|
# # # # Wearable technology offers the potential to more accurately monitor the symptoms of Parkinson’s in real time. Such information could allow for better and more precise management of the condition, as well as providing objective measures for clinical trials exploring novel therapies. Assessing some of the features of Parkinson’s, however, is not easy. Differentiating jerky involuntary movements like tremor or dyskinesias from planned movements like typing or shaking someone’s hand has proven difficult Recently, researchers at the tech giant Apple have been applying some focus to this problem and they are now sharing their results with the Parkinson’s community. In today’s post, we will review a research report presenting the results of the Apple study and discuss other recent events in wearable tech for Parkinson’s. # # # # |
 Source: Tes
Source: Tes
I used to be an Apple fan back in the day (mid-late 2000s). Wonderful user interface, superb design, lovely innovative products.
But I have to admit: gradually over time I became disenchanted with them.
Why?
The products became too expensive, the “Walled garden” mentality around the operating system frustrated me, and there has been a lack of serious innovation (a new iteration on a phone or tablet every year just doesn’t cut it… and now they are thinking of getting into the crowded space of electric cars… yippee, inspiring stuff).
Maybe we came to expect too much from them, but (personal opinion here) I think they lost their fanatical drive in the absence of Steve.
 Source: Dansilvestre
Source: Dansilvestre
[Positive way to start a post on, huh? It gets better. Stay with me]
All of that said, Apple published a research report earlier this year that deserves the Parkinson’s community’s attention and respect.
What did they report?
In February, researchers from Apple (and a group of academic collaborators) published this paper:
 Title: Smartwatch inertial sensors continuously monitor real-world motor fluctuations in Parkinson’s disease.
Title: Smartwatch inertial sensors continuously monitor real-world motor fluctuations in Parkinson’s disease.
Authors: Powers R, Etezadi-Amoli M, Arnold EM, Kianian S, Mance I, Gibiansky M, Trietsch D, Alvarado AS, Kretlow JD, Herrington TM, Brillman S, Huang N, Lin PT, Pham HA, Ullal AV.
Journal: Sci Transl Med. 2021 Feb 3;13(579):eabd7865.
PMID: 33536284 (An OPEN ACCESS preprint of this report is available by clicking here)
In this study, the researchers presented the Motor fluctuations Monitor for Parkinson’s Disease (or MM4PD) system, which is a real world monitoring system for Parkinson’s that incorporates data from the inertial sensors built into the Apple smartwatch. Their goal was to continuously track fluctuations in specific motor symptoms over time.
 Source: Wareable
Source: Wareable
Why did they want to do that?
Real-world remote monitoring of motor symptoms over time in Parkinson’s is worthy goal. It could provide an objective, more accurate measure of disease progression which would greatly help clinicians (and patients) to better manage the treatment of the condition. At present, yearly visits to a neurologist are the gold standard of available care, but this provides a very limited glimpse into the specific characteristics of individual cases. More continuous, at home measures of symptoms would provide a more granular view of treatment effectiveness, providing better quality of life.
This monitoring is particularly relevant to aspects of the condition that are generally poorly self-assessed, and here I am thinking of levodopa-induced dyskinesias. Previous research has reported that patient recall of dyskinesias is often error-prone:
 Title: Parkinsonian patients and poor awareness of dyskinesias.
Title: Parkinsonian patients and poor awareness of dyskinesias.
Authors: Pietracupa S, Latorre A, Berardelli A, Fabbrini G.
Journal: Front Neurol. 2014 Mar 20;5:32.
PMID: 24688482 (This report is OPEN ACCESS if you would like to read it)
More continuous monitoring of symptoms would also improve clinical trial efforts that are seeking to demonstrate a slowing of disease course, by offering more specific measures. Rather than requiring participants to come into research centers for periodic clinical evaluations (often off medication), methods of continuous assessment could provide more detailed data and a richer dataset.
How can we do this?
Technology has offered many different approaches. We have previously discussed methods like smart phone-based apps (Click here to read an old SoPD post on this topic). My only concern with the smart phone approach is that it requires effort from the participant being assessed. A better approach would be to have a technology that the individual wears, and completely forgets about. This way there is no conscious bias creeping into the collected data.
Smart ring technology fits this requirement (Click here to read a previous SoPD post on this topic). Most of us who are married rarely think of the ring on our finger (terrible thing to admit to, but it’s true).
 Source: Soukie
Source: Soukie
And this is wear smart watch technology could be useful. Large portions of the population wear watches and are only sporadically aware of them when they need to know the time.
Smart watch technology is already being used by a wide section of the general public as a tool for continuous monitoring of activity and fitness. Keeping track of number of steps, distance travelled, heart rate, etc.
 Source: Forbes
Source: Forbes
So naturally scientists have been exploring potential applications for smart watch technology in their respective fields of medical research. And recently researchers at the tech giant Apple have presented their efforts in this arena.
|
# RECAP #1: Wearable technology could provide the opportunity to better monitor Parkinson’s symptoms. This would allow for better management of treatment, and as a result better quality of life. Numerous approaches to wearable technology are available, and recently the tech giant Apple published a report on the use of their Smart watch for tracking Parkinson’s symptoms over time. # |
So what did the Apple researchers report?
The Apple Watch was first released on the 24th April 2015. It was quickly adopted by consumers, and became one of the best-selling wearable device with more than 100 million people using an Apple Watch by December 2020 (Source).
 Source: Apple
Source: Apple
During that time, the company has been developing a lot of apps specifically designed around the Apple Health initiative, some of which have been focused on acting as tools for clinical settings.
As part of this programme, they have developed the Motor fluctuations Monitor for Parkinson’s Disease (or MM4PD) system, to continuously track changes in resting tremor and dyskinesia over the course of hours, days, weeks and months.
What exactly do we mean by tremor?
There are many different types of tremor but only a few of them apply to Parkinson’s. In 1998, the Movement Disorder Society published a classification scheme that categorizes three types of tremor associated with Parkinson’s:
 Title: Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee.
Title: Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee.
Authors: Deuschl G, Bain P, Brin M.
Journal: Mov Disord 1998;13 Suppl 3:2-23.
PMID: 9827589
Those three types of tremor are:
1. A resting tremor with the same frequency – this tremor is the most common and it is usually inhibited during movement.
2. A resting tremor of different frequencies – this form of tremor occurs in less than 10% of people with Parkinson’s.
3. A postural tremor with a frequency varying between 4 and 9 times per second. These types of tremor are rare in the overall Parkinson’s population, but are common in the very rigid version of Parkinson’s.
And what are dykinesias?
Dyskinesias (from Greek: dys – abnormal; and kinēsis – motion, movement) are a category of movement disorders that are characterised by involuntary muscle movements. And they are certainly not specific to Parkinson’s.
But in the case of Parkinson’s, dyskinesias have generally been believed to be associated with long-term use of levodopa (also known as Sinemet or Madopar – levodopa with carbidopa and levodopa with benserazide, respectively).
 Sinemet is levodopa. Source: Drugs
Sinemet is levodopa. Source: Drugs
NOTE: Long-term use of levodopa is not a certainty for developing dyskinesias, but there is an association. It will differ from person to person.
As for how they develop, there is a lot of debate over this topic, but there are some basic details that researchers generally tend to agree on.
Before being diagnosed and beginning a course of levodopa, the locomotion parts of the brain in a person with Parkinson’s gradually becomes more and more inhibited. This increasing level of inhibition results in the slowness and difficulty in initiating movement that characterises this condition.
A person with Parkinson’s may want to move, but they can’t – they are inhibited. In effect, they are akinetic (from Greek: a-, not, without; and kinēsis – motion).
 Drawing of an akinetic individual with Parkinson’s, by Sir William Richard Gowers. Source: Wikipedia
Drawing of an akinetic individual with Parkinson’s, by Sir William Richard Gowers. Source: Wikipedia
Once inside the brain, levodopa is quickly converted into dopamine. It is changed into dopamine by an enzyme called DOPA decarboxylase, and this change rapidly increases the levels of dopamine in the brain, allowing the locomotion parts of the brain to function more normally.
The chemical conversion of L-DOPA to dopamine. Source: Nootrobox
In understanding this process, it is important to appreciate that when an levodopa tablet is consumed and levodopa enters the brain, there is a rapid increase in the levels of dopamine. This ‘spike’ in the supply of dopamine will last for the next few hours, before the dopamine is eventually used up.
As the effects of the levodopa tablet wear off, another tablet will be required. This use of multiple levodopa pills across the day gives rise to a wave-like shape to the dopamine levels in the brain over the course of the day (see the figure below). The first pill in the morning will quickly lift the levels of dopamine enough that the individual will no longer feel akinetic. This will allow them to be able to function with normal controlled movement for several hours before the levodopa begins to wear off. As the levodopa wears off, the dopamine levels in the brain drop back towards levels that will leave the person feeling akinetic and at this point another levodopa tablet is required.
 A hypothetical illustration of dopamine levels over a day
A hypothetical illustration of dopamine levels over a day
After several years of levodopa use, many people with Parkinson’s will experience a weaker response to each tablet. They will also find that they have more time during which they will be unable to move (exhibiting akinesia). This is simply the result of the slow progression of Parkinson’s – levodopa treats the motor features of the condition but only hides/masks the fact that the disease is still progressing.
To combat this shorter response time, the dose of levodopa is usually increased. This will result in increasing levels of dopamine in the brain (as illustrated by the higher wave form over time in the image below). Gradually it will take more levodopa medication-induced dopamine to lift the individual out of the akinetic state.
 Again this illustration is hypothetical (situation differs between individuals)
Again this illustration is hypothetical (situation differs between individuals)
This increasing of levodopa dosage, however, results in too much dopamine being present in the brain at times. And this situation is often associated with the gradual development of abnormal involuntary movements that appear when the levels of levodopa induced dopamine are the highest.
These are the involuntary muscle movements that we refer to as dyskinesias.
Got it. So the Apple researchers were trying to track tremor and dyskinesias over time using their Apple Watch. How did they do?
They validated their MM4PD system in 343 volunteers with Parkinson’s. This included two components: first, an in-clinic pilot study involving 118 individuals with PD. Up to 4 videoed session were conducted for each participant, and the smart watch performance was compared to assessments made by 3 movement disorder specialists.
Second, a longitudinal 6 month clinical study in a cohort of 225 participants with PD. The researcher did not restrict the test cohort to a particular subtype or level of severity of Parkinson’s. Instead they recruited a broad range of stages of Parkinson’s in order to really test the MM4PD algorithms.
The investigators also tested the smartwatch MM4PD system in 171 older volunteers in a longitudinal control study for up to 12 months. This extra group was to help the algorithms determine what was tremor or dyskinesia from normal general motor behaviour of elderly individuals.
In controlled settings, the researchers found that MM4PD was accurate and sensitive “in estimating symptom presence or severity“.
 Source: Researchgate
Source: Researchgate
Tremor was defined (based on displacement) as “slight” (<0.1 cm), “mild” (0.1-0.6 cm), “moderate” (0.6-2.2 cm), “strong” (>2.2 cm), “unknown”, and “absent”. These measures were taken during each one-minute interval the smartwatch was worn.
Tremor is very difficult for algorithms to distinguish from all the other movements our arms/wrists make on a daily basis as we go about our business.
But the MM4PD was very good at picking up tremor – matching clinician based assessments and also detecting fluctuations in tremor that corresponded with prescribed times for L-dopa treatment. Here is an example of one participant’s tremor measures by the MM4PD system during the longitudinal study:
 Source: Researchgate
Source: Researchgate
When the investigators assessed the median tremor false positive rate in over 43,300 hours of all-day data from the elderly, non-PD control group (n=171) in the longitudinal control study, they found it to be just 0.25%.
For dykinesias, the researchers developed the Choreiform Movement Score (CMS). This dyskinesia algorithm was validated across all of the 343 participants with Parkinson’s (61 with dyskinesia) involved in the study and using data from the 171 elderly, non-Parkinson’s control group.
The amount of dyskinesia detected by the MM4PD system differed significantly between individuals – demonstrating how difficult the task is – but (again) the false positive rate was just 2.0% in the elderly control group, indicating that the approach is viable.
And when individual cases are monitored over time, there are clear trends in the appearance of dyskinesias, such as “ON-OFF” fluctuations. In the image below, note the ability of the MM4PD system to differentiate between tremor (yellow/red coloured bars) and dyskinesias (blue bars):
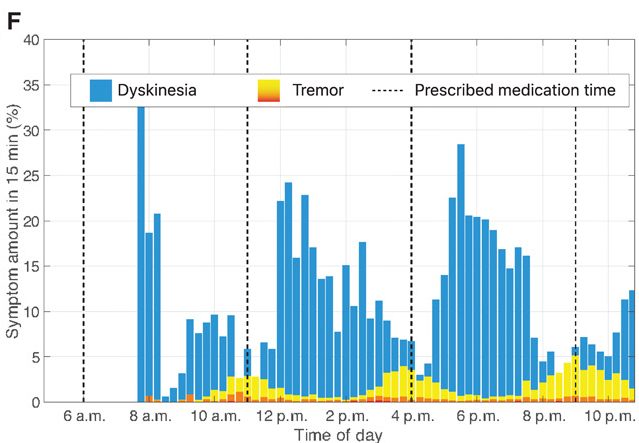 Source: Researchgate
Source: Researchgate
A couple of other remarkable notes in the report was that the MM4PD system was able to pick up changes in symptom profiles after individuals underwent surgery for deep brain stimulation, deep brain stimulation reprogramming, and “even as a subject became more adherent to their prescribed treatment plan“.
A rather remarkable achievement considering that the MM4PD system is simply converting motion sensor data captured by a wrist watch into resting tremor and dyskinesia scores.
The authors also noted that the approach is a “discreet, unobtrusive symptom monitoring without the stigma of a dedicated medical device or burden of active tasks“, which renders adherence issues obsolete (all the participant has to do is remember to wear their watch each day).
The researchers concluded that “MM4PD enables continuous symptom monitoring through a smartwatch, and can help clinicians and researchers incorporate out-of-clinic data for the treatment and study of Parkinson’s”.
|
# # RECAP #2: Researchers at the tech firm Apple have developed the “Motor Fluctuations Monitor for Parkinson’s Disease” (of MM4PD) system which was tested on their Apple Watch. The algorithms were able to convert motion sensor data captured by the wrist watch into resting tremor and dyskinesia scores, in both laboratory and real world settings. # # |
Is Apple the only big tech firm working on Parkinson’s?
No, there are a few developing various products.
But very recent developments could have implications for some of those efforts.
What do you mean?
Very recently Verily Life Sciences – which is Google/Alphabet Inc.’s research organization devoted to the study of life sciences – asked the US FDA to approve a Parkinson’s symptom assessment tool on its clinical research-focused smart watch – called “Study watch“. Verily was hoping that this would allow for their watch to be used as a data collection tool in drug development studies.
Verily had developed a Parkinson’s symptom assessment tool, called virtual motor exam (VME) for Parkinson’s disease Part III (VME Part III). The Part III refers to a component of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (or MDS-UPDRS) – which is the ‘gold standard’ clinical assessment tool used by clinicians in evaluating individuals with PD.
The current version of the MDS-UPDRS has four parts:
- I: Non-motor experiences of daily living
- II: Motor experiences of daily living/Activities of daily living
- III: Motor examination
- IV: Motor complications.
Verily had designed their software around Part III – the motor examination. The watch prompts its wearer once per week to do 8 quick motor assessments (such as a finger tapping test, stomping the foot, opening and closing the hand, and standing still).
 Verily study watch. Source: Wareable
Verily study watch. Source: Wareable
The company is also conducting a clinical study evaluating the VME Part III in 3 cohorts:
- 144 individuals with newly diagnosed with Parkinson’s
- 500 individuals with mild to moderate cases diagnosed within the last 5 years
- 105 individuals with moderate to severe cases diagnosed at least 5 years ago.
This study is being conducted in the Netherlands and Japan (Click here to read more about the details of this study), but Verily also plans to expand the study in the US, Canada, the UK and Europe by the end of this year. Participants wear the watch for 2 years, with clinical assessments included and involving the collection of biosamples.
It looks like a really thorough investigation of the natural history of Parkinson’s.
In a letter of intent sent to the FDA in January, Verily wrote that the VME Part III was designed to simplify the collection of data tracking the physical Parkinson’s symptoms. They wrote that “the exam takes less than 20 minutes”, adding that it could “be performed in the comfort of a person’s home, without the guidance of a clinician.”
But in their recently issued rejection letter, the FDA said that “the MDS-UPDRS Part III and the VME III are limited in their capacity to evaluate meaningful aspects of concepts of interest that are relevant to the patients’ ability to function in day-to-day life”.
In particular, the agency points out that the finger tapping test “in the MDS-UPDRS Part III cannot be directly interpreted as being meaningful to patients”. The FDA suggested that “a change in speech, eating and dressing (as assessed in the MDS-UPDRS Part II) represents meaningful change in how patients function in daily life”.
It is a fair point and denotes a shift by the agency towards more clinically meaningful endpoints, but where does it leave tech companies who have sunk significant resources into their devices/apps based on previous ‘gold standards’?
And it is also curious how a regulator can approve a drug one week for it’s “effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit to patients” (and, in the absence of data, broaden the cohort that can be prescribed said drug) and yet at the same time say no to a non-invasive wearable piece of technology based on a change in preference.
Loose standards on a drug that millions may be administered, but tight rules on a wrist watch…
I’m just saying.
How Verily will respond to the FDA is unknown outside of 269 E Grand Ave, South San Francisco, but they may have some help in the form of a FDA veteran: Dr Amy Abernethy, former principal deputy commissioner of food and drugs at the FDA was recently hired as president of Verily’s clinical research business.
Let’s hope they can rapidly transition their Parkinson’s programme to the regulators preference.
Are there other issues with adoption of smart watch tech in Parkinson’s?
The report from Apple reviewed in this post acknowledges that in addition to regulatory hurdles, there are other considerations that need to be addressed before wide spread adoption of smart watch-based technology. Specifically, they wrote “Integrating MM4PD into clinical
settings requires careful consideration to avoid further exacerbating socioeconomic disparities among patients with Parkinson’s”.
I thought this was a rather considerate consideration when considering all things to be considered.
So what does it all mean?
Having started on a negative note with my personal opinion about recent developments at the big tech firm Apple, I would like to finish by giving the company their props (hip-hop slang, derived from ‘propers’, both meaning “due respect”): Their Motor fluctuations Monitor for Parkinson’s Disease is a really impressive piece of work!
As we have discussed above, the measuring of tremor and (particularly) dyskinesias is extremely difficult. Consider the playing of a piano – how is an algorithm capable of blindly determining whether that is planned or involuntary movement simply from the motion of a wrist?
The Apple researchers get a solid salute from the SoPD team and we will look forward to seeing how they develop this technology further.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: Apple is a publicly traded company. The material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. Apple have not requested that this material be produced, nor has the author had any contact with the company or associated parties (except to ask for a copy of the report reviewed. This post has been produced for educational purposes only.
The banner for today’s post was sourced from biotechscope.



I may have missed it but are there any plans to make this available publicly? Perhaps as a beta?
LikeLike
What was meant by “avoid further exacerbating socioeconomic disparities among patients with Parkinson’s?” It sure wasn’t a consideration when selling a1$K cell phone. Why would you hold back an APP until you could get the government to subsidize it? Where is your heart Apple? I need this to manage my Parkinson’s and you are telling us it must have FDA approval, It is an APP! An APP you load on a watch. You aren’t concerned about socioeconomic impact, BS Apple is worried about charging as much as they can. Putting a government wrapper around it. Forcing us to pay more. That is the epitome of socioeconomic impact.
LikeLike