|
Last week the German biotech firm MODAG announced that they had secure €12M in series A funding from various venture capital investors. The company is going to use those funds to clinically develop their lead compound – Anle138b – in the neurodegenerative condition, Multiple Systems Atrophy (or MSA). In today’s post, we will discuss how Anle138b works, what Multiple Systems Atrophy is, and how this news could be good for the Parkinson’s community.
|
 Stealth mode. Source: Hackernoon
Stealth mode. Source: Hackernoon
Last week a small biotech firm in Germany came out of ‘stealth mode’.
What is stealth mode?
According to wikipedia, “in business, stealth mode is a company’s temporary state of secretiveness, usually undertaken to avoid alerting competitors to a pending product launch or other business initiative”.
After years of developing a novel drug, the German company emerged from stealth mode with €12M in series A funding, which will be used to clinically test their new treatment.
The company’s name is MODAG.
 And what is MODAG planning to do now they are out of “stealth mode”?
And what is MODAG planning to do now they are out of “stealth mode”?
They are planning to clinically test their lead compound which is called Anle138b.
The initial Phase I safety test will be conducted in healthy individuals, but then they will turn their attention to individuals with multiple systems atrophy.
What is Multiple System Atrophy?
Multiple System Atrophy (or MSA), also known as Shy-Drager syndrome, is a rare neurodegenerative condition.
It is considered an Atypical Parkinsonism.
What is an ‘Atypical Parkinsonism’?
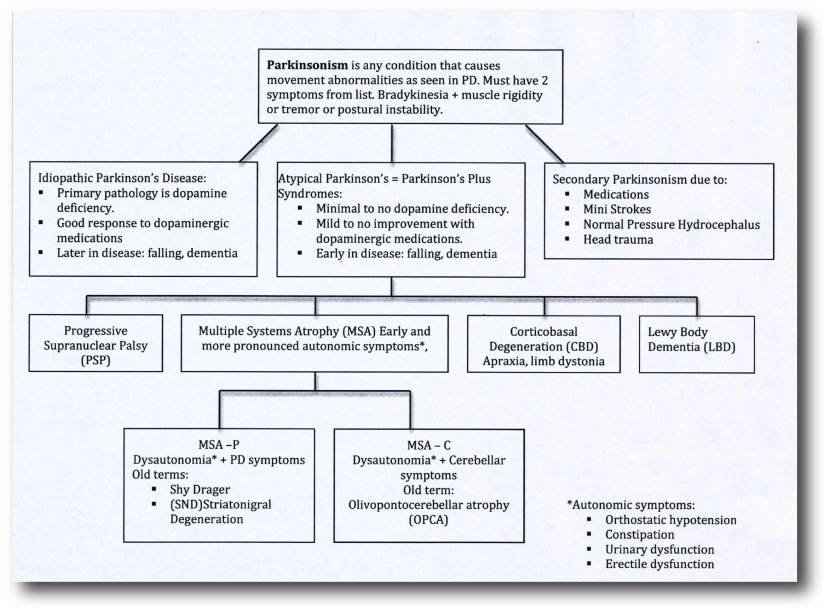
‘Parkinsonisms’ refer to a broad group of neurological conditions that cause movement features similar to those observed in “Parkinson’s disease”, such as tremors, slow movement and stiffness. The name ‘Parkinsonisms’ is often used as an umbrella term that covers “Parkinson’s disease” and all of the other ‘Parkinsonisms’.
Parkinsonisms are generally divided into three groups:
- Classical idiopathic Parkinson’s (the common spontaneous form of the condition we refer to as “Parkinson’s disease”)
- Atypical Parkinson’s or Parkinson-plus syndromes (such as multiple system atrophy and Progressive supranuclear palsy (PSP))
- Secondary Parkinson’s (which can be brought on by mini strokes (aka Vascular Parkinson’s), drugs, head trauma, etc)
 Source: Parkinsonspt
Source: Parkinsonspt
So what is the difference between Multiple System Atrophy and idiopathic Parkinson’s?
When a person first presents at the clinic with rigidity, slowness of movement and a resting tremor, it can be very difficult to differentiate between classical idiopathic Parkinson’s and other kinds of Parkinsonisms. But there are some telltale signs that can help differentiate the Multiple System Atrophy from idiopathic Parkinson’s:
- The disability progresses more rapidly in MSA
- People with MSA are poor responders to levodopa treatment over time (such as Sinemet)
- Urinary retention and orthostatic hypotension (blood pressure falls when suddenly standing up) are common
- Rigidity and bradykinesia are out of proportion to tremor
- Speech is severely affected
- Gasping and high pitched wheezing sounds when breathing are present
- In the vast majority of cases, there is an absence of dementia, though other cognitive functions can be affected
- Reduced blinking and dry eye, in addition to jerky or slower eye movements
It should be noted that the presence of one or two of these signs is not necessarily indicative of an individual having MSA.
Are there different types of MSA?
The Second Consensus Statement on the diagnosis of multiple system atrophy provides for two basic types of multiple system atrophy, based on the symptoms of the condition at the time of evaluation.
These two types are:
- MSA with predominant parkinsonism (MSA-P), which can resemble idiopathic Parkinson’s because of slow movement and stiff muscles. The terms striatonigral degeneration or parkinsonian variant are sometimes used for this category of MSA.
- MSA with cerebellar features (MSA-C). It is sometimes termed ‘sporadic olivopontocerebellar atrophy’ (or OPCA). MSA-C primarily affects balance, coordination, and speech.
Some researchers believe that there is also a third type of MSA which is a combination of the two.
How is Multiple System Atrophy diagnosed?
Currently there is no test that can definitively make or confirm the diagnosis of MSA in a living person.
Having said that, regular use of brain imaging techniques such as MRI and CT scanning can show a decrease in the size of specific brain structures affected in MSA (such as the cerebellum and pons). For example, in the image below you can see serial MRI images of the brain of a person with MSA-C over a 2-year period. Note the subtle reduction in the size of the characteristic ‘hot cross bun’ shape of the pons between image C (2004) and two years in image D (2006):
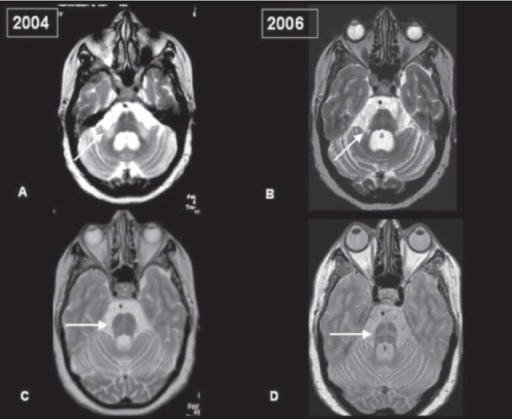 Serial MRI of brain of a person with MSA-C over a 2-year period. Source: Openi
Serial MRI of brain of a person with MSA-C over a 2-year period. Source: Openi
And in MRI brain scan images from individuals affected by MSA-P, there is a gradual darkening of the region of the brain called the putamen (indicated on the left side of the brain by a white arrow in the image below). You can see that over time as the condition progresses (from grade 0 to grade 3), the putamen becomes darker and darker.
 MRI of brain of a person with MSA-P over time. Source: e-jmd
MRI of brain of a person with MSA-P over time. Source: e-jmd
What causes Multiple System Atrophy?
I think most researchers would agree that the correct answer to this question is:
 Source: Wellbeing365
Source: Wellbeing365
But there are many researchers who believe that the development of MSA is associated with the aggregation of the Parkinson’s-associated protein, alpha synuclein.
Remind me one more time what is alpha synuclein?
Alpha synuclein sounds like a distant galaxy, but it is one of the most common proteins in our brains. It makes up about 1% of all the protein in a neuron. When alpha synuclein protein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure.
When it is first produced, alpha synuclein will look something like this:

Alpha synuclein. Source: Wikipedia
In this form, alpha synuclein is considered a monomer – which is a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). It is believed that alpha synuclein has certain functions as a monomer, but may also have specific tasks as an oligomer.
In Parkinson’s, alpha synuclein will also misfold and aggregate together to form amyloid fibrils.

Microscopic images of monomers, oligomers and fibrils. Source: Brain
And it is believed that the oligomer and fibril forms of alpha synuclein protein that aggregate together, and then go on to form what we call Lewy bodies.
 Parkinson’s associated alpha synuclein. Source: Nature
Parkinson’s associated alpha synuclein. Source: Nature
A Lewy body is referred to as a cellular inclusion, as they are almost always found inside the cell body. They are a characterisitic feature of the Parkinsonian brain.
 A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
But I thought alpha synuclein is associated with Parkinson’s?
It is, but there is some research suggesting that different types (or species) of apha synuclein may be influencing the two conditions (Click here to read more about this).
So MODAG are going to test their drug (Anle138b) in MSA?
Yes.
And what does Anle138b do?
Anle138b is a diphenyl–pyrazole compound (don’t worry about what that means), which displays potent anti-protein aggregating properties.
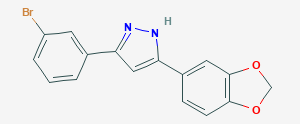 Anle138b. Source: Pubchem
Anle138b. Source: Pubchem
In particular, this compound has been shown to inhibit the formation of the toxic form oligmeric and fribrillar forms of alpha synuclein:

Title: Anle138b: a novel oligomer modulator for disease-modifying therapy of neurodegenerative diseases such as prion and Parkinson’s disease.
Authors: Wagner J, Ryazanov S, Leonov A, Levin J, Shi S, Schmidt F, Prix C, Pan-Montojo F, Bertsch U, Mitteregger-Kretzschmar G, Geissen M, Eiden M, Leidel F, Hirschberger T, Deeg AA, Krauth JJ, Zinth W, Tavan P, Pilger J, Zweckstetter M, Frank T, Bähr M, Weishaupt JH, Uhr M, Urlaub H, Teichmann U, Samwer M, Bötzel K, Groschup M, Kretzschmar H, Griesinger C, Giese A.
Journal: Acta Neuropathol. 2013 Jun;125(6):795-813
PMID: 23604588 (This article is OPEN ACCESS if you would like to read it)
In this first study the researchers discovered Anle138b by conducted a large screening study to identify for molecules that could inhibit the toxic form of alpha synuclein.
They next tested Anle138b in both cell culture and rodent models of Parkinson’s and found it to be neuroprotective and very good at inhibiting the toxic form of alpha synuclein. And the treatment looks to be very effective. In the image below you can see dark staining of toxic alpha synuclein in the left panel from the brain of an untreated mouse, but very little staining in the right panel from an Anle138b treated mouse.

Toxic form of alpha synuclein (dark staining). Source: Max-Planck
Importantly, Anle138b does not interfere with the production and normal behaviour of alpha synuclein protein in the mice, but it does act as an inhibitor of alpha synuclein aggregation (which is considered to be the toxic form of the protein). In addition, the investigators found no toxic effects of Anle138b in any of their experiments even after long-term high-dose treatment (more than one year).
And in a follow up study, the drug was effective even if it was given after the disease model had started:

Title: The oligomer modulator anle138b inhibits disease progression in a Parkinson mouse model even with treatment started after disease onset
Authors: Levin J, Schmidt F, Boehm C, Prix C, Bötzel K, Ryazanov S, Leonov A, Griesinger C, Giese A.
Journal: Acta Neuropathol. 2014 May;127(5):779-80.
PMID: 24615514 (This article is OPEN ACCESS if you would like to read it)
During the first study, the researchers had started Anle138b treatment in the mouse model of Parkinson’s at a very young age. In this study, however, the investigators used genetically engineered mice which produce high levels of a mutant version of the human alpha synuclein protein (A30-P), and began treatment only as the symptoms were starting to show. These mice begin to display Parkinson’s-like symptoms at approximately 300 days of age. The researchers began treating the mice at 350 days of age and they found that Anle138b was significantly improved the overall survival of the mice.
More recently, the researchers followed up this research by investigating Anle138b in a mouse model of MSA:
Title: Anle138b modulates α-synuclein oligomerization and prevents motor decline and neurodegeneration in a mouse model of multiple system atrophy.
Authors: Heras-Garvin A, Weckbecker D, Ryazanov S, Leonov A, Griesinger C, Giese A, Wenning GK, Stefanova N.
Journal: Mov Disord. 2019 Feb;34(2):255-263. doi: 10.1002/mds.27562. Epub 2018 Nov 19.
PMID: 30452793 (This report is OPEN ACCESS if you would like to read it)
The PLP‐hαSyn mouse model recapitulates many of the clinical and pathophysiological features of MSA, and the investigators who conducted this study used these mice to test the potential of Anle138b as a treatment against MSA. The researchers found that treating the mice for 4 months with Anle138b significantly reduced alpha synuclein accumulation, resulting in neuroprotection and a reduction of immune system activation in the mice (compared to placebo treated animals). Anle138b treatment also helped to preserve motor function in the mice.
And these results have given MODAG confidence in moving to clinical trials of Anle138b in humans.
What about Parkinson’s?
Very recently a research group at Cambridge university has demonstrated the potential of Anle138b in a new mouse model of Parkinson’s:
 Title: Depopulation of dense α-synuclein aggregates is associated with rescue of dopamine neuron dysfunction and death in a new Parkinson’s disease model.
Title: Depopulation of dense α-synuclein aggregates is associated with rescue of dopamine neuron dysfunction and death in a new Parkinson’s disease model.
Authors: Wegrzynowicz M, Bar-On D, Calo’ L, Anichtchik O, Iovino M, Xia J, Ryazanov S, Leonov A, Giese A, Dalley JW, Griesinger C, Ashery U, Spillantini MG.
Journal: Acta Neuropathol. 2019 May 31. doi: 10.1007/s00401-019-02023-x.
PMID: 31165254
In this study, the investigators generated a new mouse model of Parkinson’s (called MI2), which produces high levels of the human alpha synuclein protein which is particularly aggregation-prone (truncated 1–120 α-syn). And they engineered these mice so that this protein was only produce in the dopamine neurons of the brain.
These mice displayed a progressive reduction of dopamine levels and significant dopamine cell death starting from 6 and 12 months of age, respectively. The animals began displaying the first signs of motor issues from about 9 months of age and gross motor probelms at 20 months of age (when 50% of dopamine neurons had been lost). This mouse model appears to resemble the human condition very well.
Remarkably, treatment with Anle138b from 9 to 12 months of age restored dopamine levels, prevented dopamine cell death and improved the motor impairments in this mice. Despite the delayed treatment, Anle138b was still able to have a beneficial effect in these mice. This finding validates and expands of similar previous studies involving delayed treatment onset with Anle138b (Click here to read more about this).
This last project has been supported by the Cure Parkinson’s Trust, Parkinson’s UK, and the Michael J Fox Foundation, and it will be interesting to see if after the Phase I clinical trial of Anle138b is complete if these organisations encourage MODAG to conduct a Phase II study in folks with Parkinson’s.
So what does it all mean?
News that the German biotech firm MODAG has come out of stealth mode and is now initiating a clinical trial programme for their lead compound – the anti-protein aggregation drug Anle138b – is very encouraging. This compound has been prudently developed and the company is now confident that Anle138b is ‘ready for primetime’.
Here at the SoPD, we will be keeping a close eye on how things go with regards to the clinical development of this drug, and if determined to be safe in humans after Phase I testing, we will be keen to see this compound tested further in different Parkinsonisms.
The banner for today’s post was sourced from


The great thing about Ante138b is a new drug which can be patented. There are a few other potent A-syn inhibitors of aggregation, but those drugs are either an old cheap generic or natural substance like Baicalein. These drugs have zero commercial value. Huge development that finally an anti a-syn inhibitor of aggregation with potential to make billions and in process attack the progression of PD and Lewy body dementia.
LikeLike
Hi Alan,
Thanks for your positive comment. This is an exciting development.
Kind regards,
Simon
LikeLike
Simon
You said that “Currently there is no test that can definitively make or confirm the diagnosis of MSA in a living person” I was under the impression that MRI could reveal a charcteristic ‘hot cross bun’ pattern for MSA and that PET scans can be used for PSP. Are these not reliable tests?
Keith
LikeLike
Hi Keith,
I hope all is well. Thanks for the interesting question. They are not considered diagnostic tests, rather diagnostic aids. And there has been some recent research questioning the sensitivity of these imaging ‘biomarkers’:
https://www.ncbi.nlm.nih.gov/pubmed/22488922
Kind regards,
Simon
LikeLike