|
# # # # The cause of the cell loss and pathology associated with Parkinson’s is still unknown. While the later stages of the condition have been well investigated based on various pathological marker (ie Braak staging), the early manifestations of the condition are still a mystery. Cells of the immune system are early responders to any signs of trouble in our bodies, and recently researchers have been looking at a specific class of immune cells (called T cells) in postmortem sections of brains from people who passed away with Parkinson’s. Curiously, in their analysis the researchers found that the bulk of activity of T cells occurs before any cell loss or pathology appears. In today’s post, we will discuss what T cells are, review the new research, and explore what this could mean for potential therapies for Parkinson’s. # # # # |
 Your hematopoietic system. Source: Wikipedia
Your hematopoietic system. Source: Wikipedia
The process of hematopoiesis (or blood formation) is absolutely fascinating.
Seriously.
You start off with a single, multi-potential hematopoietic stem cell. This is called a hemocytoblast (it’s the big cell in middle of the image below):
 A hemocytoblast. Source: Pinterest
A hemocytoblast. Source: Pinterest
Given enough time, this single cell will give rise to an entire blood system, made up of many of different types of cells with very specific functions that are required for us to live normal lives.
It is a remarkable achievement of biology.
Understand that at any moment in time your blood system will contain 20-30 trillion cells (in the average human body). And as the image near the top of the post suggests, there are quite a few branches of potential cell types that these blood stem cells can generate.
Very interesting, but what does this have to do with Parkinson’s?
A lot.
And what we need to do today to make our way down one of those branches to a particular type of blood cell, which we are going to be focusing on today.
Blood accounts for approximately 7% of your body weight, and the average adult has a blood volume of roughly 5 litres (11 US pints).
There are three chief components of blood:
- Red blood cells
- White blood cells
- Plasma (92% water and 8% other stuff – think blood clotting proteins, waste, nutrients, etc)
By volume, the red blood cells constitute about 45% of whole blood, the plasma about 54.3%, and white cells about 0.7%.
 Source: KhanAcademy
Source: KhanAcademy
When it comes to the cellular parts of blood, one cubic microliter of blood contains:
- Approximately 5 million erythrocytes, which are ‘red blood cells’ that distribute oxygen around the body. These are small, bi-concave shaped cells that lack any nucleus, but this extra space allows for more hemoglobin – the critical protein involved in transportation of oxygen.
 Source: KhanAcademy
Source: KhanAcademy
- 200,000–500,000 thrombocytes, (also known as platelets) which take part in blood clotting. Thrombocytes are are not actually cells, but rather cell fragments. They are produced by large cells called megakaryocytes which each release 2-3000 platelets in their life time. Platelets are small (2-4 micrometers), lens-shaped structures that float around in the blood system waiting to help plug a rupture.
 Source: KhanAcademy
Source: KhanAcademy
- 5,000–10,000 leukocytes, which are the white blood cells that form the body’s immune system – chewing up and removing old or rogue cells or waste, as well as attacking any foreign or infectious agents.
For the rest of this post, we are going to focus on a particular type of leukocyte.
What are leukocytes?
Leucocytes (or ‘white blood cells’) are the part of the blood system involved in protecting the body against both infectious disease and foreign invaders
They can be divided into two different groups based on what they look like under a microscope:
- granulocytes – which have tiny granules inside them
- agranulocytes – which do not have tiny granules inside them (simple enough)
This division can broken down further into five major subtypes:
 Source: KhanAcademy
Source: KhanAcademy
And while it would bring me endless pleasure to discuss the roles of each of these sub-types, I will spare you that torturous fate and dedicate the rest of this post to the cell on the far right (of the image, not political persuasion).
That cell is a lymphocyte.
What are lymphocytes? And when does any of this actually get back to Parkinson’s?
When your body is attacked by a pathogen (a disease causing agent) like the common cold virus, it will elicit what is called an immune reaction. The presence of the virus inside the body will be detected by cells in the immune system and given that the virus will be clearly determined to be ‘not self’ (or not part of your body), an immune response will be initiated.
The cells that carry out the immune response are the lymphocytes.

That big cell in the middle is a lymphocyte. Source: ASH
There are basically three types of lymphocytes:
- B cells
- Natural killer cells (cool name right?)
- T cells
B cells, Natural killer cells and T cells are highly specialised blood cells that defend our body when things go wrong (which they inevitably do, it’s just the way life is!).
B cells are bone marrow-derived cells which produce Y-shaped proteins called antibodies that are used to label the invading pathogens (such as viruses), and alert other immune cells that these unwanted pathogens need to be disposed of. By sticking to pathgens, antbodies can also block them from doing their function.

Antibodies. Source: Astrazeneca
Utterly useless pub quiz fact: The ‘B’ doesn’t come from ‘bone’ or ‘blood’, but rather it comes from the name of the place these cells were discovered: the Bursa of Fabricus. The Bursa is an organ only found in birds.
 Source: Askabiologist
Source: Askabiologist
B cells do not kill pathogens, they just spend their short life producing antibodies which trap and neutralise them.
The killing of pathogens is left to the natural killer cells and T cells.
Natural killer cells were first noticed because of their ability to kill cancer tumour cells, but what made them particularly interesting was that their approach to killing: it occurred without any priming or prior activation. Natural killer cells are truly the sociopaths of the immune system – if they don’t like the looks of something, they just kill it (shoot first, ask questions later).
In the image below, you can see a large purple/blue cell (this is a natural killer cell) making contact with lots of other cells.
 A NK cell. Source: Dana-Faber
A NK cell. Source: Dana-Faber
Thymus-derived T cells kill cells, on the other hand, are ‘primed’ (by antigen presenting cells) to target very specific pathogens. They are the precision, tactical weapons of the immune system.
T cells can be identified by the presence of a T-receptor on their surface. This T-receptor identifies antigens (a molecule capable of inducing an immune response) and binds to them, which activates the T cell. Each T cell targets a specific antigen.
In a nutshell: Natural killer cells kill any thing that doesn’t look right to them, while T cells kill a specific, unique target.
And the rest of this post is going to focus on T cells.
|
# RECAP #1: Blood cells have very specific functions. Red blood cells carry oxygen to all of the tissues of the body, while white blood cells are involved in the immune response to pathogens and damage. T cells are a type of white blood cell, that each target very specific pathogens (or disease causing agents). # |
So now we get to the stuff about Parkinson’s?
Yes.
This report was recently published:
 Title: CD8 T cell nigral infiltration precedes synucleinopathy in early stages of Parkinson’s disease.
Title: CD8 T cell nigral infiltration precedes synucleinopathy in early stages of Parkinson’s disease.
Authors: Galiano-Landeira J, Torra A, Vila M, Bové J.
Journal: Brain. 2020 Oct 29:awaa269. Online ahead of print.
PMID: 33118032
In this study, the researchers wanted to look at T cells in postmortem sections of brain from people who passed away with Parkinson’s. They focused their attention on a specific region of the brain, called the substantia nigra.
What is the substantia nigra?
By the time a person is presenting the motor features characteristic of Parkinson’s (slowness of movement, rigidity, and a resting tremor) and they are being referred to a neurologist for evaluation, they will have lost approximately 50% of the dopamine producing neurons in an area of the brain called the midbrain.
 The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinsonian brain (right). Source:Memorangapp
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinsonian brain (right). Source:Memorangapp
The dopamine neurons reside in a region of the midbrain, called the substantia nigra. The substantia nigra is normally visible by eye because the dopamine neurons produce a dark pigment (called neuromelanin). As the image above illustrates, people with Parkinson’s have less of this pigmentation as a result of the lost dopamine neurons.
The investigators labelled T cells on sections of midbrain collected from 15 people diagnosed with Parkinson’s and compared them with labelled T cells on sections from 7 unaffected control brains. The researchers used two labelling markers of T cells, one highlighted CD4 T cells and the other pointed out CD8 T cells.
What’s the difference between CD4 and CD8 T cells?
T cells can be divided into subsets based on their function. Two of the primary subtypes are:
- CD4 helper T cells
- CD8 cytotoxic T cells
CD4 helper T cell, as the names suggest, ‘help’ other cells of the immune system.
CD8 cytotoxic T cells are important for immune defence against intracellular pathogens, such as viruses and bacteria, as well as cancer tumour surveillance.
 Source: Wikipedia
Source: Wikipedia
When they looked at these two types of T cells in the substantia nigra of people who passed away with Parkinson’s, the investigators found no difference in the number of CD4 T cells when compared with unaffected control samples, suggesting that there is no infiltration of CD4 helper T cells in Parkinson’s.
In the control brain, CD8 cytotoxic T cell numbers higher compared to CD4 T cells, but they were also much more abundant in the vascular spaces. When the investigators looked at the Parkinson’s brains, however, they found that the density of CD8 T cell increased 2.5 times (compared to controls), suggesting that cytotoxic T lymphocytes may be infiltrating the brain in Parkinson’s. And this infiltration correlated with the level of neuronal death in the Parkinson’s brains – as the level of cell death increased, so too did the number of CD8 cytotoxic T cells.
CD8 T cell could be observed in close proximity to (and even contacting) dopamine neurons. The researcher also found them in regions where debris from dead dopamine neurons could be found (identified by extracellular neuromelanin).
These results were very interesting for the investigators, but they wanted to dig further.
Specifically, they wanted to determine whether this CD8 T cell infiltration might be an important step in the early stages of Parkinson’s – or do the CD8 T cells simply turn up late to the party?
To evaluated this, the researchers examined sections of the substantia nigra region of the brain from 9 individuals who passed away with incidental Lewy body disease.
What is incidental Lewy body disease?
Lewy bodies are dense circular clusters of aggregated alpha synuclein protein (and other proteins) that are found in specific regions of the brain in people with Parkinson’s (Click here for more on Lewy bodies).
Lewy bodies are one of the cardinal features of the Parkinsonian brain – they are used to help make postmortem diagnoses.
 A Lewy body indicated within the cell body. Source: Alzheimer’s news
A Lewy body indicated within the cell body. Source: Alzheimer’s news
Previous studies have suggested that Lewy pathology occurs in between 8–17% of “neurologically-normal” people who are over the age of 60. That is to say, there is evidence of Lewy bodies in their brains, but they exhibited no clinical evidence of Parkinson’s.
Pathologists have termed these cases incidental Lewy body disease.
It has been proposed that cases of incidental Lewy body disease could represent a presymptomatic, early stages of Parkinson’s. And they have been used in analyses that have attempted to develop staging systems for Lewy body disorders.
Staging systems such as this one:
 Title: Unified staging system for Lewy body disorders: correlation with nigrostriatal degeneration, cognitive impairment and motor dysfunction.
Title: Unified staging system for Lewy body disorders: correlation with nigrostriatal degeneration, cognitive impairment and motor dysfunction.
Authors: Beach TG, Adler CH, Lue L, Sue LI, Bachalakuri J, Henry-Watson J, Sasse J, Boyer S, Shirohi S, Brooks R, Eschbacher J, White CL 3rd, Akiyama H, Caviness J, Shill HA, Connor DJ, Sabbagh MN, Walker DG; Arizona Parkinson’s Disease Consortium.
Journal: Acta Neuropathol. 2009 Jun;117(6):613-34.
PMID: 19399512 (This report is OPEN ACCESS if you would like to read it)
In this report, the researchers examined a set of 10 specific regions across 417 postmortem brains. They analysed the sections for alpha synuclein protein and suggested a new staging system that allowed them to classify all of the subjects with Lewy body disorders (Parkinson’s, Lewy body dementia, Alzheimer’s, etc).
The classification system that the investigators proposed involved 4 stages:
- 1. Alpha synuclein in just the olfactory region of the brain
- 2a Alpha synuclein predominantly in the brainstem
- 2b Alpha synuclein predominantly in the limbic system
- 3. Alpha synuclein in both the brainstem and limbic system
- 4. Alpha synuclein in the neocortical regions
The progression (from 1 to 4) of the cases through these stages was associated with a general worsening in terms of:
- Dopamine concentrations in the striatum (the region of the brain where most dopamine is released)
- Dopamine neurons in the substantia nigra
- Cognitive scores (such as the Mini Mental State Exam)
- Score on the motor component of the Unified Parkinson’s Disease Rating Scale Part 3.
The researchers of the new CD4/CD8 T cell study applied this staging system to all of the brains that they were analysing, including the 9 incidental Lewy body disease cases.
And this is where the story gets really interesting:
You see, the researchers found that the brains that fell into stage 1 of the staging system (Lewy pathology only in the olfactory system) had higher densities of CD8 T cells than any of the other stages.
And it is important to understand that in stage 1, there is no Lewy bodies present in the substantia nigra and no loss of dopamine neurons. This finding suggested to the researchers that CD8 T cell infiltration precedes both alpha synuclein aggregation and neuronal cell death (starting in stage 2 – see graphs below).
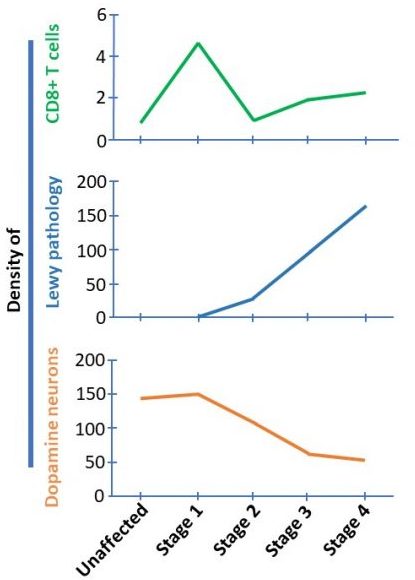 Begs the question, what attracted the CD8 T cells to go rushing into the substantia nigra so early in the disease process???
Begs the question, what attracted the CD8 T cells to go rushing into the substantia nigra so early in the disease process???
|
# # RECAP #2: Analysis of T cells in the postmortem brains of people who passed away with Parkinson’s suggest that a particular type of T cell is elevated in the PD brain. The most interesting discovery, however, is that the highest level of these cells occurs before any cell loss or Parkinson’s pathology occurs – before Parkinson’s is even diagnosed. # # |
Has this ever been reported before?
Infiltration of T-cells in the Parkinsonian brain?
Yes.
In 2009, this report was published:
 Title: Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease.
Title: Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease.
Authors: Brochard V, Combadière B, Prigent A, Laouar Y, Perrin A, Beray-Berthat V, Bonduelle O, Alvarez-Fischer D, Callebert J, Launay JM, Duyckaerts C, Flavell RA, Hirsch EC, Hunot S.
Journal: J Clin Invest. 2009 Jan;119(1):182-92.
PMID: 19104149 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers wanted to determine if Parkinson’s was associated with leukocyte infiltration of the brain, and if this process contributes to degeneration. So, like the study reviewed above, the investigators analysed postmortem sections of brain and found that levels of both CD8 and CD4 T cells are elevated in the substantia nigra of people with Parkinson’s.
 Source: PMC
Source: PMC
And other groups have reported similar results (Click here for another example)
So what can we do with this new information? Should we block the T cells to stop the cell death associated with Parkinson’s?
T cells turn up to a situation looking for trouble, and if they find it, they will kill/remove it. So the big question is not “should we be trying to stop the T cells“, but more “why are they there in the substantia nigra so early in the disease in the first place?”
What unknown initiator grabs their attention and beckons them in?
There is also another important question to ask regarding this new research: how sure are we that these cases of incidental Lewy body disease (that have high levels of CD 8 T cells) represent an early stage of Parkinson’s?
The ‘8–17% of “neurologically-normal” people who are over the age of 60‘ population do not all go on to develop Parkinson’s (1-2% of the population over the age of 60). So the question has to be asked is this a representative image of “pre-diagnosis” Parkinson’s? The researchers who conducted the CD8 T cell study seem to be pretty confident that these cases do represent an early stage of PD, but acknowledge that their report is just a “descriptive and correlative study” and that further research is required.
It would be interesting to see a post mortem analysis of T cells in the brains of individuals with REM sleep behaviour disorder (or RBD) who have not developed Parkinson’s. RBD is considered to be a prodrome (or early indicator) of Parkinson’s, and individuals with RBD have a higher risk of developing PD (Click here to read more about this).
So what does it all mean?
Researchers have recently reported that a specific type of immune cell – called a CD8 cytotoxic T cell – may be present in elevated numbers in the substantia nigra of the Parkinsonian brain before any aggregation of alpha synuclein protein or dopamine neuronal cell loss occurs. This result may be providing us with a tantalizing glimpse into the very early stages of the condition.
Determining the early initiators of pathology in the Parkinsonian brain will provide us with critical insight not only into the underlying biology of the condition, but also into how we can best slow the progression of it. It would be interesting if in the next step of this research, the investigators could somehow isolate the regions surrounding these T cells (from the postmortem tissue) and screen them for pathogens or changes in neuronal/glial function.
This new report is really intriguing and we should all be looking forward to independent replication and expansion of the initial findings.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from Niaid.

I love the depth-first dive that you do in your reports, and this article is a perfect example. You just fill in all of the background needed to understand the central topic of the article, which sometimes involves reviewing a lot of basic science. And that makes topics accessible to a lay audience that would otherwise be beyond our reach.
So it’s a project that goes beyond Parkinson’s and contributes to a more general education and empowerment of the public, and in particular of the patients whose lives are affected by the research that you are covering. It obviously involves a lot of work, and I just wanted to say that the results are valued and the effort is appreciated.
LikeLike
Hi Lou,
Thank you for the comment and kind words. Very good of you (the cheque is in the mail). Your constructive and thought provoking comments are equally appreciated! I hope all is well.
Kind regards,
Simon
LikeLike
Hello,
I am a PwP who gratefully benefits from Simon’s contributions “to a more general education and empowerment of the public, and in particular of the patients” and would like to ask something: If cytotoxic CD8 T cells normally chase viruses and bacteria, does their presence not indicate that idiopathic PD could be an infectious disease? Has there ever been a systematic search for viral or bacterial DNA (or RNA) in the substantia nigra of people who passed away with PD?
Best regards,
zz
LikeLiked by 1 person
Hi zz,
Thanks for your comment and kind words – much appreciated. Thanks also for the really interesting question. Searching for viral DNA & RNA in postmortem tissue is hard and probably fruitless. In my previous life as a research scientist I applied for funding to do “off target” screening of the UK Medical Research Council Brain Banks Network Resource (which contains DNA sequencing data from 1511 human brains, 39 of which had PD – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5204341/). The idea was to remove all human DNA from the data set and to see what was left, looking for any associations with PD. But the study was never funded, mainly because most viral infections are transcient events so the odds of finding something are rather unlikely. Plus you will only find DNA-based viruses, none of the RNA-based viruses (Hepatitis C, influenza, etc).
The better way to approach the problem is to look at blood. There is a technique called Virscan (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4844011/), which provides a pretty good history of infections for each individual by analysing antiviral antibodies in blood. I did approach the research group about having a look in Parkinson’s, but I think they were overwhelmed with requests from every other medical field at the time. Hopefully someone is doing that analysis now – all it would require is some blood samples and clinical notes (both of which are available through programs like MJFF PPMI) – just saying…
Hope this answers your question.
Kind regards,
Simon
LikeLike
Hi Simon,
thank you very much for the explanations! However, I still have two remarks/questions, both of which might be naive.
(1) You write that “most viral infections are transient events so the odds of finding something are rather unlikely”. I guess this is because once a normal cell has survived a viral infection, it gets rid of the remains of the virus. However, my understanding is that in many cases of PD, the waste disposal does not work properly and some of the garbage ends up in Lewy bodies. So maybe there is some viral DNA hidden in these Lewy bodies? (This reminds me of the newspaper headline from 3 years ago: “Scat burglar: DNA evidence left in toilet leads to arrest of thief”)
(2) Is it really known for sure that the varicella zoster virus (VZV) is not a major cause of PD? (It is a DNA virus and in this case Virscan probably would not help much since most people (in the age group of PwP) have had chickenpox.) VZV is my favourite suspect for the following reasons. (a) It knows how to survive in neurons for decades. (b) Shingles is usually initially limited to one side of the body, just like PD. Perhaps for the same reason? Maybe because the immune system (T-cells?) kicks in before it spreads further? (c) While I was taking the antiviral drug Brivudine this year to treat shingles, my PD symptoms unexpectedly improved a lot!
Thank you for your consideration and kind regards,
zz
LikeLike