|
Researchers have recently described a new method to quantify a person’s “immune age” – a measure that could act as a key determinant of future health, as well as response to disease and treatment. This novel test appears to provide a more reliable predictor for the status of one’s immune system than any other previous method. And it could be useful in other ways. In today’s post, we will discuss this new method of determining “immune age”, explore examples of how similar analysis has been used for other conditions, and consider what it could mean for Parkinson’s.
|
 Source: Emaze
Source: Emaze
Do you remember Andre Agassi?
I know he’s still around, but when I was young and less beautiful, I was a big fan. Not only of his on court achievements, but also of his charismatic off-court image.
And it certainly paid off well for him:
One of the things that Agassi taught us was that “image is everything”.
Before Agassi, tennis was a conservative sport of white shirts & shorts (McEnroe was basically as radical as things got). It was bland, conservative, and – yes, I’ll say it – boring.
Agassi not only brought colour but charisma to the game. It was shocking and disgraceful to some, but to young, naive fools like me, it was a captivating breath of much needed fresh air.
 Source: Hesaidandshesaid
Source: Hesaidandshesaid
Despite the early infatuation with the stylings of Mr Agassi, I have to admit that I have never remotely been concerned about own image. My dimensions mean that I wear what fits as opposed to what I like, and as a result the finished product is better behind a keyboard rather than speaking to a crowd.
But as I have gotten older, I have become concerned about a different kind of IMM-AGE (not a typo).
Let me explain: Recently some researchers in Israel and at Stanford University in the US published a rather remarkable research report which if replicated could have important implications for how we approach medical care.
What did they report?
Here is their published report:
 Title: A clinically meaningful metric of immune age derived from high-dimensional longitudinal monitoring.
Title: A clinically meaningful metric of immune age derived from high-dimensional longitudinal monitoring.
Authors: Alpert A, Pickman Y, Leipold M, Rosenberg-Hasson Y, Ji X, Gaujoux R, Rabani H, Starosvetsky E, Kveler K, Schaffert S, Furman D, Caspi O, Rosenschein U, Khatri P, Dekker CL, Maecker HT, Davis MM, Shen-Orr SS.
Journal: Nat Med. 2019 Mar;25(3):487-495.
PMID: 30842675
In this study, the researchers collected blood samples from 135 healthy adult individuals of different ages (63 young adults (aged 20–31 at enrollment) and 72 older adults (aged 60–96 at enrollment)) over a period of eight years.
 Source: Researchgate
Source: Researchgate
The samples were analysed using two experimental designs that resulted in two data sets for the researchers to analyse:
- an ongoing design (in which samples were annually analysed using an array of platforms, such as cellular phenotyping, cytokine-stimulation assays, and whole-blood gene-expression).
- a snapshot design (this involved longitudinal samples from 18 participants (3 young adults and 15 older adults) being simultaneously anaylsed to remove the chance of any year-to-year technical variation.
The results of this effort allowed the researchers to capture both population and individual changes in the immune system over time.
Interesting. So what did they find?
Using machine learning, the researchers identified patterns of changes in subsets of cells common to those in the study, despite the fact that there was large amounts of variation in the state of each individual immune system.
In particular, the researchers found that the changes in the immune status of an individual were more affected by personal immunosenescence measures at baseline than by actual chronological age.
Interesting. But what does that mean?
Which part?
All of it. What are immunosenescence measures?
Our immune system is made up of lots of different types of cells, and as we age the ability of those cells to function properly changes.
Hematopoietic stem cells, for example. These cells – which reside in our bone marrowm – are the cells that replenish our blood supply throughout life.
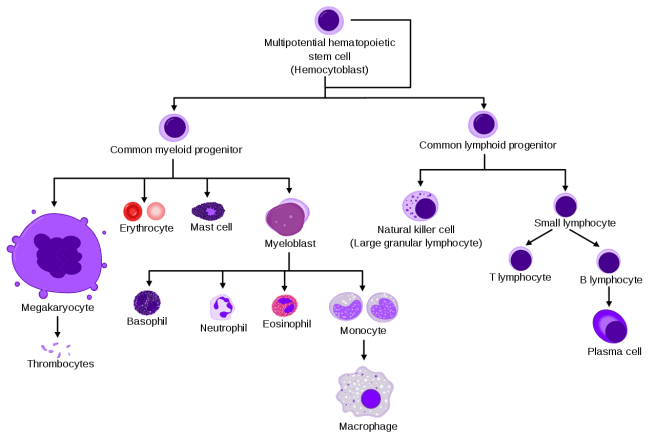 The progeny of hematopoietic stem cells. Source: Wikipedia
The progeny of hematopoietic stem cells. Source: Wikipedia
Their ability to self-renew and supply new cells, however, diminishes over time. This is due to a number of different factors (eg. the accumulation of oxidative damage to DNA, cellular metabolic activity, the shortening of telomeric terminals of chromosomes, etc).
Likewise, phagocytes.
 A schematic of a phagocyte. Source: Meducator
A schematic of a phagocyte. Source: Meducator
These cells travel around our bodies consuming and breaking down anything that should not be there. But there is a decline in the total number of them as we age, and they experience an intrinsic reduction in their ability to do their job (Click here to read more about this).
Immunosenescence is a blanket term which refers to this gradual deterioration of the immune system over time.
 Changes in T cells as we age. Source: Frontiers
Changes in T cells as we age. Source: Frontiers
This reduction is caused by natural age advancement and environmental factors. It involves both the host’s capacity to respond to infections and the development of long-term immune memory.
But it can be measured using analysis of the blood cells.
And by doing this, what the researchers found was that changes in the immune status of an individual over time were more affected by the measures of personal immunosenescence at baseline than by actual chronological age. This observation allowed the researchers to be able to calculate an immune age (or IMM-AGE) for each person at any given time.
I see. Cute name. And what did they do with this new method of measuring “IMM-AGE”?
The researchers were keen to determine if IMM-AGE was connected to overall health and longevity. So they accessed data from 2,292 participants (aged 40–90 years) in the Framingham Heart Study – a longitudinal analysis of cardiovascular disease.
 Source: BU
Source: BU
The researchers used the rich clinical and cardiovascular information – in addition to whole-blood gene-expression data – from these participants and they were able to assign each participant an approximate IMM-AGE score.
Remarkably, the investigators found that IMM-AGE was a very good predictor of mortality. In fact, the association of IMM-AGE with overall survival was >500-fold more significant than that obtained other measures of aging (such as DNA methylation age).
This finding obviously highlights the important role of immune aging in overall survival.
Wow! Fascinating. But what does any of this have to do with Parkinson’s?
This new data could significantly change the way we develop new treatments for Parkinson’s. It will be interesting to see if future therapies can be designed to better maintain one’s IMM-AGE, and this could potentially have benefits for Parkinson’s.
In addition, think about how we clinically test new therapies.
We take 200 people and we blindly/randomly split them into two “equal” groups. We then treat one group with the drug of interest and the other with a placebo. Both of these groups are apparently equally balanced. But one can envisage a future where the IMM-AGE of each individual will be part of the eligibility criteria for taking part in a clinical trial.
And think about how this kind of analysis could be applied to identifying responders in drug trials.
Que? You’ve lost me.
The research in this report was partly conducted by a biotech firm called CytoReason.
CytoReason is an Israeli company that previously published this report here:
 Title: Cell-centred meta-analysis reveals baseline predictors of anti-TNFα non-response in biopsy and blood of patients with IBD.
Title: Cell-centred meta-analysis reveals baseline predictors of anti-TNFα non-response in biopsy and blood of patients with IBD.
Authors: Gaujoux R, Starosvetsky E, Maimon N, Vallania F, Bar-Yoseph H, Pressman S, Weisshof R, Goren I, Rabinowitz K, Waterman M, Yanai H, Dotan I, Sabo E, Chowers Y, Khatri P, Shen-Orr SS; Israeli IBD research Network (IIRN).
Journal: Gut. 2019 Apr;68(4):604-614.
PMID: 29618496 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers wanted to try and determine predictive biomarkers of people with inflammatory bowel disease who do not respond to anti-TNFα therapy.
And what does any of that mean? What is inflammatory bowel disease?
Inflammatory bowel disease (or IBD) is one of these umbrella terms that is used to refer to a group of inflammatory conditions of the large and small intestine:
 The large and small intestine. Source: Adam
The large and small intestine. Source: Adam
The symptoms of IBD can include abdominal pain, diarrhoea, vomiting, rectal bleeding, severe internal cramps/muscle spasms in the region of the pelvis, and weight loss.
The most common forms of IBD are Crohn’s disease and ulcerative colitis.
There has been an increased incidence of IBD since World War II, which could be associated with increased awareness and reporting of the condition, but it could also be linked with increases in meat consumption (Click here to read more about this). For example, in 2015, an estimated 1.3% of U.S. adults (3 million) were diagnosed with IBD, which was a large increase on the levels in 1999 (0.9% or 2 million adults – Source: CDC).
And what on Earth is anti-TNFα therapy?
Tumor necrosis factor alpha (or TNFα) is a small cytokine that is involved in inflammation.
What is a cytokine?
Cytokines (from the Greek: kýtos meaning ‘container, body, cell’; and kī́nēsis meaning ‘movement’) are small proteins that are secreted by cells in the body and they have an effect on other cells.
Cytokines are a method of communication for cells.
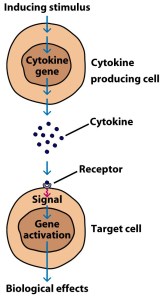
How cytokines work. Source: SBS
In the case of TNFα, the signal that is passing on to the cell it attaches to is one of inflammation.
What is inflammation?
Inflammation is an immune response to a situation in the body. The function of inflammation is to eliminate the initial cause of the problem (such as a damage causing pathogen like a virus), clear out dying cells and tissues damaged from the original insult, clear out dying cells and tissues damaged by the inflammatory process itself, and initiate tissue repair.
TNF is produced by blood cells (eg. macrophages and monocytes) during early inflammatory events. It triggers the production and release of a series of other inflammatory molecules/cytokines – further inflaming the situation.
So ‘anti-TNFα therapy’ is a treatment against inflammation?
Exactly.
Anti-TNFα therapy blocks or inhibits the production of TNFα, thus limiting inflammation signaling.
But while anti-TNFα therapy has been a major breakthrough for the treatment of IBD, more than 30% of patients will be non-responders to the treatment – meaning that anti-TNFα therapy will have no impact on the condition for these individuals.
So for a long time researchers have wanted to find biomarkers which could help to identify anti-TNFα therapy non-responders before doctors initiate treatment, saving both patient time and hospital resources.
The CytoReason investigator analysed publicly available DNA profiles of colon biopsies obtained from multiple studies of patients with IBD. Their machine learning approach (similar to the IMM-AGE method described above) suggested that increased levels of activity in two genes (Triggering receptor expressed on myeloid cells 1 (or TREM-1) and chemokine receptor type 2 (CCR2)–chemokine ligand 7 (CCL7)) were particularly apparent in non-responders.
In blood samples, the researchers found that when TREM-1 levels were high in an individuals with Crohn‘s disease at baseline, it was predictive of that individual being a non-responder to anti-TNFα treatment.
The study proposed two assays – one on biopsies and one on blood – that are clinically feasible for predicting – with high accuracy – non-responders to anti-TNFα therapy prior to initiation of treatment.
But what does any of this have to do with Parkinson’s?
It would be interesting to apply some of this sort of analysis to various aspects of Parkinson’s.
Such as?
Well, take response to Levodopa for example.
Clinical research suggests that only a portion of the Parkinson’s affected community respond well to this standard treatment:
 Title: Developing and validating Parkinson’s disease subtypes and their motor and cognitive progression
Title: Developing and validating Parkinson’s disease subtypes and their motor and cognitive progression
Authors: Lawton M, Ben-Shlomo Y, May MT, Baig F, Barber TR, Klein JC, Swallow DMA, Malek N, Grosset KA, Bajaj N, Barker RA, Williams N, Burn DJ, Foltynie T, Morris HR, Wood NW, Grosset DG, Hu MTM.
Journal: J Neurol Neurosurg Psychiatry. 2018 Dec;89(12):1279-1287
PMID: 30464029 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers conducted a data-driven analysis of clinical data to determine the existence and natural history of subtypes of Parkinson’s. They did this using two large independent cohorts of patients who were newly diagnosed with the condition and followed over time (clinical data from 1601 and 944 individuals, from the Tracking Parkinson’s and Discovery cohorts, respectively).
The results suggested 4 clusters (or subtypes) of Parkinson’s:
 Source: PMC
Source: PMC
Of interest to our discussion in this particular post is the responders to levodopa: only 1/3 of individuals with Parkinson’s are good responders (Cluster 2 – good responders – 29%, Cluster 3 – intermediate responders – 21%, Cluster 1 and Cluster 4 combined – poor responders – 50%).
It would be interesting to conduct an IMM-AGE-sort of analysis on a large cohort of good, intermediate and poor responders to levodopa to assess whether there are any key determinants that could indicate good responders early.
In addition, in every clinical trial of novel therapies we have responders and non-responders.
Rather than simply rubbishing a new treatment if there are not enough responders to shift the average, why do we not conduct an IMM-AGE-sort of analysis on the actual responders (compared to the non-responders)? This type of approach could be very useful for determining non-responders to novel experimental therapies, such as exenatide (Click here to read a previous SoPD post on the topic of responders to Exenatide).
In fact, it could be REALLY interesting to go back through old “failed” clinical trials and look at the few individuals who may have responded to each treatment and compare them to the non-responders.
Yes, there is the possibilty of pacebo responses, and yes, it would be a lot of work for someone, but surely it is an idea worth exploring.
Just a thought.
So what does it all mean?
Israeli scientists have determined a novel method of determining the age of a person’s immune system (called IMM-AGE) and they have found that it is a more accurate measure of the state of the immune system of the individual than actual cronological age.
 Source: Scientificeuropean
Source: Scientificeuropean
While this new method needs to be independently replicated and further explored, it could have important implications for how we develop new medications in the future (both the treatments and how we conduct the clinical trials). The analytical approach behind the study could also be useful to determining biomarkers for drug responders to new experimental therapies, which could give us a more efficient mechanism of determining clinical trial success.
The banner for today’s post was sourced from my-healing-hands.


Simon, thought you and readers might be interested in this interview I did with Shai Shen Orr, the lead author of the study mentioned above…
Shameless plug aside, I’d be interested to hear your thoughts on his last statement, “I think in the future targeting the immune system will be much more beneficial as it is easier to examine and has a greater impact on our health than our genetics.”
LikeLike