|
We are going to talk about a snail model of Parkinson’s disease. I kid you not. Love them or hate them, recent research on snails is helping us to better understand a potential therapeutic target for Parkinson’s disease, called Pituitary adenylate cyclase-activating polypeptide (or PACAP). In today’s post we will look at what PACAP is, outline the new snail research, and discuss what they mean for people living with Parkinson’s disease. |

The humble snail. Source: Warrenphotographic
In a recent post, I talked about a class of drugs called Dipeptidyl peptidase-4 (or DPP-4) inhibitors (Click here to read that post). DPP-4 is a ubiquitous enzyme (it is present on most cells in your body) that breaks down certain proteins.
In that post, I listed some of the proteins that DPP-4 targets – they include:
- Gastrin-releasing peptide (GRP)
- Glucagon
- Glucagon-like peptide-1 (GLP-1)
- Glucagon-like peptide-2 (GLP-2)
- Granulocyte-macrophage colony-stimulating factor (GM-CSF)
- GHRH and IGF-1
- High-mobility group box 1 (HMGB1)
- Macrophage-derived chemokine (MDC)
- Macrophage inflammatory protein-1 α (MIP-1 α), chemokine (C-C motif) ligand 3-like 1 (CCL3L1), or LD78β
- Pituitary adenylate cyclase-activating polypeptide (PACAP)
- Neuropeptide Y (NPY)
- Regulated on activation, normal T cell expressed and secreted (Rantes)
- Stromal cell-derived factor-1 (SDF-1)
- Substance P (SP)
Lots of interesting proteins with regards to Parkinson’s disease on this list, including GLP-1 which has been turned in the drug Exenatide (which has demonstrated positive effects in recent clinical trials for Parkinson’s disease – click here and here to read more about this). Another interesting protein on the list is ‘Granulocyte-macrophage colony-stimulating factor‘ (GM-CSF) which we have also discussed in a previous post (Click here to read that post). A synthetic version of GM-CSF (called Sargramostim) has recently been tested in a clinical trial of Parkinson’s disease in Nebraska, and the results of that Phase I trial have been very encouraging.
By treating people with DPP-4 inhibitors (also known as ‘gliptins’), one would be blocking the breaking down of these potentially beneficial proteins – increasing the general amount of GLP-1 and GMCSF that is floating around in the body.
EDITOR’S NOTE: DPP-4 inhibitors have not yet been clinically tested in Parkinson’s disease, and thus we have no idea if they are safe in people with this condition. They are being mentioned here purely as part of an academic discussion.
One protein on the list of DPP-4 targets above that we have not yet discussed is Pituitary adenylate cyclase-activating polypeptide (or PACAP).
And today we are going to have a look at it.
Why?
Because PACAP is really interesting.
Ok, I’ll bite: what is PACAP?
Pituitary adenylate cyclase-activating polypeptide is a peptide that exhibits potent neurotrophic and neuroprotective properties when it binds to and activates G protein–coupled receptors.
What does any of that actually mean?
Ok, let’s start with a peptide.
A peptide is similar to a protein in that it is an important part of a cells ability to do stuff. But peptides differ from a protein in that they are much smaller. Both peptides and proteins are formed by chains of amino acids, but peptides are defined as molecules that consist of between 2 and 50 amino acids, whereas proteins are anything bigger than that (50 or more amino acids).
Source: Peptidesciences
A polypeptide refers to a peptide of 10 or more amino acids (but less than 50). So PAPAP is a chain of more than 10 amino acids.
PACAP is considered a neurotrophic factor (neurotrophic = Greek: neuron – nerve; trophikós – to feed). These are chemicals produced in the brain that help to nourish the neurons and keep them alive. This neurotrophic feature of PACAP helps to make this polypeptide neuroprotective – it can keep neurons alive when faced with a toxin or an insult. PACAP achieves this via binding to G protein–coupled receptors.
What are G protein–coupled receptors?
Peptides work by binding to a receptor – like a key works with a lock.
The peptide binds to the receptor and this action activates a process (or multiple processes) associated with that receptor. An example of this peptide-receptor interaction is illustrated in the image below where Neurotensin receptor type 1 (NTS1) is presented. NTS1 is not associated with PACAP, but provided here for explanatory purposes.
Example of how a peptide (NTS1) activates a receptor. Source: Nature
The polypeptide PACAP functions via three main receptors: PAC1, VPAC1, and VPAC2.
Once one of these receptors is bound by PACAP, specific processes are activated. Some of these processes are shared between the three receptors, but there are also processes that are unique to each of the them.
Receptors of PACAP. Source: wjgnet
You have probably never heard of G protein–coupled receptors, but they involve about 4% of the protein-producing DNA in each cell of your body. They are a very common mechanism for getting things done inside of cells. When PACAP binds to one of the three receptors and they becomes activated, the part of the receptor structure inside the cell will send a signal via a messenger (called a G-protein).
There are two primary messaging pathways involving the G protein in G protein–coupled receptors:
- the cyclic adenosine monophosphate (cAMP) signal pathway
- the phosphatidylinositol signal pathway
PACAP activation of one of its G protein–coupled receptors increases the cAMP level in target cells. cAMP then goes on to activate other pathways involved in the neurotrophic and neuroprotective activities associated with PACAP.
Click here for an excellent OPEN ACCESS review of PACAP
And PACAP is neuroprotective in models of Parkinson’s?
It certainly appears to be.
Back in 2004, this research report was published:
Title: Pituitary adenylate cyclase activating polypeptide protects dopaminergic neurons and improves behavioral deficits in a rat model of Parkinson’s disease.
Authors: Reglodi D, Lubics A, Tamás A, Szalontay L, Lengvári I.
Journal: Behav Brain Res. 2004 May 5;151(1-2):303-12.
PMID: 15084446
Rats were administered PACAP (0.1mg) before injected with a neurotoxin (6-hydroxydopamine) that models Parkinson’s disease. PACAP-treated animals showed better behavioural outcomes when compared to the untreated control animals and they lost significantly less dopamine neurons. The researchers concluded that these results suggest a potential neuroprotective role for PACAP in Parkinson’s disease.
While the findings were very interesting, this first study provided little indication as to how PACAP might be having a neuroprotective effect on the cells, but subsequent research has provided clues.
Such as this study:

Title: Effect of the pituitary adenylate cyclase-activating polypeptide on the autophagic activation observed in in vitro and in vivo models of Parkinson’s disease.
Authors: Lamine-Ajili A, Fahmy AM, Létourneau M, Chatenet D, Labonté P, Vaudry D, Fournier A.
Journal: Biochim Biophys Acta. 2016 Apr;1862(4):688-95.
PMID: 26769362 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers found that pre-treating cells with PACAP before exposing them to a neurotoxin (MPTP) could counteract the toxic effects, completely rescuing the cells death (which was 50% loss in the untreated control cells that were exposed to the toxin). The investigators found that this neuroprotective effect was partly due to an inhibition of the waste disposal system (autophagy) – PACAP treatment reduced the autophagic activity both in cell culture and in a rodent model of Parkinson’s.
Are this the only research on PACAP in models of Parkinson’s?
No.
Numerous groups have now demonstrated that PACAP is having a beneficial effect in models of Parkinson’s disease, both in cell culture experiments (Click here, here and here to read examples) and in rodent models of the condition (Click here, here and here to see some of that research).
And researchers are now looking at synthetic versions of PACAP, with the goal of developing a drug that could be taken to the clinic:

Title: Characterizations of a synthetic pituitary adenylate cyclase-activating polypeptide analog displaying potent neuroprotective activity and reduced in vivo cardiovascular side effects in a Parkinson’s disease model.
Authors: Lamine A, Létourneau M, Doan ND, Maucotel J, Couvineau A, Vaudry H, Chatenet D, Vaudry D, Fournier A.
Journal: Neuropharmacology. 2016 Sep;108:440-50.
PMID: 26006268
These researchers have developed a new and improved version of PACAP, which they have called (I kid you not) Ac-[Phe(pI)(6), Nle(17)]PACAP(1-27).
Catchy name, huh? Try saying it three times really fast!
Importantly, when the investigators tested Ac-[Phe(pI)(6), Nle(17)]PACAP(1-27) in cell culture experiments they found that it stimulated cAMP production, maintained normal mitochondrial function and protected cells from exposure to a neurotoxin (MPTP). Ac-[Phe(pI)(6), Nle(17)]PACAP(1-27) also rescued dopamine neurons in MPTP-treated mice. A very pleasing result – it will be interesting to watch how Ac-[Phe(pI)(6), Nle(17)]PACAP(1-27) develops.
And what about the snail model of Parkinson’s disease you mentioned?
Ah yes.
So ten years ago, this study was published: 
Title: Behavioural and neural deficits induced by rotenone in the pond snail Lymnaea stagnalis. A possible model for Parkinson’s disease in an invertebrate.
Authors: Vehovszky A, Szabó H, Hiripi L, Elliott CJ, Hernádi L.
Journal: Eur J Neurosci. 2007 Apr;25(7):2123-30.
PMID: 17439496
In this study, the researchers investigated the idea of modelling Parkinson’s disease in the common pond snail, called Lymnaea stagnalis:
Cute huh?
But why?
For several good reasons:
-
Lymnaea stagnalis is one of the most widely used organisms in invertebrate neurobiology (we know a great deal about it)
-
They have bigger brains than flies (which are also widely used for Parkinson’s research)
-
They exhibit relatively simple behaviours – locomotion, respiration and feeding – which can be easily quantified and analysed.
- Researchers can acquire those results very quickly.
And the researchers demonstrated this by treating the snails with the commercially available organic pesticide, Rotenone – which has been associated with Parkinson’s disease. They found that it is rather toxic to these snails. At various concentrations (from 0.1 to 5 microM), the investigators found that rotenone exposure caused progressive and irreversible motor problems for the snails. In the image below, note the reduction in the distance travelled by snails exposed to rotenone. One hour after exposure the snail (in the middle panel) has travelled significantly less than the control snail (left panel), and after 4 hours most of the exposed snails were almost completely immobilized.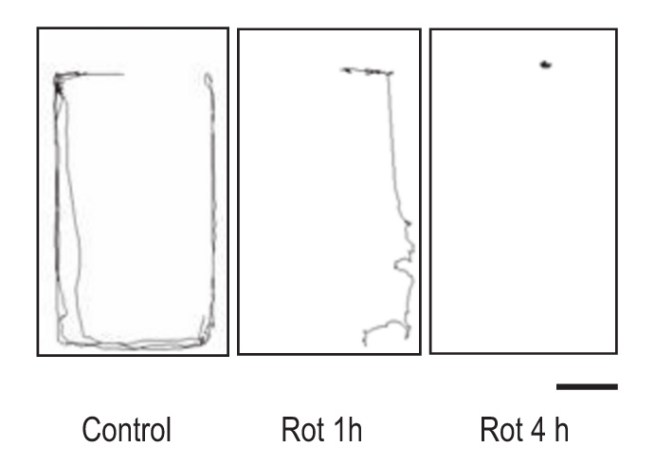
Lines indicating distance travelled by snails around an enclosure after exposure to rotenone (Rot). Source: Wiley
When the researchers looked at the brains of the snails, they found that levels of the chemical dopamine were reduced by 75%. The investigators concluded that “the rotenone-treated pond snails show many common features with widely used models of Parkinson’s disease” and they proposed that this model could be utilised for screening and testing drugs for Parkinson’s.
And that is exactly what some researchers have done recently with PACAP:
Title: Pituitary adenylate cyclase-activating polypeptide (PACAP) has a neuroprotective function in dopamine-based neurodegeneration in rat and snail parkinsonian models.
Authors: Maasz G, Zrinyi Z, Reglodi D, Petrovics D, Rivnyak A, Kiss T, Jungling A, Tamas A, Pirger Z.
Journal: Dis Model Mech. 2017 Feb 1;10(2):127-139.
PMID: 28067625 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers also observed decreased locomotor and feeding activity rotenone-treated snails, which corresponded very well with the results they obtained in rats treated with a neurotoxin (6-OHDA). By treating the snails and rats with PACAP, the researchers were able to prevent the behavioural problems caused by the two neurotoxins. Levels of dopamine in the PACAP snails were rescued by 50% compared to the untreated controls.
Interesting fact about snails: they have no blood-brain barrier (the membrane surrounding the brain in mammals; Source).
One curious difference between the snail and rat model of Parkinson’s: the researchers reported a decrease in levels of the Parkinson’s associated protein DJ-1 in both models, but PACAP had a beneficial effect on DJ-1 levels only in the rats. The remaining results between the two models were very similar though, which gave the investigators great confidence in concluding that PACAP is neuroprotective.
The only remaining question is whether the snails (and rats) that were exposed to PACAP went on to get headaches.
Que?
So, this is where the whole PACAP story gets a little tricky:
See, if you inject someone with PACAP, they are very likely to go on to get a whopping great headache over the next few hours: 
Title: PACAP38 induces migraine-like attacks in patients with migraine without aura.
Authors: Schytz HW, Birk S, Wienecke T, Kruuse C, Olesen J, Ashina M.
Journal: Brain. 2009 Jan;132(Pt 1):16-25.
PMID: 19052139 (This article is OPEN ACCESS if you would like to read it)
There are two versions of PACAP in the human body: PACAP-38 and a smaller version, PACAP-27. PACAP-38 is the most common of the two polypeptides as it represents more than 90% of the total PACAP content in your body. In this study, the researchers wanted to see if there was any difference in response to PACAP between normal healthy individuals and people who suffer from migraines.
PACAP38 infusions caused headaches in ALL of the healthy subjects and 11 out of 12 migraine sufferers!
But this is where synthetic PACAP may be very beneficial.
In the Ac-[Phe(pI)(6), Nle(17)]PACAP(1-27) study mentioned above, when the researchers injected this drug into mice they reported a fall of average arterial pressure which was lower both in intensity and duration when compared with normal PACAP. So hopefully future derivatives of PACAP will be able to provide a robust neuroprotective effect without the whacking great migraine.
Has PACAP ever been investigated in the clinic?
No, it hasn’t (that I am aware of).
What does it all mean?
Pituitary adenylate cyclase-activating polypeptide (PACAP) is a neuroprotective agent that is worthy of further investigation within the realm of Parkinson’s disease. Numerous cell culture and animal models of Parkinson’s have demonstrated that this polypeptide can have beneficial effects. The hope is now that derivatives of PACAP can be successfully generated and taken into clinical testing (without the negative side effect of stimulating headaches).
The banner for today’s post was sourced from Healthfitnessboss






nicely written
LikeLike