|
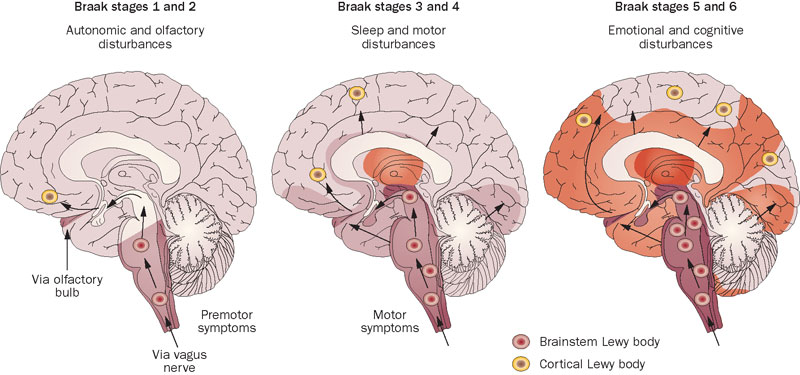
# # # # The cause of the cell loss and pathology associated with Parkinson’s is still unknown. While the later stages of the condition have been well investigated based on various pathological marker (ie Braak staging), the early manifestations of the condition are still a mystery. Cells of the immune system are early responders to any signs of trouble in our bodies, and recently researchers have been looking at a specific class of immune cells (called T cells) in postmortem sections of brains from people who passed away with Parkinson’s. Curiously, in their analysis the researchers found that the bulk of activity of T cells occurs before any cell loss or pathology appears. In today’s post, we will discuss what T cells are, review the new research, and explore what this could mean for potential therapies for Parkinson’s. # # # # |
 Your hematopoietic system. Source: Wikipedia
Your hematopoietic system. Source: Wikipedia
The process of hematopoiesis (or blood formation) is absolutely fascinating.
Seriously.
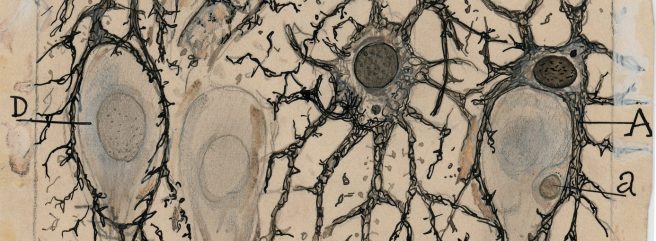
You start off with a single, multi-potential hematopoietic stem cell. This is called a hemocytoblast (it’s the big cell in middle of the image below):
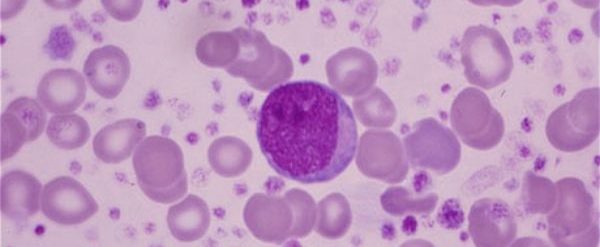 A hemocytoblast. Source: Pinterest
A hemocytoblast. Source: Pinterest
Given enough time, this single cell will give rise to an entire blood system, made up of many of different types of cells with very specific functions that are required for us to live normal lives.
It is a remarkable achievement of biology.
Understand that at any moment in time your blood system will contain 20-30 trillion cells (in the average human body). And as the image near the top of the post suggests, there are quite a few branches of potential cell types that these blood stem cells can generate.
Very interesting, but what does this have to do with Parkinson’s?