|
# # # # There is a critical need for good biomarkers in neurodegenerative research. A biomarker is an objective indicator of a medical state that can be assessed from outside a patient, and can be measured accurately and reproducibly. It could come in the form of a medical imaging application or a biological sample (such as a blood test). Recent research points towards a particular protein (referred to as GPNMB) that could be a potential biomarker for a specific subtype of Parkinson’s. In today’s post, we will review some of the research on this topic and consider how a biomarker could potentially be used in Parkinson’s research. # # # # |
 Source: TED
Source: TED
Back in 2016, some researchers reported something interesting.
They had been investigating cells collected from people with a condition called Gaucher disease (Pronounced: ‘Go-Shay’; don’t ask – we’ll discuss what it is in a moment, just let me get the intro out of the way). Specifically, the scientists were seeking potential biomarkers for Gaucher disease… and they might have found one.
Here is their report:
 Title: Elevation of glycoprotein nonmetastatic melanoma protein B in type 1 Gaucher disease patients and mouse models.
Title: Elevation of glycoprotein nonmetastatic melanoma protein B in type 1 Gaucher disease patients and mouse models.
Authors: Kramer G, Wegdam W, Donker-Koopman W, Ottenhoff R, Gaspar P, Verhoek M, Nelson J, Gabriel T, Kallemeijn W, Boot RG, Laman JD, Vissers JP, Cox T, Pavlova E, Moran MT, Aerts JM, van Eijk M.
Journal: FEBS Open Bio. 2016 Jul 30;6(9):902-13.
PMID: 27642553 (This report is OPEN ACCESS if you would like to read it)
In the study, the researchers collected cells from the spleen of an individual with Gaucher disease and looked for proteins in the cells that were higher than normal.
They found glycoprotein non-metastatic melanoma protein B (GPNMB, also known as osteoactivin) was ridiculously high. Off the charts high. They then compared GPNMB levels in blood samples collected from 59 people with Gaucher disease and 20 healthy controls. As you can see in the graph below, GPNMB levels were on average 25‐fold higher in all of the 59 people with Gaucher disease (Note: the Y axis is logarithmic):
 Source: PMC
Source: PMC
Interestingly, when the individuals with Gaucher disease started the standard treatment for the condition, the levels of GPNMB collectively dropped:
 Source: PMC
Source: PMC
And this result has been independently validated (Click here to read that report). The second study used a larger cohort of individuals with Gaucher disease (155 patients) and they found a >15-fold elevation of GPNMB in the blood of this group (compared to controls). And again these high levels were reduced when the Gaucher group started treatment:
Source: PMC
A third study found that GPNMB levels in the brains of a mouse model of Gaucher disease correlated with disease severity in the mice, and also reported elevated GPNMB levels in brain samples from patients with Gaucher disease.
All of the research groups concluded that their data supports the potential utility of GPNMB as a biomarker of Gaucher disease.
Great! But what is Gaucher disease and why is this on a website for Parkinson’s research?
Gaucher disease is a rare inherited genetic disorder characterised by the build up in cells of a large fatty molecule called glucocerebroside. The disorder results from the deficiency of the enzyme glucocerebrosidase, which usually breaks down glucocerebroside. Because the body cannot break down glucocerebroside, swollen fat-laden cells accumulate in certain areas of the body, such as the spleen, liver and bone marrow.
These cells are referred to as ‘Gaucher cells’.
 Swollen Gaucher cells (circled in red). Source: Imagebank
Swollen Gaucher cells (circled in red). Source: Imagebank
The incidence of Gaucher disease is about one in every 40,000 live births (Source), and the condition manifests itself in several way, from reduced bone density to swollen liver and spleen:
 The signs of Gaucher disease. Source: Rare2aware
The signs of Gaucher disease. Source: Rare2aware
What happens to the cells in Gaucher disease?
Macrophage are one type of cell that is particularly affected in Gaucher disease. They are a type of blood cell that is responsible for detecting, engulfing and destroying dangerous pathogens and apoptotic cells. Below is a schematic of a macrophage, consuming orange pathogens (left), digesting them, and releasing the waste (on the right):
 A schematic of a macrophage. Source: Meducator
A schematic of a macrophage. Source: Meducator
Macrophage travel the body, swallowing anything that they don’t like the look of. In order to break down everything it swallows, a macrophage must have a full complement of digestive enzymes. These enzymes are usually stored in the macrophage in small sacks known as lysosomes.
As you can see in the image below, without glucocerebrosidase, the macrophage has trouble digesting certain fatty molecules like glucocerebrosides and the lysosomes start to accumulate in the cell, causing the cell to swell up.
 Gaucher disease. Source: Gaucherawareness
Gaucher disease. Source: Gaucherawareness
Are there different types of Gaucher disease?
There are three types of Gaucher disease:
- Type I – (also called the “non-neuropathic” type) this is the most common; having said that it mainly occurs in Ashkenazi Jews (x100 more than the general population). The median age at diagnosis is 28 years of age, and life expectancy is only mildly decreased. As the “non-neuropathic” label suggests, there are no neurological symptoms.
- Type II – is characterised by neurological problems in small children. The glucocerebrosidase enzyme is barely present in the lysosomes. Prognosis is poor (death before the age of three).
- Type III – (the Swedish variety) occurs in people from the Norrbotten region in Sweden. This group develops the disease somewhat later, but most most do not survive their 30th birthday.
These subtypes of Gaucher disease are characterised by the presence or absence of neurological issues, and by the rate of disease progression and severity:
 Stages of Gaucher disease. Source: Gaucherawareness
Stages of Gaucher disease. Source: Gaucherawareness
You said Gaucher disease is inherited. So if one of my parents has Gaucher disease will it affect me?
As we mentioned above, Gaucher disease is an inherited genetic disorder. It is caused by mutations in the GBA gene (we discuss the genetics further down this post).
Humans normally have two copies of the GBA gene. If one copy of the GBA gene is faulty due to a genetic mutation, the person will not develop Gaucher’s disease, because the one remaining functional gene will be able to produce enough of the glucocerebrosidase enzyme.
Gaucher disease is considered an autosomal recessive disorder. This means that two copies of an abnormal gene must be present in the individual in order for the disease to develop. A person with just one faulty gene will not get sick, but they will be a carrier. To develop Gaucher disease, you need to have two genetic mutations in the GBA gene – one from your mother and one from your father.
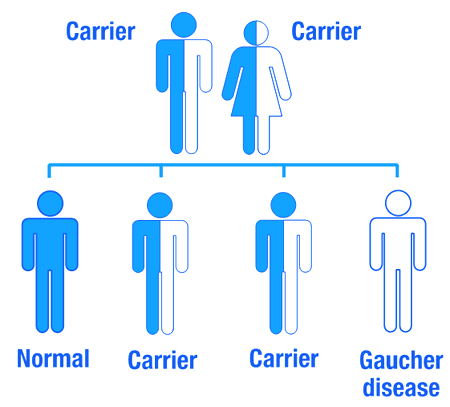 An autosomal recessive disorder. Source: Myhealthyfeeling
An autosomal recessive disorder. Source: Myhealthyfeeling
So even if one of your parents has been diagnosed with Gaucher disease, you will not necessarily develop it if the other parent does not have a mutation in their GBA gene.
|
# RECAP #1: Gaucher disease is an inherited genetic disorder characterised by the build up in cells of a large fatty molecule called glucocerebroside.. The disorder results from a deficiency of glucocerebrosidase, and there are three types of Gaucher disease. # |
Ok. All of this is interesting, but how is Gaucher disease associated with Parkinson’s?
In the 1990s, physicians began to notice a high incidence of patients with Gaucher displaying symptoms of Parkinson’s. An example of this was a report published in 1996 that described six people with Gaucher disease who also exhibited an early-onset, severe form of Parkinson’s with cognitive decline:
 Title: Occurrence of Parkinson’s syndrome in type I Gaucher disease.
Title: Occurrence of Parkinson’s syndrome in type I Gaucher disease.
Authors: Neudorfer O, Giladi N, Elstein D, Abrahamov A, Turezkite T, Aghai E, Reches A, Bembi B, Zimran A.
Journal: QJM. 1996 Sep;89(9):691-4.
PMID: 8917744 (This article is OPEN ACCESS if you would like to read it)
In this study, the Israeli researchers report on 6 people with Type I Gaucher disease (which up until that point had not been considered neuronopathic). All six of the subjects also exhibited the hallmark of a rather severe form of Parkinson’s, which made its appearance in the 4th to 6th decade of life and displayed an aggressive progression and was largely unresponsive to conventional anti-Parkinson therapy (such as L-dopa).
These initial reports were followed by many additional studies which eventually started pointing towards the GBA gene as the likely risk factor for this form of Parkinson’s, including this study:
 Title: Mutations in the glucocerebrosidase gene and Parkinson’s disease in Ashkenazi Jews.
Title: Mutations in the glucocerebrosidase gene and Parkinson’s disease in Ashkenazi Jews.
Authors: Aharon-Peretz J, Rosenbaum H, Gershoni-Baruch R.
Journal: N Engl J Med. 2004 Nov 4;351(19):1972-7.
PMID: 15525722 (This article is OPEN ACCESS if you would like to read it)
In this study, the investigators examined 99 individuals with idiopathic Parkinson’s from an Ashkenazi Jewish background. Thirty-one of them (31.3%) had one or two mutations in their GBA gene. And of all the individuals with Parkinson’s, the subjects who were carriers of GBA mutations were younger than those who were not carriers (mean age at onset being 60 years vs. 64 years).
And this result is similar to what has been seen in larger follow up studies (Click here for an example).
So genetic variations in the GBA gene are associated with Parkinson’s?
Yes. They are one of the main genetic risk factors for the condition.
It is now believed that approximately 5%–8% of people with Parkinson’s have a genetic mutation in the GBA gene (Click here and here to read more about this). According to the Michael J Fox foundation webpage on GBA “up to 10 percent of people with PD in the United States carry” a genetic variation in the GBA gene.
What do we know about the GBA gene?
GBA is a very large gene and there are numerous genetic variants spread across its length. The most common mutations are located in positions N370S and L444P. These mutations cause a reduction in the enzymatic activity of the glucocerebrosidase enzyme.
 The GBA gene and mutations associated with Parkinson’s. Source: Neurology
The GBA gene and mutations associated with Parkinson’s. Source: Neurology
And as I mentioned above generally, people with GBA-associated Parkinson’s will exhibit more progressive symptoms than people without a GBA mutation, and this has recently been confirmed in a large clinical observation study:
 Title: Features of GBA-associated Parkinson’s disease at presentation in the UK Tracking Parkinson’s study.
Title: Features of GBA-associated Parkinson’s disease at presentation in the UK Tracking Parkinson’s study.
Authors: Malek N, Weil RS, Bresner C, Lawton MA, Grosset KA, Tan M, Bajaj N, Barker RA, Burn DJ, Foltynie T, Hardy J, Wood NW, Ben-Shlomo Y, Williams NW, Grosset DG, Morris HR; PRoBaND clinical consortium.
Journal: J Neurol Neurosurg Psychiatry. 2018 [Epub ahead of print]
PMID: 29378790 (This article is OPEN ACCESS if you would like to read it)
In this multi-research centre study, the investigators studied 1893 people with Parkinson’s. Of these 171 (9.0%) had one copy of a known GBA mutation, of whom 121 (6.2%) had a genetic variant known to be associated with Parkinson’s but not causing Gaucher’s disease, and 50 (2.7%) had a genetic variant linked to both Parkinson’s and Gaucher’s. Another 28 (1.5%) carried genetic variants of unknown significance in the GBA gene. The most common GBA mutations associated with Parkinson’s in this study were ‘E362K‘, ‘T369M’ , and ‘L444P’.
Individuals with Parkinson’s associated GBA mutations were on average diagnosed 5 years earlier compared with non-carriers. They were also more likely to have postural instability and gait difficulties compared with non-carriers. In addition, they had more progressive forms of Parkinson’s (as determined by more advanced Hoehn and Yahr staging – after adjustment for age – compared with non-carriers).
No differences was observed in cognitive function between GBA mutation carriers and non-carriers. Cognitive impairment/dementia have been reported in other studies at a later stages of the condition (Click here to read more about this). This observation led the researchers to conclude that “this offers an important window of opportunity for potential disease-modifying therapy that may protect against the development of dementia”.
This is really bad, right? What if I have a GBA genetic mutation?
It is important for readers to understand that our understanding of the genetics of Parkinson’s is basic at best and that this aggressive pattern of disease progression in GBA-associated Parkinson’s is not always the case.
In addition, 2-3% of the general population will have a Parkinson’s associated GBA mutation in their DNA, but they never go on to present any of the features of Parkinson’s. And then there are cases of identical twins who both have a GBA mutations, but only one of them has developed Parkinson’s (Click here to read more about this).
Thus, the genetics of Parkinson’s is still very complex, and just because a person has a GBA mutation, it does not necessarily mean that they will go on to develop Parkinson’s or necessarily present the aggressive form of the condition.
More research is required to increase our understanding of all aspects of GCase and GBA.
|
# RECAP #2: Genetic errors (or variants) in a region of DNA called the GBA gene causes Gaucher disease. Some of these variants can also increase an individuals risk of developing Parkinson’s. GBA-associated Parkinson’s is characterised by a more rapid progression of the condition, and increased risk of cognitive complaints – but these features can vary from case to case. # |
Ok, so GPNMB could be a potential biomarkers for Gaucher disease, and Gaucher disease is associated with Parkinson’s. Could GPNMB be a biomarker for Parkinson’s?
It is a really interesting question.
One that researchers have previously investigated. In 2017, this report was published:
 Title: Increased brain expression of GPNMB is associated with genome wide significant risk for Parkinson’s disease on chromosome 7p15.3.
Title: Increased brain expression of GPNMB is associated with genome wide significant risk for Parkinson’s disease on chromosome 7p15.3.
Authors: Murthy MN, Blauwendraat C; UKBEC, Guelfi S; IPDGC, Hardy J, Lewis PA, Trabzuni D.
Journal: Neurogenetics. 2017 Jul;18(3):121-133.
PMID: 28391543 (This report is OPEN ACCESS if you would like to read it)
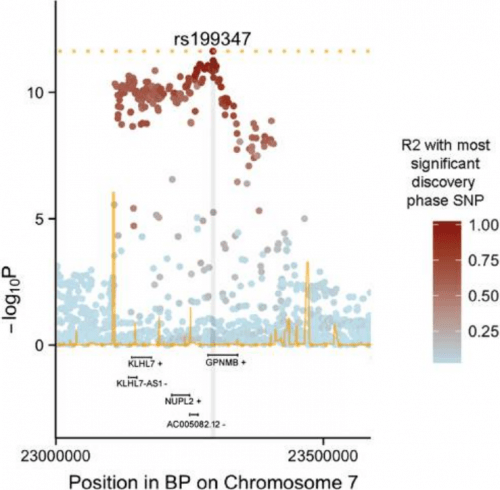
In this study, the researchers were interested in better understanding a region of DNA – tagged rs199347 – in which variants had previously been associated with increased risk of developing Parkinson’s (Click here to read more about that). In the image below, the dots represent single point variants in the DNA on a small stretch of chromosome 7. The brown colouration indicates an association with Parkinson’s:
Source: PMC
As you may also have noticed in the image above, there are five genes (functional sections of DNA) within the affected area of DNA. These genes are KLHL7, KLHL7-AS1, NUPL2, AC005082.12 and (surprise) GPNMB.
The researchers found that elevated levels of GPNMB in the brain underlies the association between Parkinson’s risk and the rs199347 region. They proposed that this could be “the causative link explaining the association of this locus with Parkinson’s”.
And this association has very recently been replicated:
 Title: Identification of Candidate Parkinson Disease Genes by Integrating Genome-Wide Association Study, Expression, and Epigenetic Data Sets.
Title: Identification of Candidate Parkinson Disease Genes by Integrating Genome-Wide Association Study, Expression, and Epigenetic Data Sets.
Authors: Kia DA, Zhang D, Guelfi S, Manzoni C, Hubbard L, Reynolds RH, Botía J, Ryten M, Ferrari R, Lewis PA, Williams N, Trabzuni D, Hardy J, Wood NW; United Kingdom Brain Expression Consortium (UKBEC) and the International Parkinson’s Disease Genomics Consortium (IPDGC)
Journal: JAMA Neurol. 2021 Apr 1;78(4):464-472.
PMID: 33523105 (This report is OPEN ACCESS if you would like to read it)
This study identified five genes with changes in total levels of activity that were associated with Parkinson’s risk. Those genes were WDR6, CD38, TMEM163, RAB29, and GPNMB.
And the increase in GPNMB levels has also been reported in studies doing more thorough examinations of brain tissue from people with Parkinson’s:
 Title: The glycoprotein GPNMB is selectively elevated in the substantia nigra of Parkinson’s disease patients and increases after lysosomal stress.
Title: The glycoprotein GPNMB is selectively elevated in the substantia nigra of Parkinson’s disease patients and increases after lysosomal stress.
Authors: Moloney EB, Moskites A, Ferrari EJ, Isacson O, Hallett PJ.
Journal: Neurobiol Dis. 2018 Dec;120:1-11.
PMID: 30149180 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers analysed GPNMB levels in three regions of the brain (the cerebellum, the caudate/putamen, and the substantia nigra. This last area is severely affected in Parkinson’s, and it is home to most of the dopamine-producing neurons in the brain. The investigators examined samples from 25 non-GBA varriant carriers who passed away with Parkinson’s and compared them with the substantia nigra from 30 control cases.
While they found no differences in GPNMB levels in the cerebellum (CB), the caudate/putamen (CP), they did observe a significant increase in GPNMB levels in the substantia nigra of the Parkinson’s cases:
 Source: PMC
Source: PMC
This research group have recently extended these results (Click here to read more about this).
And their finding has been replicated by independent researchers:
 Title: The glycoprotein GPNMB attenuates astrocyte inflammatory responses through the CD44 receptor.
Title: The glycoprotein GPNMB attenuates astrocyte inflammatory responses through the CD44 receptor.
Authors: Neal ML, Boyle AM, Budge KM, Safadi FF, Richardson JR.
Journal: J Neuroinflammation. 2018 Mar 8;15(1):73
PMID: 29519253 (This report is OPEN ACCESS if you would like to read it)
In this study, the investigators reported increased levels of GPNMB in the substantia nigra of postmortem brains of people who passed away with Parkinson’s (n=31, compared to 17 control cases).
The researchers also looked at what GPNMB could be functionally doing and they observed a potential neuroprotective effect: By adding GPNMB to cultured cells, there was a reduction in the levels of inflammatory markers and an increase in anti-inflammatory factors (like IGF-1 and arginase-1).
Interesting. So this potential biomarker GPNMB could be having a neuroprotective role. Has this ever been reported before?
Yes it has.
Title: Transgenic Overexpression of GPNMB Protects Against MPTP-Induced Neurodegeneration.
Authors: Budge KM, Neal ML, Richardson JR, Safadi FF.
Journal: Mol Neurobiol. 2020 Jul;57(7):2920-2933.
PMID: 32436108
In this study, the researchers used a genetically engineered mouse that produces high levels of GPNMB. The investigators exposed these mice to a neurotoxin (MPTP) that has classically been used to model Parkinson’s and they found that the high levels of GPNMB reduced levels of degeneration in the dopamine neurons.
And the neuroprotective potential of GPNMB has also been reported in models of amyotrophic lateral sclerosis (ALS/motor neuron disease – Click here to read more about this) and stroke (Click here to read more about that).
So GPNMB could be a biomarker and a neuroprotective treatment for neurodegeneration?
I am not saying that. A lot more research is required – particularly to determine what happens to GPNMB levels in GBA-associated Parkinson’s.
In addition, recent data indicates that cerebrospinal fluid levels of GPNMB could not distinguish between Alzheimer’s or controls (Click here to read more about this), so it is unlikely that GPNMB will be important in all neurodegenerative conditions.
But it is an interesting molecule.
So what does it all mean?
The identification of biomarkers would revolutionise research into neurodegenerative conditions, as they would provide a measure by which progression and novel disease modifying therapies could be assessed.
It is unlikely that pan-condition biomarkers will be identified (our brains and biology is slightly more complex than that), but by focusing on specific sub-types of diseases, maybe we will get lucky. Potentially genetic sub-types could be a good place to start as the individuals affected by a specific genetic variant could share a disruption to a common biological pathway.
The observation that GPNMB is so elevated in Gaucher disease is intriguing as a potential biomarker. And given the association between Gaucher disease and Parkinson’s, it could be worth investigating blood/CSF samples in some of the longitudinal GBA-associated PD cohorts that are being studied around the world.
It’s just an idea.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from FEBS




Hi. What about tavapadone the new guy in town?
LikeLike