|
We have previously discussed the importance of the right foods for people with Parkinson’s on this blog – Click here for a good example. Recently, new data from researchers in Sweden points towards the benefits of a specific component of fish in particular. It is a protein called β-parvalbumin, which has some very interesting properties. In today’s post, we discuss what beta-parvalbumin is, review the new research findings, and consider how this new information could be applied to Parkinson’s. |
A very old jaw bone. Source: Phys
In 2003, researchers found 34 bone fragments belonging to a single individual in a cave near Tianyuan, close to Beijing (China).
But it was not the beginning of a potential murder investigation.
No, no.
This was the start of something far more interesting.
Naming the individual “Tianyuan man”, the researchers have subsequently found that “many present-day Asians and Native Americans” are genetically related to this individual. His bones represented one of the oldest set of modern human remains ever found in the eastern Eurasia region.
Tianyuan caves. Source: Sciencemag
But beyond the enormous family tree, when researchers further explored specific details about his jaw bone (or lower mandible as it is called) they found something else that was very interesting about Tianyuan man:

Title: Stable isotope dietary analysis of the Tianyuan 1 early modern human.
Authors: Hu Y, Shang H, Tong H, Nehlich O, Liu W, Zhao C, Yu J, Wang C, Trinkaus E, Richards MP.
Journal: Proc Natl Acad Sci U S A. 2009 Jul 7;106(27):10971-4.
PMID: 19581579 (This research article is OPEN ACCESS if you would like to read it)
In this study, the investigators analysed the carbon and nitrogen isotopes found within bone collagen samples taken from the jaw bone of Tianyuan man. In humans, the carbon and nitrogen isotope values indicate the sources of dietary protein over many years of life.
The researchers found that a substantial portion of Tianyuan man’s diet 40,000 years ago came from freshwater fish.
Interesting preamble, but what does this have to do with Parkinson’s?
Well, eating fish is believed to have many beneficial effects against numerous age-related conditions.
From cardiovascular disease (consuming small quantities of fish is associated with a 30% risk reduction for heart attacks – Click here to read more) to dementia/Alzheimer’s (one study found that eating fish once a week or more results in a 60% lower risk for developing Alzheimer’s – Click here to read more about this), the consumption of fish is believed to have many benefits.
Source: Connectcolony
And while Tianyuan man probably ate fish simply because he was hungry, more recently researchers have been trying to identify exactly which components of the fish meat provide the beneficial effects to our health.
Many people have become very excited about one particular component, called omega-3 fatty acids.
It has widely been presented as a potential therapeutic measure against dementia. Despite the enthusiasm, however, remarkably little direct evidence for this exists in humans (Click here to read more about this).
Clinical studies have found no evidence for benefit (or harm) from omega-3Polyunsaturated fatty acids supplements in people with mild to moderate Alzheimer’s (Click here to read more about this). This hasn’t stopped the research continuing though, and there is currently a phase 2/3 clinical trial of docosahexaenoic acid – a specific omega-3 fatty acid – in Alzheimer’s (Click here to read more about that).
What about Parkinson’s and Omega-3?
In the case of Parkinson’s, there has also been a lot of pre-clinical research on omega-3 acids (Click here and here to read more about this, and click here for a review that has a good section on omega-3 fatty acids research in Parkinson’s as well as other food components), but – again – there are limited clinical results supporting the preclinical data.
Title: Depression in Parkinson’s disease: a double-blind, randomized, placebo-controlled pilot study of omega-3 fatty-acid supplementation.
Authors: da Silva TM, Munhoz RP, Alvarez C, Naliwaiko K, Kiss A, Andreatini R, Ferraz AC.
Journal: J Affect Disord. 2008 Dec;111(2-3):351-9.
PMID: 18485485
This study is the only randomised clinical trial on fish oil supplements in Parkinson’s (that I am aware of – happy to be corrected). In this pilot study, 31 individuals with Parkinson’s and depression were randomly assigned either fish oil capsules (“containing omega-3 fatty acids”) or mineral oil capsules for 12 weeks. At the end of that period, those who had been taking the fish oil capsules had statistically significant improvements in their levels of depression (compared to the mineral oil group), but they did not demonstrate any changes in their other Parkinson’s symptoms.
Fish oil capsules. Source: Indiamart
Recently a clinical trial (called ‘RLID-PD’ – Click here to find out more about that trial) has been conducted investigating the impact of docosahexaenoic acid on the development of L-dopa induced dyskinesias in Parkinson’s (Click here to find out more about dyskinesias). The preliminary results from that study suggest that for people just beginning L-dopa treatment, concomitant use of docosahexaenoic acid may help delay onset of L-dopa induced dyskinesias (Click here to see an early abstract of the results – NOTE: we are still waiting on the final results of the study).
Admittedly, both of these studies were a very small, and the depression study was too short (three months is not long enough to observe motor improvements), so it is difficult to draw too many conclusions.
And the lack of any strong clinical support for omega-3 fatty acids has not stopped researchers from focusing on other components of fish meat.
Which resulted in the research report we are going to review today:
Title: Abundant fish protein inhibits α-synuclein amyloid formation
Authors: Werner T, Kumar R, Horvath I, Scheers N, Wittung-Stafshede P
Journal: Sci Rep. 2018 Apr 3;8(1):5465.
PMID: 29615738 (This research article is OPEN ACCESS if you would like to read it)
In this study, a group of Swedish researchers wanted to look at the interactions between Atlantic cod β-parvalbumin and the Parkinson’s associated protein alpha synuclein.
What is β-parvalbumin?
Parvalbumins are proteins that represent one of the largest group of animal food allergens.
Allergens are proteins that triggers immune responses (or allergic reactions). For example, nuts are a common allergen, causing an immune response in people who are allergic to them.
Nuts. Source: Ronly
Parvalbumins are ubiquitous in animals – they are particularly abundant in the white muscle cells of many fish species. But Parvalbumins can be classified into two different types: alpha (α) and beta (β).
Structure of α-Parvalbumin (Pink) and β-Parvalbumin. Source: Wikipedia and Ebi
And the difference is important.
Most fish species are rich in β-parvalbumins – they contain about 0.2 g of β-parvalbumin per 100 g muscle tissue (that is quite a lot – source). And β-parvalbumins are the most allergenic (that is, they cause the biggest allergic reaction).
Seafood allergies are relatively common – 2.3% of the general population – but fish allergies specifically are much less common (only 0.4% of the population – Source). Allergic reactions to fish are mainly caused by parvalbumins, which are relatively heat stable (meaning that they are not destroyed by cooking the meat).
But why were the Swedish researchers looking at β-parvalbumin?
An interesting feature of parvalbumins is that they can bind together and form what is known as an amyloid state.
What is an amyloid state?
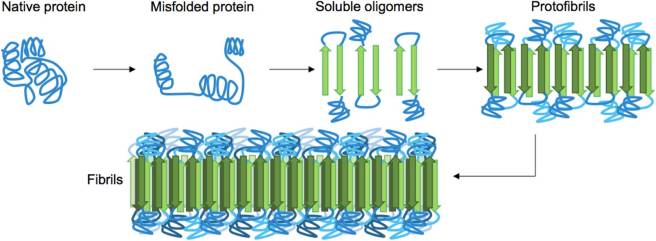
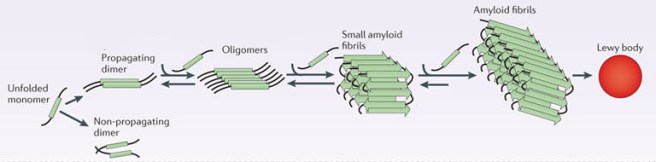
Amyloid states occur when a protein become folded in such a way that allows many copies of that particular protein to stick together. Quite often this clustering (or aggregation) occurs when the protein has been misfolded.
Small aggregates of these amyloid-proned proteins are called oligomers, and as they combine with other oligomers, they form what are referred to as fibrils.
Source: Spiedigitallibrary
In Parkinson’s, we see this property in the “bad guy” protein, alpha synuclein. It has this ability to form amyloid states, which results in the production of fibrils and (subsequently) the formation of Lewy bodies (the characteristic circular clumps of alpha synuclein that appear in cells in the Parkinsonian brain):
Parkinson’s associated alpha synuclein. Source: Nature
If you want to read more about amyloid state proteins (particularly in the context of disease), click here for a good review. And please don’t assume that amyloid clustering of proteins is only a disease-related phenomenon. There is a lot of evidence now of these amyloid-clusters of proteins having functional roles (Click here to read more about this).
And β-parvalbumin can form these amyloid states as well?
Yes it can.
There are actually a lot of proteins that can form amyloid states. In the case of β-parvalbumin, this amyloid state is believed to explain the allergic properties of the protein:
Title: Fish β-parvalbumin acquires allergenic properties by amyloid assembly.
Authors: Martínez J, Sánchez R, Castellanos M, Fernández-Escamilla AM, Vázquez-Cortés S, Fernández-Rivas M, Gasset M.
Journal: Swiss Med Wkly. 2015 May 29;145:w14128.
PMID: 26023765 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers found that forming an amyloid state protects β-parvalbumin from being broken down by stomach acid (after it has been consumed). The amyloid state also helped the protein to cross through the intestinal wall, and the amyloid formation resulted in the presentation of the epitope that makes β-parvalbumin an allergen.
Importantly, the researchers also report that the anti-amyloid compound epigallocatechin gallate (also known as EGCG) prevented β-parvalbumin forming an amyloid state, AND reduced the presentation of the epitope. We have previously discussed the potentially magical powers of EGCG in the context of Parkinson’s (Click here to read that post).
Ok, so what did the Swedish researchers in this new study do?
Knowing that human amyloid proteins can interact with each other, the researchers were wondering whether – once fish meat is consumed and absorbed into the host blood stream – fish β-parvalbumin may also interact with some of the human amyloid proteins.
To test this idea, the researchers collected Atlantic cod β-parvalbumin and tested it in a host of experiments with human alpha synuclein.
Atlantic cod. Source: Wikipedia
Through their experiments, the researchers discovered that β-parvalbumin robustly inhibits amyloid formation of alpha synuclein. It blocks the formation of alpha synuclein aggregates completely.
But it was the way that it did this that was particularly interesting.
The investigators found that the anti-amyloid effect involved alpha synuclein actually binding to the β-parvalbumin amyloid structures. In this fashion, β-parvalbumin “scavenges” alpha synuclein, binding it up so that it cannot form the aggregates.
What is the normal function of β-parvalbumin in the cell?
Parvalbumin is a calcium-binding albumin protein.
Calcium? Again?
I am assuming that you are referring to the string of recent research reports dealing with calcium (Click here and here to read SoPD posts on these reports).
Yes, calcium is currently a big deal in the world of Parkinson’s research.
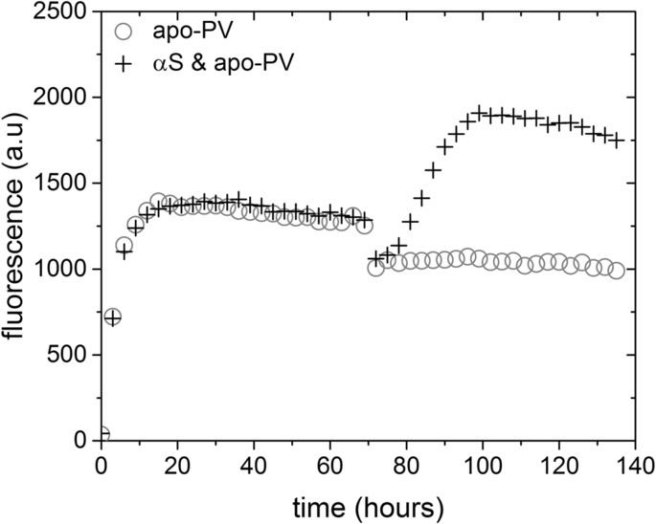
And in this current study, the researchers in Sweden found that β-parvalbumin adopted a very stable amyloid state in the absence of calcium. As soon as they added calcium to their experiments, β-parvalbumin would stop forming amyloid structures.
Importantly, the addition of calcium and the loss of β-parvalbumin amyloid structures resulted in the increase of alpha synuclein amyloid aggregates. As you can see in the image below, when β-parvalbumin and alpha synuclein were maintained in a low calcium solution they did not aggregate much over time. But as soon as the investigators added calcium to the solution (at 70 hours), there was an increase in alpha aggregation (the upward curve in crosses).
Source: Nature
This result suggested that in order for β-parvalbumin to have its anti-amyloid effect on alpha synuclein, their interaction needs to be in a low calcium environment.
But given this anti-amyloid effect β-parvalbumin has on alpha synuclein, the researchers concluded that “fish intake may provide health benefits through PV amyloid interactions that prevent neurodegenerative processes”
The researchers even provided some dietary advice: “Fish is normally a lot more nutritious at the end of the summer, because of increased metabolic activity. Levels of parvalbumin are much higher in fish after they have had a lot of sun, so it could be worthwhile increasing consumption during autumn,” – Nathalie Scheers, Assistant Professor in the Department of Biology and Biological Engineering, and a researcher on the study (Source).
Interesting. Is there any evidence of a high-fish diet helping with Parkinson’s?
This is difficult to answer as fish is not eaten in isolation. There has, however, been some research looking at diet and the risk of developing Parkinson’s, and it puts fish into the groups of food that are associated with a lower risk of developing Parkinson’s:
Title: Prospective study of dietary pattern and risk of Parkinson disease.
Authors: Gao X, Chen H, Fung TT, Logroscino G, Schwarzschild MA, Hu FB, Ascherio A.
Journal: Am J Clin Nutr. 2007 Nov;86(5):1486-94.
PMID: 17991663 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers conducted a prospective study involving the medical details of 49 692 men and 81 676 women from two huge US databases: the Nurses’ Health Study (NHS) and the Health Professionals Follow-Up Study (HPFS).
NHS study was started in 1976 when 121,700 female registered nurses (aged 30 to 55 years) completed a mailed questionnaire. They provided an overview of their medical histories and health-related behaviours. The HPFS study was established in 1986, when 51,529 male health professionals (40 to 75 years) responded to a similar questionnaire. Both the NHS and the HPFS send out follow-up questionnaires every 2 years.
By analysing all of that data, the investigators found 508 cases of Parkinson’s. When they looked at the diets of these individuals, the researchers found that a high intake of fruit, vegetables, fish, and poultry and a low intake of saturated fat lowered the risk of developing Parkinson’s. Exactly how much impact the fish component had on this effect is still to be determined.
So does this mean I should eat more fish?
Not necessarily.
With regards to the β-parvalbumin study, the results need to be independently before we get too carried away. In addition, as Dr Beckie Port of Parkinson’s UK pointed out in her discussion of this research, ‘we don’t yet know if [β-parvalbumin] can cross the blood brain barrier to enter the brain’ (the blood brain barrier being the protective membrane that surrounds our brain).
Plus radically increasing the amount of fish in the diet of a person with Parkinson’s is not really a good idea, particularly if they are taking L-dopa.
Que?
Good food is important.
Particularly for an individual affected by Parkinson’s.
But too much foods that are high in protein should be avoided or eaten in moderation if you have Parkinson’s. And fish is high in protein.
The reason for this is because L-dopa is an amino acid, and protein competitively inhibits L-dopa absorption. This competition reduces the effectiveness of L-dopa therapy (Click here to read more about this).
Thus, one should be careful eating too much fish.
I have fish allergies, how can I get some of this β-parvalbumin stuff?
Again, the results need to be independently replicated.
But while we are waiting for those results, drink green tea. It is full of the anti-amyloid compound epigallocatechin gallate (EGCG – Click here to read more about this).
Source: BBC
So what does it all mean?
Researchers in Sweden have published the results of a very interesting study that highlights the anti-amyloid effect of fish-derived β-parvalbumin on aggregating alpha synuclein. Their findings indicate that the β-parvalbumin achieves its effect by scavenging alpha synuclein, and this phenomenon is dependent on low calcium levels.
One important consideration when interpreting the results of this new research is that all of the experiments were conducted in solutions, not in cells. Thus, there is a question of how biologically relevant the anti-amyloid properties of β-parvalbumin are – particularly in the high calcium environment of a neuron.
It will thus be interesting to see if the results can be replicated in cells, and hopefully such experiments will include other amyloid-proned proteins like beta-amyloid (Alzheimer’s) and amylin (type-2 diabetes). The Swedish researchers have already indicated that they are certainly planning to follow up the research. And they have suggested that that follow up work will also include investigations of how β-parvalbumin is distribute within human tissues after consumption (Source).
We shall see what happens. And until those results become available, I’ll try not to be koi.
Not a bad pun. Cod do batter though.
The banner for today’s post was sourced from 24-hrbait













Simon,
Once again thank you for such an interesting post.
Sanity check. On the results presented we would expect an association between fish consumption (high) and PD prevalence (low).
Fish consumption in Portugal, the highest in the EU in 2013, was 53.8kg and the PD death-rate/100000/year, age standardized, was 3.66/100000. For Hungary, the lowest in the EU, it was 5.1kg and the death-rate was 1.76/100000.
For fish consumption, see http://neweconomics.org/wp-content/uploads/2017/03/NEF_Fish_Dependence_2017_2.pdf
For a measure of Parkinson’s see http://www.worldlifeexpectancy.com/cause-of-death/parkinson-disease/by-country/
Of course, this contrary result could reflect poor quality data, genetics or the specifics of the food preparation, rather than fish consumption per se, or other confounding features such as the fish species used, and reporting limitations, but it needs consideration.
John
LikeLike
Hi John,
Glad you liked the post. I think it is very difficult to assess the impact of eating fish has on PD (at both the population and individual level). The Japanese eat a lot of fish and have a lower rate of PD, but they also drink a lot of EGCG-filled green tea. It is very difficult to view one component in isolation. Plus there is wide variations in levels of beta-P and omega 3 in different species of fish. And on top of that there is also the concerns about metals, toxins etc in fish (though I doubt the levels are high enough to really have a major impact). Thus, I really struggle with the prevalence data.
Thanks again.
Kind regards,
Simon
LikeLike
thanks
LikeLike
John,
you nailed a question that is in my mind – what do the epidemiological data say about this?
I wonder though if global mortality statistics are a reliable proxy for the incidence of idiopathic PD ? – with regional variance in under-diagnosis, and the different ways in which cultures handle/respond to death. Can we get “non-death” clinical incidence data instead?
Also I’m also wondering about the potential for longer-term cross-cultural comparisons – could we compare (over time) clinical diagnosis incidence data for say Japan (a culture that has until recently relied primarily on fish protein) vs landlocked south-east asian countries (avoiding comparison with Westernized cultures due to the plethora of confounding variables). Or how about regional comparisons – coastal provinces of India vs inland provinces?
LikeLike
Hi Alex,
Thanks for your comment. Like I said in my response to John’s comment, I have a hard time with these sorts of comparisons. Having said that, I’m sure in this modern age of data, that the information you are suggesting will be available somewhere. It is simply a case of finding it I guess. But unfortunately I for one am not sure where to start looking.
Kind regards,
Simon
LikeLike
Dkdc – You’re welcome
LikeLike
I think Laurie Mischleys epidemiological data supports eating fish. I don’t remember how strongly and I am not trained to read such data and represent it accurately.
LikeLike
Hi Dkdc,
You are right. Fish oil was one particular component of diet that was associated with a lower rate of progression in Parkinson’s. I remember writing about it in the post we did on Laurie’s research a while back (https://scienceofparkinsons.com/2017/09/18/food/). Thanks for mentioning it.
Kind regards,
Simon
LikeLike
Simon,
Thank you for yet another informative article! OK – I’ll order the fish more often.
I’d like take exception however to a recommendation you make in this post (noting that you usually assiduously avoid making such admonitions) – but this is the second time in recent times that you advise people with Parkinson’s to moderate or avoid consumption of high-protein foods. Of course I understand that this is “boilerplate” in the L-dopa prescribing guidelines, and often repeated by clinicians – however, a critical examination of the paper you cite (Virmani et. al., 2016) hardly supports such a recommendation. The conclusions in that paper specifically claim applicability to a relatively narrow population of patients – and even there, I have serious concerns about the reliability of the underlying clinical data. I’d want to take a poll of PWPs and ask how many of them note a consistent decrease in L-dopa dose effectiveness when taking the medication within a short time-frame of consuming a high-protein meal (vs. the corollary). I wonder if one would truly be able to detect an effect of “high protein” distinct from any other meal. And given that this guidance is “out there”, even then I’d bee looking for a “nocebo” effect.
The reason I’m pushing back hard on this is that the choice to reduce or avoid protein has many implications quite aside from possible interactions with anti-parkinsonian medication. Among them for example, protein is necessary for building and maintaining muscle, and for supporting a body that exercises regularly (see where I’m going with this?). In addition, avoiding high-protein will often concomitantly reduce the intake of saturated fats (many will say that’s a good thing) but I urge them to reread recent data on the benefits of saturated fats (in moderation) – let’s not forget that cholesterol and other fats are quite literally the energy source for brain cells. Lastly a “low-protein” diet will often emphasize intake of carbohydrates – and in Western cultures in particular- that will entail reliance on grains, particularly wheat. While I do know that you (Simon) and I are overdue a longer discussion of the state of knowledge regarding gluten and its possible implications in neurodegenerative disease (that is for another time), I think it is generally acknowledged that a diet high in grains is not ideal either (all sorts of potential for metabolic dissregulation going all the way up to diabetes).
For now I think we’d agree that the key (as in so many things) is “moderation”, and most importantly that it is up to the individual patient to discuss with their doctor what the ideal diet should be. I do suggest however, if hearing guidance to “avoid” protein, they might want to clarify what that recommendation is based on, and particularly to verify if it is more correctly gudance to avoid taking l-dopa and high-protein foods within 30 minutes of each other. This distinction is important, because it allows for consumption of (perhaps necessary/useful/beneficial) protein at other times outside of the 30-minute timeframe of oral administration of medication.
LikeLike
Hi Alex,
Touche! You have caught me there. Several times I have mentioned on this site the ‘fact’ about being cautious with regards to high protein diets, and I have never actually questioned it or looked into it in any depth. It is definitely a topic for a future post (one to add to the list). Protein does competitively inhibit L-dopa absorption, but as you wisely point out this is not a reason to cut back on protein. Everything in moderation – agreed.
And I have been asked by several readers to do a post on glutens so I will look forward to our discussion on that topic.
Thanks for the great comment.
Kind regards,
Simon
LikeLike
Can beta-Parvalbumin inhibit or excrete the aggregation of synuclen in endoplasmic reticulum ?
LikeLike
This is a great website for ideas for rock band names. “endoplasmic reticulum” will be my next band ! – Just a little levity that I hope is not obtrusive.
LikeLike
Ha! I’ll go for “Metaiodobenzylguanidine scintigraphy”! Levity is good 🙂
LikeLike
I’m not sure. And I’m not sure if anyone knows. It’s a really good question though – a lot of research attention is now focused on the ER in PD (https://scienceofparkinsons.com/2018/05/03/berserker/). I will look into it.
Kind regards,
Simon
LikeLike