepsy?
Narcolepsy is a rare long-term brain condition that results in a person suddenly falling asleep at inappropriate times. It is thought to be caused by a reduction in levels of a brain chemical called hypocretin (also known as orexin), which plays a key role in the regulation of sleep. This deficiency is believed to be the result from the immune system mistakenly attacking the regions of the brain that make hypocretin.
This video shows what living with the conditions is like:
The pharmacological treatment of narcolepsy involves stimulants which help to keep the brain awake during day time hours, and sodium oxybate which helps to improve the quality of sleep at nighttime – which is often very poor in narcolepsy.
Sodium oxybate increases the levels of slow wave sleep and it is taken in two doses: one at bedtime and another one around four hours later.
Got it. So what happened with the researchers gave the mice sodium oxybate?
They saw a significant increase in slow wave sleep activity during both day-time and night-time periods after long term treatment with sodium oxybate.
It should be noted that long-term treatment in older VMAT2 mice did not have an effect on motor ability.
Interesting.
Indeed, but what the researchers did next was where this particular story gets really interesting.
What did they do next?
Well, the scientists knew from previous research on Alzheimer’s that pharmacological sleep enhancement can reduce the accumulation of beta amyloid protein in the brains of genetically engineered mice.
What is aggregated beta amyloid protein?
Beta amyloid has long been considered to be the villain in Alzheimer’s.
It is a piece of a larger protein that sits in the outer wall of nerve cells where it has various functions. In certain circumstances, specific enzymes can cut it off and it floats away.

The releasing of Beta-Amyloid. Source: Wikimedia
Beta amyloid is a very “sticky” protein and it has been believed that free floating beta amyloid proteins begin sticking together (or aggregating), gradually building up into the large amyloid plaques. And these large plaques were considered to be involved in the neurodegenerative procession of Alzheimer’s.
This was one of the first studies to report that sleep enhancement can reduce the accumulation of aggregated beta amyloid protein:
 Title: Amyloid-beta dynamics are regulated by orexin and the sleep-wake cycle.
Title: Amyloid-beta dynamics are regulated by orexin and the sleep-wake cycle.
Authors: Kang JE, Lim MM, Bateman RJ, Lee JJ, Smyth LP, Cirrito JR, Fujiki N, Nishino S, Holtzman DM.
Journal: Science. 2009 Nov 13;326(5955):1005-7.
PMID: 19779148 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that chronic sleep restriction significantly increased the levels of beta amyloid protein aggregation in the brains of genetically engineered Alzheimer’s mice, while sleep enhancement (using a dual orexin receptor antagonist) decreased levels of beta amyloid in the brain.
This result suggested that good sleep patterns are required for helping to clear accumulating protein from inside the brain.
Naturally, the researchers of the new report that we are reviewing today started wondering if sleep enhancement – using sodium oxybate – might reduce levels of Parkinson’s-associated alpha synuclein protein aggregation in the brain.
We talk about alpha synuclein a lot on this website (click here to read a recent SoPD post to learn more about its association with Parkinson’s). If you are unfamiliar with it, just understand that in many cases of PD, alpha synuclein protein started to accumulate and aggregate together inside of neurons.
How did the researchers assess the effect of sleep enhancement on alpha synuclein?
The investigators genetically engineered a mouse that produced a lot of a version of alpha synuclein that carries a particular mutation (A53T) that makes it prone to aggregation. They then treated some of these mice with sodium oxybate (and the rest with a placebo solution) and evaluated their levels of alpha synuclein aggregation and also their sleep patterns.
 Source: Pinterest
Source: Pinterest
The researchers found that alpha synuclein accumulation in these genetically engineered mice was associated with disrupted sleep patterns, and that long-term treatment with sodium oxybate not only improved those sleep patterns, but also reduced the levels of alpha synuclein in their brains.
One interesting note here: The researchers found a gender difference with this result – the effect of sodium oxybate was stronger in the male mice than the female mice. In the male mice, the reduction in alpha synuclein load was approximately 70%, compared to only 30% in the female mice.
In the discussion of their report the researchers explore this finding, making note of the “increasing numbers of drugs presenting sexually dimorphic responses” and they point towards the Women’s brain project website.
In their extensive discussion, the researchers also explore the potential mechanism of action for the results, pointing towards improved homeostasis of protein handling and also better functioning of the glymphatic system (which we have discussed in a previous SoPD post – click here to read that post).
The investigators concluded their study by stating that the “promising results of this study provide a unique framework that encourages further exploration of the role of slow wave sleep in Parkinson’s pathology”, and I’m inclined to agree with that.
|
# # RECAP #2: Researchers are exploring different ways of improving levels of slow wave sleep. Pharmacological treatments, such as sodium oxybate, have been shown to increase slow wave activity in sleep. Scientists have also noted that increasing slow wave sleep in model of neurodegenerative conditions can help with the reduction/clearance of aggregated proteins – which are believed to be influential in the progressive nature of these conditions. # # |
Has sodium oxybate ever been explored in other neurodegenerative conditions associated with protein aggregation?
Yes it has.
In 2014, researchers reported that gamma-hydroxybutyrate (aka sodium oxybate) can protect cells from cell death in cellular models of Alzheimer’s:
 Title: Gamma-hydroxybutyrate, acting through an anti-apoptotic mechanism, protects native and amyloid-precursor-protein-transfected neuroblastoma cells against oxidative stress-induced death.
Title: Gamma-hydroxybutyrate, acting through an anti-apoptotic mechanism, protects native and amyloid-precursor-protein-transfected neuroblastoma cells against oxidative stress-induced death.
Authors: Wendt G, Kemmel V, Patte-Mensah C, Uring-Lambert B, Eckert A, Schmitt MJ, Mensah-Nyagan AG.
Journal: Neuroscience. 2014 Mar 28;263:203-15.
PMID: 24456637
In this study, the researchers treated both normal cells and cells that produce high levels of beta amyloid with a chemical (hydrogen peroxide) that causes great stress on the cells. They then treated the cells with sodium oxybate and found that the treatment could – efficiently and in a dose-dependent manor – rescue the cells.
One year later, the same research group published a second report which tested sodium oxybate in a mouse model of Alzheimer’s:
 Title: gamma-Hydroxybutyrate (Xyrem) ameliorates clinical symptoms and neuropathology in a mouse model of Alzheimer’s disease.
Title: gamma-Hydroxybutyrate (Xyrem) ameliorates clinical symptoms and neuropathology in a mouse model of Alzheimer’s disease.
Authors: Klein C, Mathis C, Leva G, Patte-Mensah C, Cassel JC, Maitre M, Mensah-Nyagan AG.
Journal: Neurobiol Aging. 2015 Feb;36(2):832-44.
PMID: 25457559
In this study, the researchers treated genetically engineered mice (which were generated to produce high levels of a mutant form of beta amyloid) with sodium oxybate and they reported reduced levels of aggregated beta amyloid in the brains, less associated cell death, and a reduction in the cognitive deficits typically seen in these mice. Importantly, this effect was achieved with clinically relevant doses of the drug.
Has sodium oxybate been tested in people with Parkinson’s?
Yes it has.
Sodium oxybate is clinically used in the treatment of daytime sleepiness, which can be a problem in Parkinson’s due to dysfunctional sleep.
This study was reported in 2018:
 Title: Sodium Oxybate for Excessive Daytime Sleepiness and Sleep Disturbance in Parkinson Disease: A Randomized Clinical Trial.
Title: Sodium Oxybate for Excessive Daytime Sleepiness and Sleep Disturbance in Parkinson Disease: A Randomized Clinical Trial.
Authors: Büchele F, Hackius M, Schreglmann SR, Omlor W, Werth E, Maric A, Imbach LL, Hägele-Link S, Waldvogel D, Baumann CR.
Journal: JAMA Neurol. 2018 Jan 1;75(1):114-118.
PMID: 29114733 (This report is OPEN ACCESS if you would like to read it)
This was a randomised, double-blind, placebo-controlled, crossover study that involved 11 individuals with Parkinson’s who also suffered from excessive daytime sleepiness. The participants took sodium oxybate (or placebo) at bedtime and again 3-4 hours later. The treatment was administered for 6 weeks and then taken away for another 2-4 week washout period (Click here to read more about the details of this study).
The results indicate that the sodium oxybate treatment “exhibited an electrophysiologically defined positive treatment response” in 8 (67%) of the participants. It also significantly enhanced subjective sleep quality and objectively improved the amount of slow-wave sleep.
Overall, sodium oxybate was well tolerated by the participants in the study, but it was found to induce obstructive sleep apnea (where breathing is momentarily cut off) in 2 participants. Despite this, the researchers concluded that their results warrant larger follow-up trials with longer treatment durations.
And it should be noted here that Dr Christian Baumann who was the senior researcher on this clinical trial is also on the list of authors of the preclinical research report that has been reviewed in today’s post.
It will be interesting to learn what he has planned for 2022.
But is there any other evidence that sodium oxybate might lower alpha synuclein aggregation in humans?
This clinical study was published last year:
 Title: Sleep Deprivation Affects Tau Phosphorylation in Human Cerebrospinal Fluid.
Title: Sleep Deprivation Affects Tau Phosphorylation in Human Cerebrospinal Fluid.
Authors: Barthélemy NR, Liu H, Lu W, Kotzbauer PT, Bateman RJ, Lucey BP.
Journal: Ann Neurol. 2020 May;87(5):700-709.
PMID: 32057125 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in the effect that sleep deprivation has on tau protein in human cerebrospinal fluid – this is the fluid that surrounds the brain.
What is Tau?
In the image below you will see a fluorescent photo of two cells that have been labelled with two different dyes: one that highlights the nucleus (in blue) and another that stains the microtubules (in green). Everything else in the cell (membrane and all) is left invisible by this labelling process:

Tau protein (green) inside two cells. Source: The human protein atlas
Pretty huh?
Pretty. But what is Tau?
Microtubules are the highways and byways inside a cell.
They are structures inside of cells that are involved with both cellular and intracellular transportation. That is to say, these skeleton-like structures help cells move from place to place within the body (‘cellular transportation’), but they also aid in the movement of proteins from place to to within the cell (‘intracellular transportation’). On top of this, microtubules also play an important function in cell division.
The stability and maintenance of these microtubles relies on proteins like Tau.
Tau is a microtubule-associated protein – meaning it directly interacts with microtubules. It functions by stabilising these highways and byways inside a cell. Basically, this protein holds together the roads which allows vesicles (the small bags of material) to be moved around the cell (by transporter proteins like kinesin).
 TAU: a stabilising presence. Source: Hindawi
TAU: a stabilising presence. Source: Hindawi
In neurodegenerative conditions like Alzheimer’s, progressive supranuclear palsy, and corticobasal syndrome, there is a build up of Tau protein, which accumulates and clusters (or ‘aggregates’) in a similar fashion to alpha synuclein.
So in this small clinical study the researchers wanted to look at the effect that sleep deprivation has on Tau in cerebrospinal fluid. They recruited 8 healthy volunteers (aged 30–60 years), who were randomised to “sleep deprivation for 36 hours”, “increased sleep with sodium oxybate”, or “normal sleep” (control).
They found that sleep deprivation did affect Tau levels in cerebrospinal fluid.
While this result is interesting, we are more interested in the protein that the researchers used as a control measure: alpha synuclein.
The investigators found that overnight cerebrospinal fluid levels of alpha synuclein concentrations increased during sleep deprivation by approximately 80% (compared to controls). And when the investigators administered sodium oxybate to some of the participants and they observed that this treatment kept alpha synuclein levels at the same as those of the control group:
 Source: PMC
Source: PMC
In addition, there is a very old report (1973) indicating that sodium oxybate can improve PD symptoms (Click here to read the abstract), but I do not have access to this report so I can’t say much more about it.
Where can I get some of this sodium oxybate stuff?
First thing to remember is that all of this is preliminary experimental data. It really needs to be carefully clinically tested in some large placebo controlled trials.
Second, sodium oxybate may be approved by the U.S. FDA for the treatment of narcolepsy, but it carries a strict risk evaluation and mitigation strategy (REMS) program mandated by the FDA – this means that careful monitoring of the medication is required as there is high potential for serious adverse effects.
In addition, the US label for sodium oxybate carries a black box warning. This is due to the agent being a central nervous system depressant, with the potential to cause seizures, coma, or death (particularly if used in combination with other depressants, such as alcohol).
Thus, some caution must be taken in terms of future exploration of this research.
And then of course there is the price tag on this drug…
Sodium oxybate originally developed for the treatment of narcolepsy by a biotech company called Orphan Medical. It was approved by the FDA in 2002 (and given the trade name Xyrem). Orphan Medical was then acquired by the biotech company Jazz Pharmaceuticals in 2005 for $122.6 million.
Jazz Pharmaceuticals very quickly started increasing the price of Xyrem (a 1-milliliter dose of Xyrem cost $2.04 in 2007, but that price rose to $19.40 by 2014, representing an >800% increase in price – source). This situation is resolving itself, however, as the key chemical composition patents covering Xyrem have already expired (back in 2019), and remaining patents protecting its method of delivery are set to expire between 2023 and 2025. This opens up the market for generic companies to provide cheaper alternatives to Xyrem.
All of this to say, it is an expensive drug that needs to be approached with caution. Please do not do anything without consulting your physician.
So what is going to happen next?
Well, UCB Pharmaceutical is acknowledged in the new preclinical report as a sponsor of the study, so I will be curious to see what they do with this data.
 In addition, there are other biotech companies in this space that may also take an interest in this new result. For example, XWPharma recently completed Phase I testing of their oxybate-based agent XW10172 (Click here to read more about this).
In addition, there are other biotech companies in this space that may also take an interest in this new result. For example, XWPharma recently completed Phase I testing of their oxybate-based agent XW10172 (Click here to read more about this).
In their study, healthy volunteers were treated with XW10172 – an immediate-release oral formulation that is “rapidly absorbed and metabolised to release oxybate at concentrations that matched equal molar doses of sodium oxybate, currently approved for treatment of narcolepsy“. And XW10172 maintains those desired concentrations over 6-7 hours following oral ingestion.
The interesting aspect of XWPharma’s approach is that they are focused on Parkinson’s as their primary indication for XW10172, and they plan “to initiate a Phase 2 study in the coming months in patients with Parkinson’s disease suffering from sleep disorders“.
The current results could open up a new avenue of investigation for some of these biotech companies developing Xyrem generics or sodium oxybate-like drugs.
So what does it all mean?
At many times in the writing of this post, I thought I seriously need to get more sleep (it is currently 2am). And I have to admit that I really like sleep. So much so that I have deep sympathy for those in the Parkinson’s community whose sleep patterns are affected by the condition. There is nothing like a good night sleep to help clear the head (that almost reads like a pun given the contents of today’s post).
I was really intrigued by the report we have reviewed today. It points towards a mechanism by which the brain may be cleansing itself of rubbish during our deep sleep state. And as we age, perhaps this process is disrupted, leaving us vulnerable to a build up of proteins like alpha synuclein (which may cause a feed-forward cycle perhaps). Thus, better sleep may be a means of slowing progression in neurodegenerative conditions and improving overall brain function.
All of that said, I think I’m going to go to bed now…
Good night!
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, some of the biotech companies that are mentioned in this post are publicly traded company. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with the company or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from Wikipedia






Good post, Thanks
Simon – any comment on Gabapentin? Gabapentin also increases slow wave sleep. A known cheap drug but not without a few issues I hear. Low doses help me sleep.
“Conclusions: Gabapentin enhances slow-wave sleep in patients with primary insomnia. It also improves sleep quality by elevating sleep efficiency and decreasing spontaneous arousal. The results suggest that gabapentin may be beneficial in the treatment of primary insomnia.”
https://pubmed.ncbi.nlm.nih.gov/20124884/
LikeLike
Hi DKDC,
Thanks for the comment – that’s really interesting! I was not aware of this. Certainly worth further exploration. I will look into it.
Kind regards,
Simon
LikeLike
I should note that I only know this because I was prescribed gabapentin for back and hip arthritis affecting my nerves there. It seemed to help me more with sleep.
Don’t take my post as a recommendation of what one should take for PD – I have no medical training. And my PD has progressed at about an average rate, I think.
LikeLike
Thanks DKDC – noted (always wise to have disclaimers attached to all posts and comments) 🙂
LikeLike
A few more links in case any of them are helpful. I should add that my MDS neurologist told me “gabapentin is not something used to treat Parkinson’s.” There must be some downside to using it, or other medications do what it does – only better.
Gabapentin leads to better uprds scoreshttps://pubmed.ncbi.nlm.nih.gov/15190235/
Gabapentin not PROVEN good for PD pain, or for all pd pain? There are various kinds of pain in PDhttps://www.karger.com/Article/Fulltext/494686
Use in PD mentioned by the Parkinson’s Foundationhttps://www.parkinson.org/sites/default/files/attachments/Medications-Treatment-Guide-to-Parkinsons-Disease.pdf
A Moderate Drug Interaction exists between gabapentin and Sinemet … This may make the symptoms of Parkinson’s disease worse.https://www.drugs.com/drug-interactions/gabapentin-with-sinemet-1147-0-515-250.html
Gabapentin helps with tremors?https://forum.parkinsons.org.uk/t/gabapentin/10007
LikeLike
Interesting, as ever. Thank you.
LikeLike
Thanks Ben – glad you liked it.
Kind regards,
Simon
LikeLike
Simon, Is gammahydroxybutyrate an agonist for GPR109A ? Butyrate and niacin are, and niacin has recently been shown to reverse some symptoms of PD.
LikeLiked by 1 person
Hi Peter,
Thanks for the interesting comment.
Beta-hydroxybutyrate is an endogenous ligand for GPR109A, but I am afraid I’m not sure about gamma.
And regarding niacin, are you referring to the Chong study published in June (https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC8245760/)?
Kind regards,
Simon
LikeLike
Simon, I read this with great interest,,particualrly the section on other atypical parkinsonisms. It spounds very promising. Thakns for sstaying up late,to post it [but you should take care of yourself.]. Keith
LikeLike
Thanks Keith – I’m glad you found it interesting.
I’m off to bed right now!
Kind regards,
Simon
LikeLike
Excellent stuff. Thank you Simon. Your blog seems to be the best place for finding out about PD progress, at least without a medical library subscription service, and then i’d need years of training to sift through to find the useful stuff.
Gratefully yours,
Tom R
LikeLiked by 1 person