|
# # # # Researchers in California have been conducting a different kind of Parkinson’s clinical trial. Rather than testing a drug or a special diet/exercise regime, they have been giving participants in their study a regular infusion of plasma. If you remove all of the cells from blood, the yellowish liquid that remains is called plasma. In medicine, plasma is usually used to boost a patient’s blood volume to help reduce shock. But recently researchers have been experimenting with giving older individuals infusions of plasma collected from young individuals to see if this has any beneficial effects. A group of researchers at Stanford University have been leading a study examining the safety of infusions of plasma (collected from young people) in a cohort of individuals with Parkinson’s. This week they published the results of their study. In today’s post, we will discuss what plasma is made of, why young plasma may help in neurodegenerative conditions, and review the results of the new study. # # # # |
 Source: KhanAcademy
Source: KhanAcademy
There are three chief components of blood:
- Red blood cells
- White blood cells
- Plasma
Red blood cells carry oxygen to distant parts of the body and they also remove carbon dioxide. And by volume, the red blood cells constitute about 45% of whole blood. White blood cells are the immune cells, fighting off infections etc. And they – this may surprise you – make up only 0.7% of whole blood.
That might sound like a tiny fraction, but understand that within a single drop of blood (50 ul) there are approximately 5 million red blood cells, and 5,000 to 25,000 white blood cells.
 Apologies to the squeamish. Source: Science
Apologies to the squeamish. Source: Science
And in total the human body contains about 4.5 litres (or 1.2 gallons) of blood. That’s a whole lot of drops. Plenty of white blood cells to help keep us healthy.
And what about plasma?
Plasma is the stuff that all of the red and white blood cells sit in. It has a yellowish tinge to it, and it makes up the other 54.3% of whole blood.
It contains 92% water and 8% ‘other stuff’.
Apologies for the very technical term (‘other stuff’), but there is a great deal of interesting stuff in that ‘other stuff’.
What do you mean ‘interesting’?
Interesting is a word I over use on this blog (Hi Chris), but plasma has garnered huge interest from the medical research world in recent years.
It contains all kinds of blood clotting proteins, waste, nutrients, dissolved salts (electrolytes), etc. But there are also a lot of beneficial factors in that ‘other stuff’.
Some of which may be useful in conditions like Parkinson’s.
What do you mean?
Back in 2005, some researchers published this report:
 Title: Rejuvenation of aged progenitor cells by exposure to a young systemic environment
Title: Rejuvenation of aged progenitor cells by exposure to a young systemic environment
Authors: Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA.
Journal: Nature. 2005 Feb 17;433(7027):760-4.
PMID: 15716955
In this study, the scientists wanted to examine the influence of the ‘other stuff’ floating around in the plasma on dividing cells in various tissues of aged mouse. They were seeking to test if ‘other stuff’ in blood from young mice could improve the function of the dividing cells in older mice.
How did they test this? By injecting young plasma?
Uh, no. They did it using a slightly different approach: They established parabiotic pairings of mice.
What are parabiotic pairings?
Ok, before we go any further – if you are even remotely squimmish, please skip this section and go down to recap #1.
Parabiotic pairing of mice is a technique used in scientific research to evaluate the effect of a shared blood system. That is to say, this approach requires the surgical joining of two mice (each with different conditions – eg. fat mouse and lean mouse) and then they co-habitate with a combined blood system for a given period of time to determine if one condition can influence another – see image below:
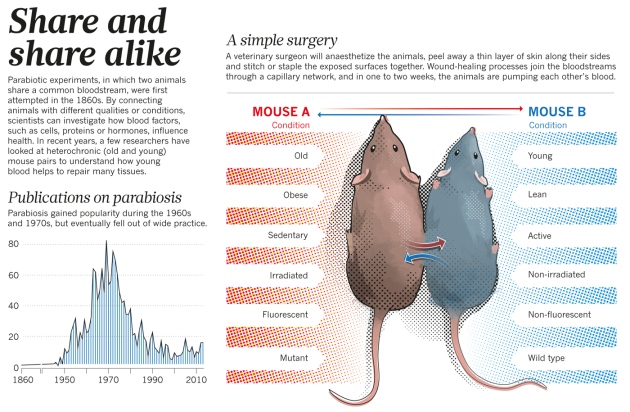 Source: Nature
Source: Nature
For a good OPEN ACCESS historical review of parabiotic pairing research – Click here.
In the current study, the researchers shared the circulatory system of young and old mice (this is called a heterochronic parabiosis) for 5 weeks. This combination exposed the old mice to factors that are present in blood (including all the ‘other stuff’ in the plasma) of young mice, and allowed the researchers to determine if this exposure benefited the older mice.
Eeekkk! Sounds awful… But what did they find?
The researchers found evidence of improved rejuvenation in certain cell types that they analysed. For example, they noted the restoration of specific biological pathways (such as Notch signalling) as well as the proliferative capacity in the satellite cells of the older mice. Satellite cells are muscle stem cells, they are the precursors to skeletal muscle cells.
The researchers also analysed different cell types in the liver and noted similar rejuvenation results.
Given these results, the researchers concluded that “the age-related decline of progenitor cell activity can be modulated by systemic factors that change with age“.
And subsequent studies have found similar results – they also demonstrated the reverse effect: Young animals exposed to older systemic factors exhibited reduced rejuvenative properties (Click here and here to read more about this).
What about the brain?
This question has been addressed in similar studies to the one described above – and the results have been published in multiple reports, such as this one:
 Title: The ageing systemic milieu negatively regulates neurogenesis and cognitive function.
Title: The ageing systemic milieu negatively regulates neurogenesis and cognitive function.
Authors: Villeda SA, Luo J, Mosher KI, Zou B, Britschgi M, Bieri G, Stan TM, Fainberg N, Ding Z, Eggel A, Lucin KM, Czirr E, Park JS, Couillard-Després S, Aigner L, Li G, Peskind ER, Kaye JA, Quinn JF, Galasko DR, Xie XS, Rando TA, Wyss-Coray T.
Journal: Nature. 2011 Aug 31;477(7362):90-4. doi: 10.1038/nature10357.
PMID: 21886162 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers used the heterochronic parabiosis model of young and old mice to determine if young blood could counteract/reverse the decline in neurogenesis (the turn-over of new brain cells) which is associated with normal aging.
Neurogenesis in the adult human and rodent brain primarily isolated to two regions: the subventricular zone of the striatum and the subgranular zone of the hippocampus (see the green regions in the image below).
 Neurogenesis. Source: Development
Neurogenesis. Source: Development
As we age, these regions exhibit less and less neurogenesis.
The investigators who conducted this study found that blood-based factors (that ‘other stuff’ we were talking about above) can inhibit or promote adult neurogenesis – in an age-dependent fashion (in mice at least). Older mice who were exposed to the young mouse blood system exhibited signs of increased neurogenesis.
And again, the reverse effect was also reported: by exposing young mice to the old systemic environment (or to the ‘other stuff’ in the plasma from old mice), the researchers reported decreased neuronal activity (using electrophysiology) and reduced performance on contextual fear conditioning and spatial memory tests.
|
# RECAP #1: Plasma is the portion of blood that does not involve cells. It makes up about half of the volume of blood, and contains many proteins that may have beneficial effects. Researchers have demonstrated that plasma from young mice can improve the functioning of certain cell types in older mice. # |
Interesting. What about conditions like Parkinson’s and Alzheimer’s? Can young plasma improve neurodegenerative conditions?
Great question. And the research community were fast to jump on this idea. Firstly, they wanted to look at whether young plasma could improve cognitive function, which led to reports like this one:
 Title: Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice.
Title: Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice.
Authors: Villeda SA, Plambeck KE, Middeldorp J, Castellano JM, Mosher KI, Luo J, Smith LK, Bieri G, Lin K, Berdnik D, Wabl R, Udeochu J, Wheatley EG, Zou B, Simmons DA, Xie XS, Longo FM, Wyss-Coray T.
Journal: Nat Med. 2014 Jun;20(6):659-63.
PMID: 24793238 (This report is OPEN ACCESS if you would like to read it)
In this study, after five weeks of sharing a circulatory system with young mice, the older mice exhibited evidence of increased connections between mature neurons in a region of the brain called the hippocampus, as well as increased synaptic plasticity (neuronal activity as measured by electrophysiology) in that region.
On the cognitive level, the researchers also demonstrated that systemic administration of blood plasma (collected from young mice) into aged mice not only improved the age-related impairments in a fear conditioning test, but also in a spatial learning/memory assessment.
The researchers concluded that their “data demonstrate that exposure to young blood counteracts aging at the molecular, structural, functional and cognitive levels in the aged hippocampus”.
Given all of these intriguing results, the researchers involved with this study then shifted their focus to test young plasma in models of Alzheimer’s.
And this report was published in 2016:
 Title: Preclinical Assessment of Young Blood Plasma for Alzheimer Disease.
Title: Preclinical Assessment of Young Blood Plasma for Alzheimer Disease.
Authors: Middeldorp J, Lehallier B, Villeda SA, Miedema SS, Evans E, Czirr E, Zhang H, Luo J, Stan T, Mosher KI, Masliah E, Wyss-Coray T.
Journal: JAMA Neurol. 2016 Nov 1;73(11):1325-1333.
PMID: 27598869 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers used a genetically engineered mouse that produces a mutated versions of the Alzheimer’s-associated protein amyloid precursor protein (two mutations: hAPP751 cDNA containing the London (V717I) and Swedish (K670M/N671L) variants – Click here to read more about these mice). In these mice, by 5-6 months, the characteristic hallmarks of Alzheimer’s – beta amyloid-containing amyloid plaques – were present in various regions of the brain.
Parabiotic pairings with young mice were set up at middle age (10 months of age) and later age (16 months of age). After 5 weeks exposure to a young blood system, the researchers found that amyloid plaques were not affected by the treatment (they looked the same between groups), nor was the microglial activation (the resident immune cells in the brain) affected.
But what the researchers did note was “a near complete restoration in levels of synaptic and neuronal proteins after exposure to young blood“. And this effect was associated with improved performance in a fear conditioning test and a spatial learning/memory assessment.
The conclusions of this study was that “Factors in young blood have the potential to ameliorate disease in a model of Alzheimer’s”.
Interesting, but how can this be tested in humans?
Researchers have conducted these types of studies in humans, but it hasn’t involved… um surgically attaching young and old people together.
Rather, the studies have involved collecting plasma from young individuals (typically university students, and ideally not after a hard night at the pub!).
 I’m giving blood in the morning. Source: HuffingtonPost
I’m giving blood in the morning. Source: HuffingtonPost
These samples of plasma can then be infused into older individuals who are taking part in the studies.
And this has already been done?
Yep. For example, there was a clinical study in Alzheimer’s involving young plasma infusion which reported last year:
 Title: Safety, Tolerability, and Feasibility of Young Plasma Infusion in the Plasma for Alzheimer Symptom Amelioration Study: A Randomized Clinical Trial.
Title: Safety, Tolerability, and Feasibility of Young Plasma Infusion in the Plasma for Alzheimer Symptom Amelioration Study: A Randomized Clinical Trial.
Authors: Sha SJ, Deutsch GK, Tian L, Richardson K, Coburn M, Gaudioso JL, Marcal T, Solomon E, Boumis A, Bet A, Mennes M, van Oort E, Beckmann CF, Braithwaite SP, Jackson S, Nikolich K, Stephens D, Kerchner GA, Wyss-Coray T.
Journal: JAMA Neurol. 2019 Jan 1;76(1):35-40.
PMID: 30383097 (This report is OPEN ACCESS if you would like to read it)
This study was designed to test the safety, tolerability, and feasibility of young plasma infusions in individuals with mild to moderate Alzheimer’s. It was a small randomised clinical trial, consisting of a double-blind crossover group of 9 patients and also an open-label group of 9 patients. The treatment involved a weekly infusion of young plasma for each of 4 weeks.
The researchers found that the treatment was safe and well tolerated and they suggested that young plasma infusion warrants further investigation in a larger clinical study to assess potential efficacy.
|
# # RECAP #2: Exposure to young plasma has had beneficial effects in models of Alzheimer’s, improving not only performance on cognitive tests but also increasing the number of synapses in various regions of the brain. These results have led to clinical trials exploring young plasma infusion which have demonstrated that this treatment approach is safe and well tolerated. Larger studies are now required to determine if plasma infusions can have any benefits in individuals with Alzheimer’s. # # |
Has plasma infusion ever been tested in the context of Parkinson’s?
As far as I’m aware, there have NOT been any preclinical models exploring parabiotic pairings in Parkinson’s research (please correct me if I am wrong here). And I am not aware of any plasma infusion work in preclinical models.
But there has been a clinical study assessing the safety, tolerability, and feasibility of young plasma infusions in people with Parkinson’s. And this week the results of that study were published:
 Title: Safety of Plasma Infusions in Parkinson’s Disease.
Title: Safety of Plasma Infusions in Parkinson’s Disease.
Authors: Parker JE, Martinez A, Deutsch GK, Prabhakar V, Lising M, Kapphahn KI, Anidi CM, Neuville R, Coburn M, Shah N, Bronte-Stewart HM.
Journal: Mov Disord. 2020 Jul 7. Online ahead of print.
PMID: 32633860 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers wanted to explore the safety and tolerability of young frozen plasma in people with Parkinson’s. 15 participants (including 5 women) completed the study – they had an average age of 63 years, and a disease duration of 8 years. The participants received 1 unit of young fresh frozen plasma twice a week for 4 weeks, and they were followed up for 4 weeks after the infusions ended.
The results suggest that not only was the treatment safe and well tolerated, but elevated baseline measures of inflammation (such as blood levels of tumor necrosis factor‐α) were decreased 4 weeks after the infusions ended.
The study was too small and short to provide any meaningful measures of efficacy, but the investigators still made some exploratory clinical assessments. There was no significant change detected by blinded rater video assessment of Parkinson’s motor score (using the UPDRS III score) – but as I say: small, short study.
The researchers conducting the sutdy suggested that the results “warrant further therapeutic investigations in PD and provide safety and feasibility data for plasma therapy in people with PD who may be at higher risk for severe complications of COVID‐19“.
EDITOR’S NOTE HERE: Please note that the researchers are not suggesting that folks with PD are at a higher risk, but rather that it is useful to know that plasma infusion can be safely used.
In addition to this study in a Parkinson’s cohort, another clinical study exploring plasma infusion in people with Progressive Supranuclear Palsy (or PSP) – a condition simiar to Parkinson’s – has been conducted. This was a 6 month Phase 1 study involving 5 individuals with PSP who received once per month infusions of young plasma (Click here to read more about this).
 Title: Open-Label Phase 1 Futility Studies of Salsalate and Young Plasma in Progressive Supranuclear Palsy.
Title: Open-Label Phase 1 Futility Studies of Salsalate and Young Plasma in Progressive Supranuclear Palsy.
Authors: VandeVrede L, Dale ML, Fields S, Frank M, Hare E, Heuer HW, Keith K, Koestler M, Ljubenkov PA, McDermott D, Ohanesian N, Richards J, Rojas JC, Thijssen EH, Walsh C, Wang P, Wolf A, Quinn JF, Tsai R, Boxer AL.
Journal: Mov Disord Clin Pract. 2020 Apr 10;7(4):440-447.
PMID: 32373661
The results of this study – like the Alzheimer’s and Parkinson’s studies – suggest that the treatment is safe and well tolerated. And while this was a very small open label study, the clinical results collected during this study suggest that “young plasma had a detectable effect on disease progression in PSP“. But given the size of this cohort (n=5) it is difficult to interpret this result.
It is apparent that plasma infusion is safe and tolerable, but larger, double-blind, placebo controlled trials may be required to determine any potential efficacy. Before then, however, it might be useful to see some preclinical young plasma infusion studies to assess if further clinical work is justified for PD.
|
# # # RECAP #3: The recently published results of a clinical trial evaluating young plasma infusion in individuals with Parkinson’s found the treatment to be well tolerated and safe, with some additional signs of beneficial effects. Another, long study in people with Progressive Supranuclear Palsy (or PSP) – a condition simiar to Parkinson’s – also found the method to be safe and tolerable. # # # |
So the usual conclusion: More research required? This stuff is years away from being clinically approved?
For plasma infusions? Yes.
But there are alternatives which might be closer to being potentially clinically approved.
Such as?
A California-based biotech company called Alkahest has taken a slightly different approach to the idea of young plasma research.  Alkahest have been developing treatments that focus on ‘plasma fractions’ (basically specific parts of the ‘other stuff’ in plasma). They have attempted to define the specific components of plasma that are having the beneficial effects and pooling them together into treatments.
Alkahest have been developing treatments that focus on ‘plasma fractions’ (basically specific parts of the ‘other stuff’ in plasma). They have attempted to define the specific components of plasma that are having the beneficial effects and pooling them together into treatments.
What are these treatments?
There are currently two treatments that are being clinically tested: GRF6021 and GRF6019?
Both of these experimental therapies are made up of approximately 400 proteins (source), and they have been developed by Alkahest and manufactured in collaboration with Grifols.
 Grifols is a Spanish pharmaceutical manufacturer, which is one of the world’s largest producers of blood plasma-based products (a lot of experinence/expertise in this field!).
Grifols is a Spanish pharmaceutical manufacturer, which is one of the world’s largest producers of blood plasma-based products (a lot of experinence/expertise in this field!).
GRF6021 is being clinically tested in Parkinson’s, while GRF6019 is being evaluated in two clinical trials for mild and late-stage Alzheimer’s (Click here and here to read more about those trials).
What do we know about the Parkinson’s trial?
In December 2018, Alkahest announced that they had dosed the first participant in a Phase II clinical trial of their product GRF6021 in people with Parkinson’s and cognitive impairments (Click here for the press release). This study is being supported by the Michael J Fox Foundation.
The study is a randomised, double-blind, placebo-controlled Phase II study in 90 people with Parkinson’s and cognitive impairment. It will be assessing the safety and tolerability of GRF6021 over a period of 7 months. The treatment (or placebo) is being administered by intravenous infusion for 5 consecutive days at Week 1 and Week 13 of the study.
Safety and tolerability is the primary end point, but change from baseline in the MDS-UPDRS score is a secondary end point so we will hopefully get a measure of whether the treatment is having any effect (Click here to read more about the details of this clinical study). The trial is being conducted across six U.S. research centers and is scheduled to finish in November 2020.
We look forward to seeing those results.
So what does it all mean?
COVID is demonstrating that as we age our bodies become weaker in their ability to fight off infection and disease. For the last decade, researchers have been exploring the cell-less portion of blood (plasma) to determine whether components of this liquid could have beneficial effects not only on normal aging, but also on diseases associated with aging (like Parkinson’s and Alzheimer’s).
Preclinical experiments suggest that components of young blood may rejuvenate old tissues, and clinical trials evaluating plasma infusion in humans with neurodegenerative conditions indicate that this treatment is safe and tolerable.
More sophisticated approaches are now being applied to this field of research, with biotech firms establishing exactly which fractions of plasma may be having the potential rejuvenating effects. Hopefully later this year we will learn from clinical trial results if their efforts are bearing fruit.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from Inverse


Are there issue s around not introducing plasma products from older people into younger ones?
LikeLike
Hi Eirwen,
I hope all is well – thanks for the interesting question. I guess it should be looked at, huh? Especially in the context of COVID! If ever I find myself in hospital in shock I will be asking to see the menu, and checking the vintages. Intriguing thought.
Kind regards,
Simon
LikeLike