|
The clustering (or aggregation) of misfolded proteins is a key feature of many neurodegenerative conditions. These aggregating proteins are collectively referred to as ‘amyloid’ proteins, and the way that they have misfolded allows many copies of these proteins to stick together. Amyloid proteins are associated with more than 50 medical conditions (from Alzheimer’s, ALS, Huntinton’s and Parkinson’s through to rheumatoid arthritis and diabetes). In addition to being public enemy no. 1 for their respective conditions, amyloid proteins also share another curious feature: They glow when exposed to specific wavelengths of light (like near-infrared). In today’s post, we will look at what we mean by ‘amyloid proteins’, what this new research found, and how this property could be extremely useful in the tracking of Parkinson’s over time.
|
 Source: Yoursalesplaybook
Source: Yoursalesplaybook
If you have recently sent me an email, you may not have had a response. I apologise profusely for this, but I have gradually become inundated with questions and requests, and have had a hard time keeping up (in addition: family and day job take priority).
I do get some wonderfully titled emails though, which immediately grab the attention.
For example, the other day I recieved an email entitled:
“So, will my head glow in a disco?”
A brief glance at the contents confirmed suspicions that the sender was referring to this new research report:
 Title: Ultraviolet–visible–near-infrared optical properties of amyloid fibrils shed light on amyloidogenesis
Title: Ultraviolet–visible–near-infrared optical properties of amyloid fibrils shed light on amyloidogenesis
Authors: Pansieri J, Josserand V, Lee S-J, Rongier A, Imbert D, Sallanon MM, Kövari E, Dane TG, Vendrely C, Chaix-Pluchery O, Guidetti M, Vollaire J, Fertin A, Usson Y, Rannou P, Coll J-L, Marquette C, & Forge V
Journal: Nature Photonics, published 13th May 2019
PMID: N/A
Previously researchers have described an intrinsic ultraviolet–visible optical property to amyloid proteins.
What does that mean?
Which part?
All of it. What is “an intrinsic ultraviolet–visible optical property”?
Ultraviolet (or UV) light is a band of the electromagnetic spectrum (see image below) with wavelength from 10 nm to 400 nm, shorter than that of visible light but longer than X-rays.
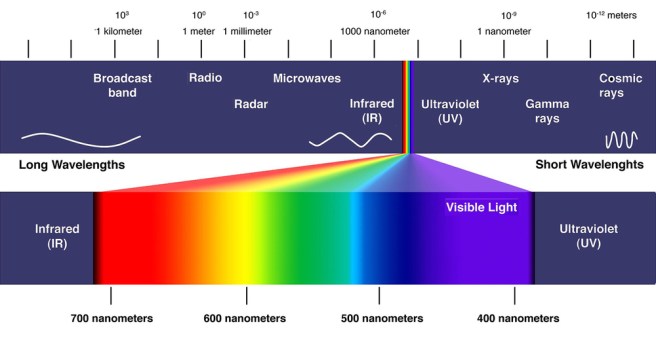 Source: Dailyoptician
Source: Dailyoptician
This video helps to explain what UV light is:
(An interesting party trick with UV light: Tonic water contains the chemical quinine, which absorbs UV light and then re-emits it as visible light. This means that tonic water appears to glow in UV light when other material remain dark)
But what is meant by “Ultraviolet–visible”?
Ultraviolet–visible refers to part of the ultraviolet and the full visible spectral regions where they meet/overlap.
Basically the border between UV and visible light.
Thus, an “intrinsic ultraviolet–visible optical property” refers to the ability of a material to emit a detectable signal when exposed to light from that borderland between UV and visible light.
And researchers have previously reported that amyloid proteins have an “intrinsic ultraviolet–visible optical property” – they glow in UV-visible light.
Interesting. But what are amyloid proteins.
An amyloid protein is one which folds into a particular shape which allows many copies of that protein to stick together.
When a protein is first formed, it must be folded into their correct functioning shape in order for it to do its assigned task.
But if a protein remains partially or completely unfolded, then it can acquire a shape which may encourage them to stick to other similarly shaped proteins, resulting in the production of amyloid assemblies. These assemblies are called fibrils.
 Source: Quora
Source: Quora
Amyloid proteins are associated with a lot of medical conditions (Click here for a partial list).
Are they associated with Parkinson’s?
Yes, the Parkinson’s associated protein alpha synuclein can become an amyloid protein.
What is alpha synuclein?
Alpha synuclein sounds like a distant galaxy, but it is one of the most common proteins in our brains. It makes up about 1% of all the protein in a neuron. When alpha synuclein protein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure.
When it is first produced, alpha synuclein will look something like this:

Alpha synuclein. Source: Wikipedia
In this form, alpha synuclein is considered a monomer – which is a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). It is believed that alpha synuclein has certain functions as a monomer, but may also have specific tasks as an oligomer.
In Parkinson’s, alpha synuclein will also misfold and aggregate together to form amyloid fibrils.

Microscopic images of Monomers, oligomers and fibrils. Source: Brain
And it is believed that the oligomer and fibril forms of alpha synuclein protein that aggregate together, and then go on to form what we call Lewy bodies.
 Parkinson’s associated alpha synuclein. Source: Nature
Parkinson’s associated alpha synuclein. Source: Nature
A Lewy body is referred to as a cellular inclusion, as they are almost always found inside the cell body. They are a characterisitic feature of the Parkinsonian brain.
 A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
I see. So amyloid fibrils of alpha synuclein glow in UV-visible light?
Apparently they do.
But this new study found something really interesting.
You see, UV light does not penetrate tissue very well. Thus, the potential utility of the intrinsic ultraviolet–visible optical property has been limited.
But in this new study we are reviewing in today’s post, the investigators reported that amyloid fibrils illuminated with near-infrared radiation also emit a dim, near-infrared signal.
What is near-infrared radiation?
Infrared radiation is on the opposite side of visible light to UV, and it has longer wavelengths than those of visible light.
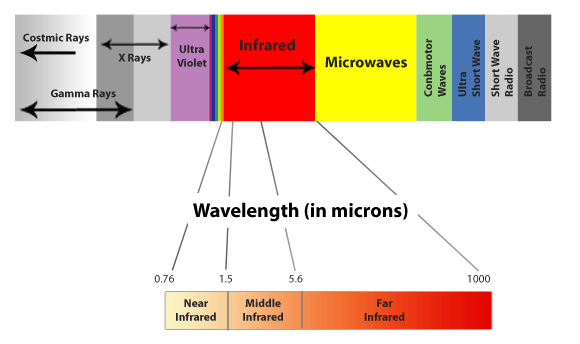 Source: Wjgnet
Source: Wjgnet
Near-infrared is the border land between visible light and infrared.
And the reason this finding is important is because near-infrared falls within what is refered to as the “diagnostic window” – unlike UV light, near-infrared radiation can penetrate relatively deeply into tissue (1-2cm), which increases its potential utility.
When the researchers added a chemical to break up the amyloid proteins, the emitted signal vanished in the presence of near-infrared light.
The researchers characterised this effect on 13 different proteins which are capable of forming amyloid fibres – including alpha synuclein – but the bulk of the presented research focused on the Alzheimer’s-associated amyloid protein beta amyloid.
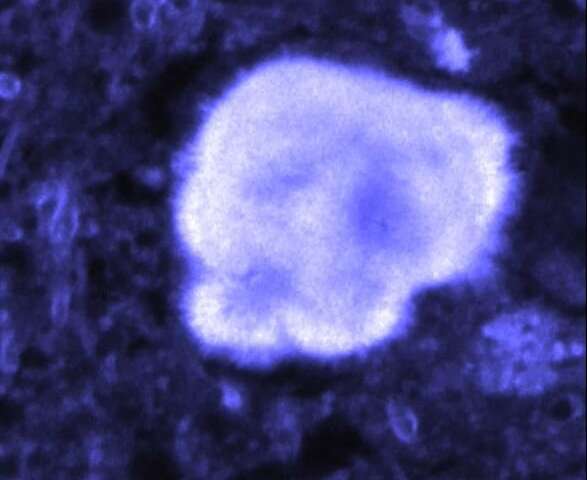 Beta amyloid protein under near-infrared light. Source: Phys
Beta amyloid protein under near-infrared light. Source: Phys
To explore this interesting property further, the researchers next exposed sections of brains from an aged mouse model of Alzheimer’s (18 month old mice producing high levels of human beta amyloid protein) to near-infrared light.
They found that amyloid protein deposits in the tissue could be easily detected – critically without the need for staining the sections with any special fluorescent markers. No obvious signal could be seen in the absence of infrared light. And no signal was observered on brain sections from normal, aged control mice.
One particularly interesting feature of the emission (from a research standpoint) is that it was so intense. Intense enough to allow the researchers to analyse the structure of the amyloid deposits within sections of brain from a person who had passed away with Alzheimer’s.
But the acid test, was whether the researchers could observe the effect on live mice.
 Source: Pinterest
Source: Pinterest
The brains of live animals were imaged noninvasively and without the use of any injected or applied contrast agent. The researchers reported that the signal being emitted by the heads of aged Alzheimer’s mice (the same model mentioned above) was 6 times more intense compared with that in the control mice.
The investigators admit that there is still a long way to go with this technology – at present it is just an interesting observation. There are some major technical hurdles, and some researchers are not sure how this could be useful in humans short of cutting holes in the skull.
That said, the investigators behind the study have been reported as saying “It’s not for tomorrow because it will necessitate further technological advances”, but then adding: “we have plans” (Source).
It will be interesting to see what those plans are.
Interesting indeed. Is this the first time anyone has noticed this ‘glowing’ property of amyloid proteins?
Actually, No.
As far back as 2003, researchers had noted this curious feature:
 Title: Live tissue intrinsic emission microscopy using multiphoton-excited native fluorescence and second harmonic generation
Title: Live tissue intrinsic emission microscopy using multiphoton-excited native fluorescence and second harmonic generation
Authors: Zipfel, W. R. et al.
Journal: Proc. Natl Acad. Sci. USA (2003) 100, 7075–7080.
PMID: 12756303 (This report is OPEN ACCESS if you would like to read it)
And it has been reported by numerous follow up reports (Click here, here, here, here, here, and here to read more examples). So this is certainly not the first time this property of amyloid proteins has been reported, but it is the best characterisation of the effect to date.
So will my head glow in a disco?
I’m just taking an uneducated guess here, but most likely not.
Sorry to disappoint anyone hoping for the opposite answer.
The amount of aggregated alpha synuclein protein in the Parkinsonian brain is tiny compared to the total amount of alpha synuclein protein in the brain. And even then, the total amount of alpha synuclein in the brain is less than 1% of the total amount of protein in the brain.
So no, the head of a person with Parkinson’s will not glow in a disco.
So what does it all mean?
The discovery of an interesting property of amyloid proteins could open new doors with regards to imaging and monitoring numerous neurodegenerative conditions. Better methods of visualising these proteins – which do not require the administration of any specific reagents – could be a major step forward not only for research, but also in early detection of disease and monitoring of conditions over time.
The future of this research is now an engineering matter. Devising clever machines that can emit and detect tiny signals will be challenging, but hopefully not impossible.
We will wait and see how this research progresses.
The banner for today’s post was sourced from marcodevisser

Shelter from the mail blizzard: You do so much to help those of us in the PD community–could we possibly help you? What possibility might there be in having a site where all the questions with which you are bombarded are posted, and those who feel qualified by experience or education could volunteer replies? This would be something like the site “Quora”, where participants volunteer to answer in fields where they have some experience or expertise. It would take some technically savvy folks to create it, and some scientific folks to moderate it for crazy or off-topic posts, the first two obvious problems (of many!). If you think it would be helpful, you might just post a “Who’s interested?” note in your reply section. Thanks again–to you and your family, both.
LikeLike